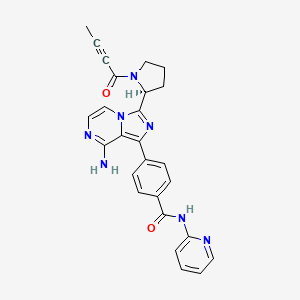
Acalabrutinib
Overview
Description
Acalabrutinib is a small molecule inhibitor of Bruton tyrosine kinase, used primarily in the treatment of mantle cell lymphoma, chronic lymphocytic leukemia, and small lymphocytic lymphoma . It is marketed under the brand name Calquence and was developed to be more potent and selective than its predecessor, ibrutinib .
Preparation Methods
Synthetic Routes and Reaction Conditions: Acalabrutinib is synthesized through a multi-step process involving the formation of key intermediates. The synthesis begins with the preparation of 4-(8-amino-3-(1-but-2-ynoylpyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-N-(pyridin-2-yl)benzamide . The reaction conditions typically involve the use of solvents like acetonitrile and reagents such as potassium dihydrogen phosphate .
Industrial Production Methods: The industrial production of this compound involves large-scale synthesis using high-performance liquid chromatography for purification . The process is optimized to ensure high yield and purity, with stringent quality control measures in place .
Chemical Reactions Analysis
Covalent Binding Mechanism to BTK
Acalabrutinib forms an irreversible covalent bond with C481 in BTK through a Michael addition reaction facilitated by its 2-butynamide moiety . The reaction proceeds via:
- Proton transfer-nucleophilic attack (PT-NA): The thiol group of C481 attacks the β-carbon of the 2-butynamide group.
- Tautomerization: Formation of a stable enol intermediate, followed by irreversible conversion to the keto form .
Key Findings:
- Water-assisted mechanism: Ab initio simulations confirm that a water molecule in BTK’s active site reduces the reaction barrier by stabilizing the transition state .
- Free energy barriers:
This difference in activation barriers explains this compound’s ≥80-fold selectivity for BTK over interleukin-2-inducible T-cell kinase (ITK) .
Metabolic Reactions
This compound undergoes CYP3A4-mediated oxidation and glutathione conjugation to form metabolites :
| Metabolic Pathway | Major Metabolites | Activity Relative to Parent Drug |
|---|---|---|
| CYP3A4 oxidation | ACP-5862 (major) | 50% potency for BTK inhibition |
| Glutathione conjugation | M25 (minor) | Inactive |
- ACP-5862 accounts for 34.7% of recovered drug mass, with a half-life of 6.9 hours .
- Elimination: 84% fecal, 12% renal .
Reaction Kinetics and Selectivity
Kinetic simulations using coupled differential equations reveal:
- BTK inhibition: 97–99% occupancy at steady state with 100 mg twice-daily dosing .
- Off-target reactivity: Minimal inhibition of Src-family kinases (IC₅₀ >1 μM) due to the 2-butynamide group’s lower electrophilicity compared to ibrutinib’s acrylamide .
Comparison with Ibrutinib:
| Parameter | This compound | Ibrutinib |
|---|---|---|
| BTK IC₅₀ (nM) | 3.0 | 0.5 |
| ITK IC₅₀ (nM) | 800 | 10 |
| T-cell kinase inhibition | None | LCK/SRC inhibition |
| Source: Biochemical assays |
Solubility and Stability
- pH-dependent solubility: Highly soluble at acidic pH (e.g., gastric fluid) but poorly soluble at neutral pH .
- Degradation pathways: Hydrolysis of the 2-butynamide group under alkaline conditions .
Reaction-Driven Design Advantages
This compound’s optimized reactivity profile addresses limitations of first-generation BTK inhibitors:
- Reduced off-target effects: The 2-butynamide group’s lower intrinsic reactivity minimizes covalent binding to non-target cysteines (e.g., EGFR, ITK) .
- Enhanced pharmacokinetics: Rapid absorption (Tₘₐₓ: 0.75 hours) and short half-life (0.9 hours) enable twice-daily dosing without cumulative toxicity .
Scientific Research Applications
Chronic Lymphocytic Leukemia (CLL)
Acalabrutinib has been extensively studied in patients with CLL, particularly those who are intolerant to ibrutinib or have relapsed disease. Key findings include:
- Efficacy : In a cohort of 33 patients intolerant to ibrutinib, this compound demonstrated an overall response rate of 79% . A systematic review involving six clinical trials with 808 patients reported a pooled overall response rate of approximately 85%, indicating robust efficacy .
- Safety Profile : Compared to first-generation BTK inhibitors like ibrutinib, this compound has a more favorable safety profile, with reduced risks of atrial fibrillation and other adverse events .
- Combination Therapies : Ongoing studies are evaluating the efficacy of this compound in combination with other therapies, including immunomodulatory drugs, to enhance treatment outcomes .
Mantle Cell Lymphoma (MCL)
This compound is also indicated for MCL, where it has shown significant clinical activity:
- Clinical Trials : In clinical settings, this compound has been effective in both treatment-naive and relapsed/refractory MCL patients. The Phase II trial results indicated a high overall response rate and manageable toxicity .
- Long-Term Outcomes : Data suggest that this compound may improve progression-free survival rates compared to traditional therapies .
Other B-cell Malignancies
Research is ongoing into the use of this compound for other hematological malignancies:
- Waldenström Macroglobulinemia : Preliminary studies indicate potential efficacy in this condition, with ongoing trials assessing its role .
- Diffuse Large B-cell Lymphoma (DLBCL) : Trials are exploring the use of this compound as part of combination regimens for DLBCL, although results are still emerging .
COVID-19
Recent studies have investigated the potential use of this compound in managing severe respiratory syndrome associated with COVID-19. Its immunomodulatory effects may help mitigate hyper-inflammatory responses observed in severe cases . Early data suggest possible benefits in reducing cytokine storms, although further research is required to establish its efficacy in this context.
Case Studies
A selection of notable case studies highlights the effectiveness and safety of this compound:
Mechanism of Action
Comparison with Similar Compounds
Ibrutinib: The first-generation Bruton tyrosine kinase inhibitor.
Zanubrutinib: Another second-generation Bruton tyrosine kinase inhibitor.
Comparison: Acalabrutinib is more selective and has fewer off-target effects compared to ibrutinib . It also demonstrates a lower rate of adverse events such as atrial fibrillation and hypertension compared to zanubrutinib .
This compound stands out due to its improved safety profile and higher selectivity, making it a preferred choice for many patients .
Biological Activity
Acalabrutinib is a second-generation Bruton’s tyrosine kinase (BTK) inhibitor primarily used in the treatment of chronic lymphocytic leukemia (CLL) and other B-cell malignancies. Its development was driven by the need for more selective therapies with improved safety profiles compared to first-generation BTK inhibitors like ibrutinib. This article reviews the biological activity of this compound, highlighting its mechanism of action, pharmacokinetics, clinical efficacy, and safety profile based on diverse research findings.
This compound works by covalently binding to cysteine 481 in the BTK active site, inhibiting BTK enzymatic activity. This inhibition disrupts downstream signaling pathways crucial for B-cell proliferation and survival, particularly in malignant B-cells. The selectivity of this compound for BTK over other kinases minimizes off-target effects, which is a significant advantage over ibrutinib .
Pharmacokinetics
- Absorption: this compound has a geometric mean absolute bioavailability of approximately 25%, with peak plasma concentrations reached within 0.75 hours post-administration.
- Distribution: The mean steady-state volume of distribution is around 34 L, indicating extensive tissue distribution.
- Protein Binding: It exhibits high protein binding (approximately 97.5%) to human plasma proteins.
- Metabolism: this compound is primarily metabolized by CYP3A4, and its active metabolite, ACP-5862, contributes to its pharmacological effects .
Clinical Efficacy
The efficacy of this compound has been demonstrated in multiple clinical trials involving patients with CLL and other hematological malignancies. Key findings from significant studies are summarized in the table below.
| Study | Patient Population | Dosage | Overall Response Rate (ORR) | Complete Response Rate (CR) | Duration of Response (DOR) |
|---|---|---|---|---|---|
| ACE-CL-001 | Treatment-naive CLL | 100 mg twice daily | 97% | 7% | Not reached (48-month DOR rate: 97%) |
| Phase II Study (ibrutinib-intolerant) | Relapsed/refractory CLL | 100 mg twice daily | 61% | Not reported | Median PFS not reached |
| ACE-LY-003 | Relapsed/refractory marginal zone lymphoma (MZL) | 100 mg twice daily | 52.2% | 12.5% | Median PFS: 27.4 months |
Safety Profile
This compound is generally well-tolerated, but it does have associated adverse events (AEs). In the pivotal studies:
- Common AEs: Hypertension (22%), atrial fibrillation (5%), infections (reported in 57% of patients), and bleeding events.
- Serious AEs: Occurred in about 38% of patients, with some requiring treatment discontinuation due to second primary cancers or severe infections .
Case Studies
- Chronic Lymphocytic Leukemia : In a phase 1/2 trial involving previously untreated CLL patients, this compound demonstrated a high response rate and durable remissions over a median follow-up of four years. This study highlighted the drug's effectiveness in achieving significant tumor reduction while maintaining a manageable safety profile .
- Marginal Zone Lymphoma : Another study focused on patients with relapsed/refractory MZL showed that this compound monotherapy resulted in an ORR of 52.2%, indicating its potential as a therapeutic option for this patient population .
Q & A
Basic Research Questions
Q. What are the standard experimental protocols for assessing acalabrutinib’s BTK inhibition efficacy in preclinical models?
- Methodological Answer : Use kinase activity assays (e.g., ADP-Glo™) to quantify BTK inhibition in cell-free systems. For cellular models, employ B-cell lymphoma lines (e.g., TMD8) with phospho-BTK (Y223) Western blotting to confirm target engagement. Dose-response curves should be generated using serial dilutions (e.g., 0.1–1000 nM) to determine IC₅₀ values .
Q. How do researchers validate the specificity of this compound against off-target kinases?
- Methodological Answer : Utilize kinome-wide profiling (e.g., KINOMEscan®) to assess selectivity across >400 kinases. Follow-up with functional assays (e.g., calcium flux in T-cells for ITK inhibition) to confirm lack of off-target effects. Cross-reference results with published selectivity profiles from clinical trials (e.g., ELEVATE-TN) .
Q. What statistical models are recommended for analyzing time-to-progression data in this compound clinical trials?
- Methodological Answer : Apply Kaplan-Meier survival analysis with log-rank tests for unadjusted comparisons. For adjusted analyses, use Cox proportional hazards models incorporating covariates (e.g., TP53 mutation status, prior therapies). Address non-proportional hazards via time-dependent covariates or stratified models .
Advanced Research Questions
Q. How can researchers resolve contradictions in long-term efficacy data between this compound and comparator therapies (e.g., ibrutinib)?
- Methodological Answer : Conduct meta-analyses of pooled trial data (e.g., ELEVATE-TN, ASCEND) using random-effects models to account for heterogeneity. Validate findings with real-world evidence (RWE) from registries (e.g., Flatiron Health). Sensitivity analyses should test assumptions about progression rates and treatment crossover effects .
Q. What experimental designs are optimal for studying this compound resistance mechanisms in chronic lymphocytic leukemia (CLL)?
- Methodological Answer : Use longitudinal patient-derived xenograft (PDX) models or CRISPR-Cas9 mutagenesis screens to identify mutations (e.g., BTK C481S, PLCG2). Validate via single-cell RNA sequencing to detect clonal evolution. Pair with phosphoproteomics to map adaptive signaling pathways (e.g., PI3K/AKT) .
Q. How should researchers address uncertainties in economic models comparing this compound to chemoimmunotherapy?
- Methodological Answer : Incorporate probabilistic sensitivity analysis (PSA) to model parameter uncertainty (e.g., progression-free survival, drug costs). Use partitioned survival models with state transitions aligned with trial endpoints (e.g., ELEVATE-TN). Validate against alternative distributions (e.g., generalized gamma vs. exponential) for extrapolation .
Q. What frameworks ensure rigor in formulating research questions about this compound’s combination therapies?
- Methodological Answer : Apply PICO (Population: CLL patients; Intervention: this compound + venetoclax; Comparison: monotherapy; Outcome: MRD negativity) or FINER (Feasible, Interesting, Novel, Ethical, Relevant) criteria. Align hypotheses with preclinical synergy data (e.g., enhanced apoptosis in co-culture assays) .
Q. Data Analysis & Reproducibility
Q. What steps ensure reproducibility in this compound pharmacokinetic/pharmacodynamic (PK/PD) studies?
- Methodological Answer : Standardize bioanalytical methods (e.g., LC-MS/MS for plasma concentration). Report PK parameters (AUC, Cmax, t₁/₂) with variability metrics (CV%). Share raw data and analysis scripts via repositories (e.g., Dryad) .
Q. How can researchers mitigate bias in observational studies of this compound’s cardiovascular safety profile?
- Methodological Answer : Use propensity score matching to balance covariates (e.g., age, hypertension). Apply competing risk regression (Fine-Gray model) to account for non-cardiac deaths. Validate with adjudicated endpoint data from RCTs .
Q. Translational Research
Q. What biomarkers are critical for stratifying patients in this compound trials?
- Methodological Answer : Prioritize BTK occupancy (flow cytometry), serum B-cell cytokines (e.g., CXCL13), and genomic markers (e.g., del(17p)). Validate in multicenter cohorts using harmonized assays .
Q. How can multi-omics data be integrated to predict this compound response heterogeneity?
Properties
IUPAC Name |
4-[8-amino-3-[(2S)-1-but-2-ynoylpyrrolidin-2-yl]imidazo[1,5-a]pyrazin-1-yl]-N-pyridin-2-ylbenzamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C26H23N7O2/c1-2-6-21(34)32-15-5-7-19(32)25-31-22(23-24(27)29-14-16-33(23)25)17-9-11-18(12-10-17)26(35)30-20-8-3-4-13-28-20/h3-4,8-14,16,19H,5,7,15H2,1H3,(H2,27,29)(H,28,30,35)/t19-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
WDENQIQQYWYTPO-IBGZPJMESA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC#CC(=O)N1CCCC1C2=NC(=C3N2C=CN=C3N)C4=CC=C(C=C4)C(=O)NC5=CC=CC=N5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC#CC(=O)N1CCC[C@H]1C2=NC(=C3N2C=CN=C3N)C4=CC=C(C=C4)C(=O)NC5=CC=CC=N5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C26H23N7O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID401026209 | |
| Record name | Acalabrutinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID401026209 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
465.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Freely soluble in water at pH values below 3 but is practically insoluble in water at pH values above 6 | |
| Record name | Acalabrutinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11703 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Mantle Cell Lymphoma (MCL) is a rare yet aggressive type of B-cell non-Hodgkin lymphoma (NHL) with poor prognosis. Subsequently, relapse is common in MCL patients and ultimately represents disease progression. Lymphoma occurs when immune system lymphocytes grow and multiply uncontrollably. Such cancerous lymphocytes may travel to many parts of the body, including the lymph nodes, spleen, bone marrow, blood, and other organs where they can multiply and form a mass(es) called a tumor. One of the main kinds of lymphocytes that can develop into cancerous lymphomas are the body's own B-lymphocytes (B-cells). Bruton Tyrosine Kinase (BTK) is a signalling molecule of the B-cell antigen receptor and cytokine receptor pathways. Such BTK signaling causes the activation of pathways necessary for B-cell proliferation, trafficking, chemotaxis, and adhesion. Acalabrutinib is a small molecule inhibitor of BTK. Both acalabrutinib and its active metabolite, ACP-5862, act to form a covalent bond with a cysteine residue (Cys481) in the BTK active site, leading to inhibition of BTK enzymatic activity. As a result, acalabrutinib inhibits BTK-mediated activation of downstream signaling proteins CD86 and CD69, which ultimately inhibits malignant B-cell proliferation and survival Whereas ibrutinib is typically recognized as the first-in-class BTK inhibitor, acalabrutinib is considered a second generation BTK inhibitor primarily because it demonstrates highter selectivity and inhibition of the targeted activity of BTK while having a much greater IC50 or otherwise virtually no inhibition on the kinase activities of ITK, EGFR, ERBB2, ERBB4, JAK3, BLK, FGR, FYN, HCK, LCK, LYN, SRC, and YES1. In effect, acalabrutinib was rationally designed to be more potent and selective than ibrutinib, all the while demonstrating fewer adverse effects - in theory - because of the drug's minimized off target effects. | |
| Record name | Acalabrutinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11703 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
1420477-60-6 | |
| Record name | 4-[8-Amino-3-[(2S)-1-(1-oxo-2-butyn-1-yl)-2-pyrrolidinyl]imidazo[1,5-a]pyrazin-1-yl]-N-2-pyridinylbenzamide | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1420477-60-6 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Acalabrutinib [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1420477606 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Acalabrutinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11703 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Acalabrutinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID401026209 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | ACALABRUTINIB | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/I42748ELQW | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















