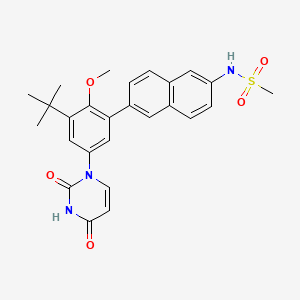
Dasabuvir
Overview
Description
Dasabuvir, sold under the brand name Exviera, is an antiviral medication used for the treatment of chronic Hepatitis C virus (HCV) infection. It is a non-nucleoside inhibitor of the HCV RNA-dependent RNA polymerase, specifically targeting the NS5B polymerase . This compound is often used in combination with other antiviral agents such as ombitasvir, paritaprevir, and ritonavir to achieve a sustained virologic response (SVR) in patients .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of Dasabuvir involves multiple steps, starting from commercially available starting materials. The key steps include the formation of the methanesulfonamide moiety and the coupling of the naphthyl and pyrimidinyl groups. The reaction conditions typically involve the use of organic solvents, catalysts, and controlled temperatures to ensure high yield and purity .
Industrial Production Methods: Industrial production of this compound follows a similar synthetic route but is optimized for large-scale manufacturing. This includes the use of continuous flow reactors, automated systems, and stringent quality control measures to ensure consistency and compliance with regulatory standards .
Chemical Reactions Analysis
Metabolic Pathways
Dasabuvir undergoes extensive hepatic metabolism, primarily mediated by cytochrome P450 (CYP) enzymes and subsequent conjugation reactions:
- Metabolite M1 : A hydroxylated derivative with reduced antiviral activity compared to the parent compound .
- Elimination : 94.4% excreted in feces (26.2% as unchanged drug), 2% in urine (0.03% unchanged) .
Enzyme Inhibition and Drug-Drug Interactions
This compound exhibits no significant inhibition or induction of CYP enzymes, but its metabolism is sensitive to CYP2C8 inhibitors/inducers:
Oxidative Stability and Degradation
Structural features influence this compound’s stability under physiological and experimental conditions:
Synthetic and Manufacturing Considerations
While synthetic details are proprietary, key steps involve:
- Naphthalene sulfonamide coupling with tert-butyl-methoxy-phenyl intermediates .
- Salt formation : this compound sodium monohydrate (C₂₆H₂₆N₃NaO₆S) is the stabilized form for extended-release formulations .
| Parameter | Value |
|---|---|
| Molecular Weight | 493.58 g/mol (free base) |
| Solubility | Slightly soluble in water |
| pKa | 8.2 (pyrimidinedione), 9.2 (sulfonamide) |
Pharmacokinetic Parameters
Key pharmacokinetic data from clinical studies:
| Parameter | Value (Mean ± SD) | Source |
|---|---|---|
| Cₘₐₓ | 1,980 ± 520 ng/mL | |
| AUC₀–₂₄ | 12,300 ± 3,450 ng·h/mL | |
| Tₘₐₓ | 4 hours | |
| Absolute Bioavailability | 70% |
Experimental Findings
Scientific Research Applications
Clinical Applications
Dasabuvir is utilized in combination with other direct-acting antivirals (DAAs) to enhance treatment efficacy. The following table summarizes key clinical trials and their findings regarding this compound's efficacy:
| Trial Name | Treatment Regimen | Duration (weeks) | Sustained Virologic Response (%) |
|---|---|---|---|
| AVIATOR | Paritaprevir/r + Ombitasvir + this compound + Ribavirin | 8 | 87.5 |
| SAPHIRE I | Paritaprevir/r + Ombitasvir + this compound + Ribavirin | 12 | 95.3 (genotype 1a) |
| SAPHIRE II | Paritaprevir/r + Ombitasvir + this compound + Ribavirin | 12 | 98.0 (genotype 1b) |
| Open-label Phase IIa | This compound monotherapy followed by combination therapy | 28 | 87.5 |
These trials demonstrate that this compound-containing regimens achieve high rates of SVR, particularly when combined with other DAAs such as paritaprevir and ombitasvir .
Combination Therapies
This compound is often used in conjunction with other antiviral agents to improve treatment outcomes. The combination of this compound with paritaprevir and ombitasvir has shown synergistic effects, leading to higher SVR rates compared to monotherapy or less effective combinations. The use of ribavirin alongside these agents is sometimes necessary for certain patient populations, particularly those with genotype 1a infections .
Safety and Tolerability
This compound has been generally well-tolerated in clinical studies. Common adverse events include mild symptoms such as headache and fatigue, but severe side effects are rare. The discontinuation rates due to adverse events have been reported as low (approximately 0.6%) across various studies, indicating a favorable safety profile .
Case Studies
Several case studies have documented the successful application of this compound in real-world settings:
- Case Study 1 : A cohort of patients with chronic HCV genotype 1a infection underwent treatment with a regimen including this compound and achieved an SVR rate exceeding 90%. Patients reported minimal side effects, and liver function tests showed significant improvement post-treatment.
- Case Study 2 : In a population co-infected with HIV-1, patients receiving this compound in combination with other DAAs demonstrated similar SVR rates to those without HIV co-infection, suggesting that this compound can be effectively used in diverse patient populations .
Mechanism of Action
Dasabuvir exerts its antiviral effects by inhibiting the HCV RNA-dependent RNA polymerase encoded by the NS5B gene. It binds to the palm domain of the NS5B polymerase, inducing a conformational change that renders the polymerase unable to elongate viral RNA. This inhibition prevents the replication of the viral genome, leading to a reduction in viral load and ultimately achieving a sustained virologic response .
Comparison with Similar Compounds
Efavirenz: Another non-nucleoside inhibitor used in the treatment of HIV infection.
Tipranavir: A protease inhibitor used in combination with other antiretroviral agents for the treatment of HIV.
Comparison: Dasabuvir is unique in its specific targeting of the HCV NS5B polymerase, whereas Efavirenz and Tipranavir target different viral enzymes. This compound’s high specificity for the NS5B polymerase makes it particularly effective against HCV genotype 1, while Efavirenz and Tipranavir are used for HIV treatment .
Biological Activity
Dasabuvir targets the NS5B polymerase enzyme, which is crucial for viral RNA replication. By binding to the allosteric site of the NS5B polymerase, this compound inhibits its activity, thereby preventing the replication of HCV. This mechanism is distinct from that of nucleoside analogs, allowing this compound to be effective against various HCV genotypes.
Structure-Activity Relationship (SAR)
The chemical structure of this compound consists of a 1-[(2S)-2-(4-((2-(4-(trifluoromethyl)phenyl)thiazol-2-yl)methylthio)-phenyl)thiazol-4-yl)methyl]-pyrrolidine-2-carboxylic acid derivative. Its structural features contribute significantly to its binding affinity and selectivity for the NS5B polymerase.
Pharmacokinetics
This compound exhibits favorable pharmacokinetic properties:
- Absorption : Rapidly absorbed with peak plasma concentrations achieved within 1–3 hours post-administration.
- Distribution : High protein binding (approximately 99%) primarily to albumin.
- Metabolism : Primarily metabolized by cytochrome P450 enzymes (CYP2C8 and CYP3A4).
- Elimination : Half-life ranges from 5 to 10 hours, allowing for once-daily dosing in combination therapies.
Table 1: Pharmacokinetic Profile of this compound
| Parameter | Value |
|---|---|
| Bioavailability | ~50% |
| Peak Plasma Concentration | 1–3 hours |
| Protein Binding | ~99% |
| Metabolism | CYP2C8, CYP3A4 |
| Elimination Half-Life | 5–10 hours |
Clinical Efficacy
This compound is typically used in combination with other antiviral agents such as Ombitasvir, Paritaprevir, and Ritonavir. Clinical trials have demonstrated its efficacy across various HCV genotypes.
Case Studies
- Study on Genotype 1 HCV : A phase III clinical trial evaluated the efficacy of this compound in combination with Ombitasvir and Paritaprevir in treatment-naive patients with genotype 1 HCV. Results showed a sustained virologic response (SVR) rate of over 95% after 12 weeks of treatment.
- Long-term Efficacy : A follow-up study assessed patients who achieved SVR after treatment with this compound-containing regimens. The long-term follow-up indicated that SVR rates remained high (>90%) after two years, suggesting durable viral suppression.
Table 2: Clinical Trial Results for this compound
| Study Type | Population | Treatment Regimen | SVR Rate (%) |
|---|---|---|---|
| Phase III Trial | Treatment-naive Genotype 1 HCV | This compound + Ombitasvir + Paritaprevir | >95 |
| Long-term Follow-up | SVR Achievers | Same regimen | >90 |
Safety Profile
The safety profile of this compound has been evaluated in multiple clinical trials. Common adverse effects include:
- Fatigue
- Nausea
- Headache
- Insomnia
Serious adverse events are rare but can include liver enzyme elevations and hypersensitivity reactions.
Table 3: Adverse Events Associated with this compound
| Adverse Event | Incidence (%) |
|---|---|
| Fatigue | 10 |
| Nausea | 8 |
| Headache | 6 |
| Insomnia | 5 |
| Liver Enzyme Elevation (ALT/AST) | <5 |
Resistance Profile
Resistance to this compound can occur, particularly in patients with prior treatment experience. Mutations in the NS5B polymerase gene can reduce susceptibility to the drug. Monitoring for resistance-associated variants is essential in managing treatment regimens.
Q & A
Basic Research Questions
Q. What is the primary mechanism of action of dasabuvir against hepatitis C virus (HCV)?
this compound acts as a non-nucleoside inhibitor (NNI) of the HCV NS5B RNA-dependent RNA polymerase (RdRp). It binds to the palm domain of NS5B, inducing conformational changes that inhibit viral RNA synthesis. Key evidence includes its EC₅₀ values of 7.7 nM (genotype 1a) and 1.8 nM (genotype 1b) in replicon assays . Methodologically, RdRp inhibition is validated via in vitro polymerase activity assays and subgenomic replicon systems .
Q. How is this compound’s in vitro antiviral activity assessed against HCV genotypes?
Antiviral efficacy is quantified using subgenomic replicon systems transfected into hepatoma cell lines (e.g., Huh-7). Key metrics include:
- EC₅₀ : Determined via dose-response curves (Table 1).
- Selectivity index (SI) : Ratio of cytotoxic concentration (CC₅₀) to EC₅₀.
| Genotype | EC₅₀ (nM) | Reference |
|---|---|---|
| 1a (H77) | 7.7 | |
| 1b (Con1) | 1.8 |
Replicon resistance profiling involves maintaining cells under drug pressure to select for mutations (e.g., C316Y, M414T) .
Q. Which metabolic enzymes are involved in this compound’s clearance?
this compound is metabolized primarily by CYP2C8 (60%) and CYP3A4 (30%). The major metabolite, M1 (hydroxylated tert-butyl), undergoes glucuronidation and sulfation. Methodological validation includes:
- Radiolabeled studies : [¹⁴C]-dasabuvir administered to humans, with metabolites identified via LC-MS/MS .
- Recombinant CYP assays : Incubation with human liver microsomes to confirm enzyme contributions .
Q. What experimental methods quantify this compound and its metabolites in biological matrices?
Stability-indicating HPLC-DAD and UPLC-MS/MS are used:
- HPLC-DAD : Validated for this compound and degradation products (e.g., alkaline DP1/DP2) with LOD 0.12 µg/mL and LOQ 0.37 µg/mL .
- UPLC-MS/MS : Quantifies this compound, M1, and co-administered DAAs (e.g., ombitasvir) in plasma, with deuterated internal standards (e.g., C13D3-dasabuvir) .
Advanced Research Questions
Q. How does this compound’s activity extend to non-HCV viruses (e.g., EV-A71, dengue)?
Repurposing studies use structure-based virtual screening and functional assays:
- EV-A71 : this compound inhibits viral replication (IC₅₀ = 1.8 µM) via ROCK1 interaction, validated by qPCR, TCID₅₀, and cytokine profiling (MCP-1, TNF-α reduction) .
- Dengue : Plaque reduction assays show 1 µM this compound reduces viral titers in co-infection models, though efficacy requires further validation .
Q. What methodologies identify resistance mutations in HCV NS5B after this compound exposure?
Resistance profiling involves:
- In vitro selection : Replicons cultured under escalating this compound concentrations (10–100× EC₅₀) yield mutations (e.g., C316N, S556G) .
- Computational mutagenesis : Molecular dynamics simulations analyze mutation effects on binding affinity (e.g., C445F disrupts hydrogen bonding with Asn291) .
Q. How do structural biology techniques elucidate this compound-NS5B interactions?
- Docking studies : this compound’s binding to the palm I site is modeled using NS5B crystal structures (PDB: 4WTD). Key interactions include hydrophobic contacts with Phe193 and hydrogen bonds with Asn291 .
- Site-directed mutagenesis : Mutant NS5B (e.g., M414V) is expressed in replicons to assess reduced this compound susceptibility (EC₅₀ shifts >10-fold) .
Q. What pharmacokinetic (PK) considerations apply to this compound in hepatic impairment models?
PK studies in cirrhotic patients reveal:
- AUC changes : +325% in CP-C patients (Child-Pugh C) due to reduced CYP2C8 activity .
- Protein binding : Unbound fraction increases from 0.61% (healthy) to 0.42% (CP-C), impacting free drug exposure .
Methodologically, population PK models integrate covariates (e.g., albumin, bilirubin) to predict dose adjustments .
Q. Why is this compound combined with ombitasvir/paritaprevir/ritonavir in HCV therapy?
The regimen targets multiple viral proteins:
- NS3/4A protease (paritaprevir), NS5A (ombitasvir), NS5B (this compound).
- Synergy : EC₅₀ values decrease 10-fold in combination vs. monotherapy .
- Resistance barrier : Non-overlapping resistance profiles (e.g., NS5A Y93H vs. NS5B C316N) reduce virologic failure risk .
Q. How does this compound’s anti-inflammatory activity modulate viral pathogenesis?
In EV-A71-infected THP-1 cells, this compound reduces pro-inflammatory cytokines (MCP-1, TNF-α) via:
- qPCR/ELISA : Quantifies cytokine mRNA/protein levels post-treatment .
- Mechanistic link : Inhibition of ROCK1 signaling, validated via siRNA knockdown .
Contradictions and Limitations
- Genotype specificity : this compound inhibits EV-A71 and CVA10 (Enterovirus A) but not CVB1 (Enterovirus B), suggesting structural constraints in RdRp binding .
- Resistance mutations : NS5B variants (e.g., Y448H) reduce this compound’s efficacy in HCV genotype 1a but not 1b .
Methodological Gaps
- In vivo models : Limited data on this compound’s efficacy in animal models of dengue or EV-A71.
- Cross-resistance : Impact of NS5B mutations on other palm-domain inhibitors (e.g., tegobuvir) remains underexplored .
Properties
IUPAC Name |
N-[6-[3-tert-butyl-5-(2,4-dioxopyrimidin-1-yl)-2-methoxyphenyl]naphthalen-2-yl]methanesulfonamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C26H27N3O5S/c1-26(2,3)22-15-20(29-11-10-23(30)27-25(29)31)14-21(24(22)34-4)18-7-6-17-13-19(28-35(5,32)33)9-8-16(17)12-18/h6-15,28H,1-5H3,(H,27,30,31) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
NBRBXGKOEOGLOI-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)(C)C1=CC(=CC(=C1OC)C2=CC3=C(C=C2)C=C(C=C3)NS(=O)(=O)C)N4C=CC(=O)NC4=O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C26H27N3O5S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID301025953 | |
| Record name | Dasabuvir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID301025953 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
493.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
Dasabuvir is a non-nucleoside inhibitor of the HCV RNA-dependent RNA polymerase encoded by the NS5B gene, which is essential for replication of the viral genome. Based on drug resistance mapping studies of HCV genotypes 1a and 1b, dasabuvir targets the palm domain of the NS5B polymerase, and is therefore referred to as a non-nucleoside NS5B-palm polymerase inhibitor. The EC50 values of dasabuvir against genotype 1a-H77 and 1b-Con1 strains in HCV replicon cell culture assays were 7.7 nM and 1.8 nM, respectively. By binding to NS5b outside of the active site of the enzyme, dasabuvir induces a conformational change thereby preventing further elongation of the nascent viral genome. A limitation of binding outside of the active site is that these binding sites are poorly preserved across the viral genotypes. This results in a limited potential for cross-genotypic activity and increased potential for development of resistance. Dasabuvir is therefore limited to treating genotypes 1a and 1b, and must be used in combination with other antiviral products. | |
| Record name | Dasabuvir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB09183 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
1132935-63-7 | |
| Record name | Dasabuvir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1132935-63-7 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Dasabuvir [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1132935637 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Dasabuvir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB09183 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Dasabuvir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID301025953 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | DASABUVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/DE54EQW8T1 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Synthesis routes and methods I
Procedure details













Synthesis routes and methods II
Procedure details













Synthesis routes and methods III
Procedure details










Synthesis routes and methods IV
Procedure details










Synthesis routes and methods V
Procedure details










Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













