Dabrafenib
Overview
Description
Dabrafenib is a kinase inhibitor used primarily in the treatment of cancers associated with mutations in the BRAF gene, such as melanoma, non-small cell lung cancer, and thyroid cancer . It is marketed under the brand name Tafinlar and was first approved for medical use in the United States in May 2013 .
Preparation Methods
The synthesis of dabrafenib involves several key steps, including sulfamidation, halogenation, thiazole cyclization, acylation, and pyrimidine cyclization . The process begins with 3-(3-amino-2-fluorophenyl)-3-oxa-propionate as the starting material. This compound undergoes a series of reactions to form the final product, this compound . Industrial production methods are designed to be concise and mild, making them suitable for large-scale manufacturing .
Chemical Reactions Analysis
Synthetic Methods
The synthesis of Dabrafenib involves the construction of a 1,3-thiazole ring. This is typically achieved through the direct ring closure of thioamide (as a 1,3-binuclear reagent) with an α-carbonyl halide (as a 1,2-amphiphilic reagent) .
Key steps in this compound synthesis:
- Sulfonyl chloride and aniline react under basic conditions to yield sulfonamide .
- Methyl pyrimidine reacts with sulfonamide .
- The product undergoes α-bromination with N-bromosuccinimide (NBS) to yield a 1,2-amphiphilic reagent .
- The 1,2-amphiphilic reagent reacts with 1,3-parent nucleotides to close the ring, forming an intermediate .
- The intermediate reacts with ammonia to produce this compound .
A preparation method of this compound involves taking 3-(3-amino-2-fluorophenyl)-3-oxa-propionate as a starting material .
Metabolism
This compound undergoes metabolism primarily through oxidation of the t-butyl group, forming hydroxy-dabrafenib, which is further oxidized to carboxy-dabrafenib . Subsequent decarboxylation produces desmethyl-dabrafenib via a pH-dependent decarboxylation .
Metabolic Reactions:
- This compound is metabolized via cytochrome P450 isoenzymes CYP3A4 and CYP2C8 .
- The major route of elimination of this compound involves a combination of oxidative metabolism (48% of the dose) and biliary excretion .
- Hydroxy-dabrafenib has a twofold higher potency as an inhibitor of mutant BRAF .
- Desmethyl-dabrafenib is metabolized by CYP3A4 to oxidative metabolites .
Other Chemical Reactions
- This compound can be directly and ATP-competitively bound to RIP3 protein, causing selective inhibition on RIP3 over RIP1, RIP2, and RIP5 .
- This compound decreases TSZ-caused-RIP3-mediated Ser358 phosphorylation of MLKL and disrupts the interaction between RIP3 and MLKL .
- This compound induces cytochrome P450 isoenzyme (CYP) 3A4-mediated metabolism and may induce other enzymes, including CYP2B6, CYP2C8, CYP2C9, and CYP2C19 .
- This compound inhibits cell proliferation through an initial G1 cell cycle arrest, followed by cell death .
Impurity Formation
During laboratory optimization, an unknown impurity at RRT ~ 0.94 was observed in the final this compound product when chloro pyrimidine intermediate is converted to this compound . In that aromatic fluoro group . Phenyl amino this compound impurity can be formed when chloro pyrimidine intermediate is reacted with excess ammonium hydroxide .
Scientific Research Applications
Treatment of Melanoma
Monotherapy for Metastatic Melanoma
Dabrafenib is approved as a monotherapy for unresectable or metastatic melanoma in patients with BRAF V600E mutations. The approval was based on clinical trials demonstrating improved response rates and survival benefits compared to traditional therapies. For instance, a pivotal study showed that patients treated with this compound had a median overall survival of 25.5 months compared to 18.2 months for those receiving standard care .
Combination Therapy with Trametinib
The combination of this compound with trametinib, a MEK inhibitor, has been shown to enhance therapeutic efficacy. In a randomized trial involving 423 patients, the combination therapy achieved an overall response rate of 67%, significantly higher than the 51% observed with this compound alone . Furthermore, the combination resulted in longer progression-free survival (9.3 months vs. 8.8 months) and improved six-month survival rates (93% vs. 85%) compared to this compound monotherapy .
Adjuvant Therapy
This compound combined with trametinib has also been investigated as an adjuvant therapy for patients with resected stage III melanoma with BRAF V600 mutations. A study involving 870 patients showed that this combination led to a 25% reduction in the risk of death compared to placebo after a follow-up period of nearly ten years . The median relapse-free survival was significantly better in the treatment group (hazard ratio for relapse or death: 0.52) compared to placebo .
Tissue-Agnostic Indications
This compound's application has expanded beyond melanoma to include various solid tumors with BRAF V600 mutations, regardless of tissue origin. This includes cancers such as non-small cell lung cancer and anaplastic thyroid cancer. The FDA has granted tissue-agnostic approval based on data from basket trials that demonstrated consistent response rates across different tumor types . For example, in the NCI-MATCH study, this compound plus trametinib showed an overall response rate of approximately 38% in rare cancers harboring BRAF mutations .
Case Study: Advanced Melanoma
In a clinical trial assessing the efficacy of this compound plus trametinib in previously untreated advanced melanoma patients, researchers found that this combination not only improved response rates but also enhanced quality of life metrics among participants .
Research Findings: Long-term Outcomes
Long-term follow-up studies have indicated that approximately one-third of patients receiving first-line treatment with this compound plus trametinib experience sustained benefits, highlighting the potential for durable responses in this patient population .
Summary Table: Efficacy of this compound in Various Applications
| Application | Study Type | Overall Response Rate | Median Progression-Free Survival | Notes |
|---|---|---|---|---|
| Monotherapy for Melanoma | Phase III | ~50% | Not specified | Approved for unresectable melanoma |
| Combination with Trametinib | Phase III | 67% | 9.3 months | Significant improvement over monotherapy |
| Adjuvant Therapy | Phase III | Not specified | Longer than placebo | Reduced risk of death by 25% |
| Tissue-Agnostic Indications | Basket Trials | ~38% | Not specified | Approved for various solid tumors |
Mechanism of Action
Dabrafenib is a competitive and selective inhibitor of the BRAF kinase, specifically targeting the ATP pocket of the enzyme . It has a higher affinity for mutant forms of BRAF, including BRAF V600E, BRAF V600K, and BRAF V600D . By inhibiting BRAF, this compound disrupts the MAPK signaling pathway, leading to cell cycle arrest and apoptosis in cancer cells . This mechanism of action makes this compound effective in treating cancers driven by BRAF mutations .
Comparison with Similar Compounds
Dabrafenib is often compared with other BRAF inhibitors, such as vemurafenib and encorafenib . While all three compounds target the BRAF kinase, this compound has a unique chemical structure that allows for a different binding affinity and selectivity . This uniqueness contributes to its distinct pharmacokinetic and pharmacodynamic profiles . Other similar compounds include trametinib, which is often used in combination with this compound to enhance its therapeutic effects .
Biological Activity
Dabrafenib is a selective inhibitor of the BRAF kinase, primarily used in the treatment of melanoma with specific BRAF mutations. Its biological activity has been extensively studied, revealing a complex mechanism of action that extends beyond simple BRAF inhibition. This article synthesizes findings from various studies, including preclinical and clinical data, to provide a comprehensive overview of this compound's biological activity.
This compound specifically targets the BRAF V600E mutation, which is prevalent in several cancers, particularly melanoma. The compound inhibits the BRAF kinase activity, leading to decreased phosphorylation of downstream effectors in the MAPK pathway, specifically MEK and ERK. This inhibition results in:
- Cell Cycle Arrest : this compound induces an initial G1 cell cycle arrest in tumor cells, followed by apoptosis (programmed cell death) due to sustained ERK inhibition .
- Tumor Growth Inhibition : In xenograft models of melanoma, this compound administration resulted in significant tumor growth inhibition, downregulating proliferation markers such as Ki67 and upregulating cell cycle inhibitors like p27 .
Target Profile
Recent studies have highlighted this compound's unique target profile compared to other BRAF inhibitors like vemurafenib. While both compounds effectively inhibit BRAF and CRAF, this compound also significantly inhibits additional kinases such as NEK9 and CDK16. The inhibition of these kinases contributes to its efficacy against tumors that do not harbor typical BRAF mutations:
- NEK9 Inhibition : this compound has been shown to inhibit NEK9 with an IC50 value of 1-9 nM, which is crucial for its anti-proliferative effects in NRAS- and KRAS-mutant cancer cell lines .
- CDK16 Inhibition : The compound also affects CDK16, leading to increased expression of p27 and subsequent cell cycle arrest .
Clinical Efficacy
This compound has demonstrated significant clinical efficacy in patients with BRAF V600E/K-mutated melanoma. A notable study involving 20 children with BRAFV600E-mutated Langerhans Cell Histiocytosis showed promising results, indicating its potential beyond adult melanoma treatment .
Case Study: Pediatric Use
A phase II trial assessed the effectiveness of this compound in pediatric patients with LCH. Results indicated:
- Overall Response Rate : 75% of patients achieved a complete or partial response.
- Safety Profile : The treatment was well-tolerated with manageable side effects.
Combination Therapy
The combination of this compound with MEK inhibitors (such as trametinib) has shown enhanced efficacy and safety profiles by mitigating paradoxical MAPK pathway activation observed with monotherapy. This combination therapy approach reduces the occurrence of skin lesions and improves overall tumor response rates:
| Treatment Approach | Tumor Response Rate | Side Effects |
|---|---|---|
| This compound Monotherapy | 40% | High incidence of skin lesions |
| This compound + Trametinib | 70% | Reduced skin lesions, manageable |
Q & A
Basic Research Questions
Mechanistic Validation Q: How does dabrafenib selectively inhibit BRAF V600E-mutant kinase activity, and what methodological approaches validate its target specificity in preclinical models? A: Preclinical validation involves comparative kinase panel screening to confirm selectivity for mutant BRAF over wild-type isoforms. Cell viability assays in isogenic BRAF-mutant vs. wild-type cell lines, coupled with xenograft studies in immunodeficient mice, are critical. Western blotting for pERK suppression and tumor growth inhibition metrics (e.g., %TGI) quantify target engagement .
Analytical Method Development Q: What validated HPLC-MS methods are recommended for quantifying this compound in pharmacokinetic studies, and how do parameters like LOD/LOQ ensure reliability? A: Reverse-phase HPLC with tandem mass spectrometry (LC-MS/MS) using deuterated internal standards (e.g., this compound-d9) achieves sensitivity down to 1 ng/mL. Validation per ICH Q2(R1) guidelines requires assessing linearity (5–500 ng/mL), precision (CV <15%), and accuracy (85–115%). LOD/LOQ calculations via signal-to-noise ratios (3:1 and 10:1, respectively) ensure reliable detection in biological matrices .
Advanced Research Questions
Model System Comparisons Q: How do 2D and 3D melanoma cell culture models differ in their response to this compound, particularly regarding apoptosis and senescence? A: 3D spheroids exhibit delayed apoptosis (caspase-3/7 activation) and increased senescence (β-galactosidase staining) compared to 2D monolayers due to reduced drug penetration and microenvironmental stress. Flow cytometry for Annexin V/propidium iodide and senescence-associated secretory phenotype (SASP) cytokine profiling (e.g., IL-6, IL-8) differentiate these responses .
Combination Therapy Synergy Q: What synergistic mechanisms underlie the combination of this compound with MEK inhibitors like trametinib, and how do longitudinal RPPA analyses inform pathway dynamics? A: Co-inhibition prevents MAPK pathway reactivation via feedback ERK signaling. Reverse-phase protein array (RPPA) analysis at serial timepoints (e.g., 0, 24, 72 hours) tracks phospho-protein dynamics (e.g., pMEK, pERK rebound). Synergy scores (e.g., Chou-Talalay CI <1) quantify interaction effects in dose-matrix assays .
Survival Predictors Q: Which baseline factors (e.g., LDH levels, metastasis sites) are multivariate predictors of long-term survival in this compound-treated melanoma patients? A: Cox regression analyses of pooled COMBI-d/v trials identified lactate dehydrogenase (LDH) >ULN (HR=1.8), ≥3 metastatic organ sites (HR=1.5), and ECOG ≥1 (HR=1.3) as negative predictors. Complete response (CR) status correlates with 5-year OS of 71% vs. 19% for non-CR patients .
Resistance Mechanisms Q: What in vitro models identify MAPK reactivation as a resistance mechanism to this compound, and what pharmacodynamic markers track early adaptive responses? A: Long-term culture of BRAF-mutant cells with incremental this compound doses induces MAPK rebound (pERK recovery at 8–12 weeks). RNA-seq identifies upregulated RTKs (e.g., EGFR, PDGFRβ). Early pharmacodynamic markers include increased DUSP6 mRNA and phosphorylated RSK .
Non-Melanoma Applications Q: How do response rates to this compound/trametinib vary across BRAF-mutant NSCLC, thyroid cancer, and gliomas, and what genomic co-alterations influence therapeutic thresholds? A: In NSCLC (ORR=64% vs. 33% in gliomas), co-occurring PI3K/AKT mutations reduce efficacy. Thyroid cancer responses correlate with BRAF V600E allele burden (>30% by NGS). CDKN2A deletions in gliomas associate with shorter PFS (HR=2.1) .
Senescence vs. Apoptosis Q: What experimental techniques differentiate this compound-induced senescence from apoptosis in BRAF-mutant cells, and how does β-galactosidase staining correlate with caspase activation? A: Concurrent SA-β-gal staining (senescence) and caspase-3/7 glo assays (apoptosis) at 72-hour treatment. Senescent cells show G1 arrest (flow cytometry) without caspase activity, while apoptotic cells exhibit sub-G1 peaks and PARP cleavage .
Cross-Trial Analysis Q: How do pooled analyses of COMBI-d/v trials address heterogeneity in survival endpoints when combining this compound studies with different comparator arms? A: Multivariate network meta-analysis adjusts for trial-specific factors (e.g., COMBI-d used this compound monotherapy control, COMBI-v used vemurafenib). Bayesian hierarchical models account for between-study variance in OS/PFS hazard ratios .
Pharmacokinetic Variability Q: What physiologically-based pharmacokinetic (PBPK) models explain this compound’s variable exposure in hepatic impairment, and what sampling strategies optimize trough level monitoring? A: PBPK simulations incorporating CYP3A4/CYP2C8 hepatic extraction ratios predict 2.3-fold AUC increase in moderate impairment. Limited sampling strategies (LSS) using trough (Cmin) and 2-hour post-dose (C2hr) concentrations achieve r²>0.85 for AUC estimation .
Properties
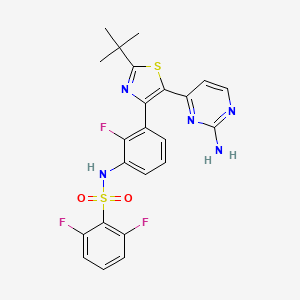
IUPAC Name |
N-[3-[5-(2-aminopyrimidin-4-yl)-2-tert-butyl-1,3-thiazol-4-yl]-2-fluorophenyl]-2,6-difluorobenzenesulfonamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C23H20F3N5O2S2/c1-23(2,3)21-30-18(19(34-21)16-10-11-28-22(27)29-16)12-6-4-9-15(17(12)26)31-35(32,33)20-13(24)7-5-8-14(20)25/h4-11,31H,1-3H3,(H2,27,28,29) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
BFSMGDJOXZAERB-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)(C)C1=NC(=C(S1)C2=NC(=NC=C2)N)C3=C(C(=CC=C3)NS(=O)(=O)C4=C(C=CC=C4F)F)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C23H20F3N5O2S2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID20152499 | |
| Record name | Dabrafenib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID20152499 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
519.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
very slightly soluble at pH 1 | |
| Record name | Dabrafenib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08912 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Dabrafenib is a competitive and selective BRAF inhibitor by binding to its ATP pocket.. Although dabrafenib can inhibit wild-type BRAF, it has a higher affinity for mutant forms of BRAF, including BRAF V600E, BRAF V600K, and BRAF V600D. BRAF is a serine/threonine protein kinase and is involved in activating the RAS/RAF/MEK/ERK or MAPK pathway, a pathway that is implicated in cell cycle progression, cell proliferation, and arresting apoptosis.Therefore, constitutive active mutation of BRAF such as BRAF V600E is frequently observed in many types of cancer, including melanoma, lung cancer, and colon cancer. | |
| Record name | Dabrafenib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08912 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
1195765-45-7 | |
| Record name | Dabrafenib | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1195765-45-7 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Dabrafenib [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1195765457 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Dabrafenib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08912 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Dabrafenib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID20152499 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | N-[3-[5-(2-aminopyrimidin-4-yl)-2-tert-butyl-1,3-thiazol-4-yl]-2-fluorophenyl]-2,6-difluorobenzenesulfonamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.215.965 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | DABRAFENIB | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/QGP4HA4G1B | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details





Synthesis routes and methods III
Procedure details






Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













