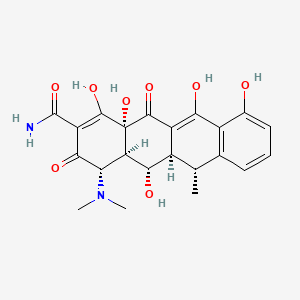
Doxycycline
Overview
Description
Doxycycline is a broad-spectrum antibiotic belonging to the tetracycline class. It is used to treat a variety of bacterial infections, including bacterial pneumonia, acne, chlamydia infections, Lyme disease, cholera, typhus, and syphilis. It is also used to prevent malaria . This compound was first patented in 1957 and came into commercial use in 1967 . It is available as a generic medicine and is on the World Health Organization’s List of Essential Medicines .
Preparation Methods
Doxycycline is synthetically derived from oxytetracycline. The preparation involves several steps:
Chlorination and Dehydration: Oxytetracycline undergoes chlorination and dehydration to form 11 alpha-chloro-6-methenyl oxytetracycline p-toluenesulfonate.
Hydrogenation: This intermediate is then subjected to a one-step or two-step hydrogenation process to produce this compound p-toluenesulfonate.
Refinement: Finally, the product is refined to form this compound hydrochloride.
Chemical Reactions Analysis
Oxidative Degradation via Fenton Process
Doxycycline reacts with hydroxyl radicals (- OH) generated in the Fenton process (Fe²⁺/H₂O₂). Optimal degradation conditions and outcomes include:
| Parameter | Optimal Value | Residual this compound (mg/L) | TOC Reduction (%) |
|---|---|---|---|
| H₂O₂ concentration | 611 mg/L | 0 | ≤30 |
| Fe²⁺ concentration | 25 mg/L | ||
| Temperature | 35°C |
-
H₂O₂ concentration is the most statistically significant variable (ANOVA p < 0.05) .
-
Degradation follows pseudo-first-order kinetics, producing intermediates such as 4a,11a-anhydrotetracycline and 6-epithis compound .
Photodegradation
This compound is highly sensitive to light, especially under alkaline conditions:
| Condition | Degradation Efficiency (%) | Half-Life (t₁/₂) |
|---|---|---|
| pH 4.0 (dark) | <5 | >48 h |
| pH 7.2 (simulated sun) | 95 | 1.2 h |
| UV/H₂O₂ (pH 7.0) | 99.6 | 6 min |
-
Photodegradation generates aromatic byproducts and quinone derivatives , which exhibit reduced antimicrobial activity .
-
Dissipation kinetic constant () increases linearly with pH (R² = 0.98) .
Hydrolysis and Thermal Degradation
Hydrolytic stability varies with temperature and pH:
| Medium | Degradation Products | Kinetic Order |
|---|---|---|
| Acidic (pH < 3) | Anhydrothis compound | Zero-order |
| Alkaline (pH > 8) | β-Diketone and lactone derivatives | Second-order |
| High humidity (75% RH) | Isothis compound | Mixed-order |
-
Thermal degradation at 40°C reduces this compound content by 12% in 6 months .
-
Hydrolysis at C-4 and C-6 positions dominates in aqueous solutions .
Ozonation
Ozone rapidly degrades this compound via electrophilic attack on its phenolic and amine groups:
| Ozone Dose (mg/L) | Degradation Efficiency (%) | Toxicity Reduction (%) |
|---|---|---|
| 27.7 | 99.9 | 100 (Vibrio fischeri) |
| 15.0 | 85 | 75 |
-
Ozonation produces non-toxic carboxylic acids (e.g., oxalic acid) and completely eliminates antimicrobial activity .
Interaction with Metal Ions
This compound forms stable chelates with divalent and trivalent metals, altering its reactivity:
| Metal Ion | Stability Constant (log K) | Effect on Degradation |
|---|---|---|
| Fe³⁺ | 24.5 | Inhibits Fenton process |
| Ca²⁺ | 7.8 | Reduces solubility |
| Mg²⁺ | 7.2 | Enhances photolysis |
Solubility and Stability Profile
This compound’s solubility influences its reactivity in different solvents:
| Solvent | Solubility (mg/mL) | Stability |
|---|---|---|
| Water | 0.1 | pH-dependent |
| 0.1M HCl | 50 | Stable |
| Methanol | 0.5 | Degrades |
Advanced Oxidation Processes (AOPs)
Comparative efficiency of AOPs in this compound degradation:
| Method | Time (min) | Efficiency (%) | Byproducts Identified |
|---|---|---|---|
| UV/H₂O₂ | 6 | 99.6 | Low-toxicity acids |
| Ozonation | 10 | 99.9 | Non-toxic fragments |
| Photoperoxidation | 30 | 95 | Toxic intermediates |
Scientific Research Applications
Clinical Applications
Infectious Diseases Treatment
Doxycycline is primarily indicated for treating a variety of bacterial infections, including:
- Rickettsial Infections : Effective against Rocky Mountain spotted fever and typhus.
- Sexually Transmitted Infections (STIs) : Proven efficacy in reducing the incidence of chlamydia, gonorrhea, and syphilis, especially among high-risk populations such as men who have sex with men (MSM) and transgender women. A notable study indicated a reduction in STIs by about 70% when administered within 72 hours post-exposure .
- Respiratory Tract Infections : Used for infections caused by pathogens like Mycoplasma pneumoniae and Haemophilus influenzae.
- Malaria Prophylaxis : Recommended for travelers to endemic regions as an alternative to primaquine, particularly due to its safety profile in patients with glucose-6-phosphate dehydrogenase deficiency .
Non-Infectious Conditions
this compound also has applications in treating non-infectious conditions:
- Acne Treatment : Effective against Propionibacterium acnes, commonly associated with acne.
- Chronic Inflammatory Conditions : Used for conditions like rosacea and periodontitis.
- Cancer Research : Investigated for its potential anti-cancer properties, including inhibition of cell proliferation and induction of apoptosis in certain cancer types .
Emerging Research Applications
Post-Exposure Prophylaxis (PEP)
Recent studies have highlighted this compound's role as a preventive measure against STIs. In clinical trials, participants taking this compound PEP reported significant reductions in STI acquisition rates. For instance, a study involving 501 participants demonstrated a relative risk reduction of 70% for chlamydia and syphilis when this compound was taken within 72 hours after condomless sex .
| Study | Population | Outcome | Reduction in STIs |
|---|---|---|---|
| IPERGAY Study | MSM and Transgender Women | First bacterial STI infection | Chlamydia: 70%, Syphilis: 73% |
| DoxyPEP Trial | HIV-positive/PrEP Users | Incidence of STIs | Gonorrhea: 55-60% |
| NIH Study | MSM and Transgender Women | Acquisition of STIs | Two-thirds reduction |
Case Studies and Research Findings
Numerous studies have documented the efficacy and safety of this compound in various contexts:
- A randomized controlled trial demonstrated that participants who took this compound within 72 hours after potential exposure to STIs had significantly lower rates of infection compared to those who did not receive the medication .
- Research on the long-term effects of this compound use has shown no significant increase in antimicrobial resistance among common pathogens when used as directed for conditions like acne or malaria prophylaxis .
Mechanism of Action
Doxycycline exerts its effects by inhibiting bacterial protein synthesis. It binds reversibly to the 30S ribosomal subunit, preventing the association of aminoacyl-tRNA with the bacterial ribosome . This inhibition of protein synthesis is bacteriostatic, meaning it prevents the growth and reproduction of bacteria. This compound also targets the apicoplast in malaria-causing parasites, disrupting their function .
Comparison with Similar Compounds
Doxycycline is often compared with other tetracyclines such as tetracycline, oxytetracycline, chlortetracycline, demeclocycline, lymecycline, methacycline, and minocycline . Compared to these compounds, this compound has several unique features:
Absorption: This compound is more reliably absorbed orally than older tetracyclines.
Lipophilicity: It is more lipophilic, allowing it to pass more easily through bacterial lipid bilayers.
Half-life: This compound has a longer serum half-life, allowing for less frequent dosing.
Similar compounds include:
Biological Activity
Doxycycline, a member of the tetracycline class of antibiotics, exhibits a broad spectrum of biological activities, particularly its potent antibacterial effects. This article delves into the various aspects of this compound's biological activity, including its mechanisms of action, pharmacokinetics, clinical applications, and recent advancements in drug delivery systems.
This compound exerts its antibacterial effects primarily by inhibiting bacterial protein synthesis. It binds to the 30S ribosomal subunit, preventing the association of aminoacyl-tRNA with the ribosome, which is crucial for protein translation. This mechanism is effective against a wide range of both gram-positive and gram-negative bacteria, including anaerobic pathogens .
Pharmacokinetics
This compound is characterized by:
- Rapid Absorption : It is rapidly and nearly completely absorbed when administered orally, with food not significantly affecting its bioavailability.
- Tissue Penetration : this compound achieves high concentrations in various tissues, including the lungs, kidneys, and reproductive organs. This extensive tissue distribution enhances its therapeutic efficacy in treating infections localized in these areas .
- Half-Life : The drug has a prolonged half-life, allowing for once-daily dosing in many cases. Importantly, it does not accumulate in patients with renal insufficiency and is minimally removed during hemodialysis .
Clinical Applications
This compound is widely used in clinical practice for various infections:
- Respiratory Tract Infections : Effective against atypical pneumonia and other respiratory infections.
- Skin and Soft Tissue Infections : Commonly prescribed for acne and other skin infections.
- Genitourinary Infections : Used to treat conditions such as gonorrhea and syphilis.
- Periodontal Disease : Recent studies suggest that this compound may improve glycemic control in diabetic patients with periodontal disease, showing a reduction in HbA1c levels .
Recent Advances in Drug Delivery
Recent research has focused on enhancing the efficacy of this compound through novel drug delivery systems:
- Nanoparticle Encapsulation : this compound-loaded nanoparticles have shown improved antibacterial activity compared to free this compound. For example, studies indicate that encapsulating this compound in polymeric nanoparticles enhances its cellular penetration and sustained release, making it more effective against intracellular pathogens like E. coli .
- Combination Therapies : Combining this compound with other agents (e.g., curcumin) in liposomal formulations has demonstrated synergistic effects against bacterial pathogens while maintaining low cytotoxicity to human cells .
Case Studies
Several case studies highlight the effectiveness of this compound in specific clinical scenarios:
- Diabetes Management : A randomized controlled trial involving 2565 participants indicated that scaling and root planing (SRP) combined with this compound treatment led to significant reductions in HbA1c levels among diabetic patients at 3 to 4 months post-treatment .
- Intravenous Use : In severe infections requiring hospitalization, intravenous administration of this compound has been shown to achieve comparable serum concentrations to oral dosing, providing flexibility in treatment options for critically ill patients .
Summary Table of this compound's Biological Activity
| Activity | Details |
|---|---|
| Mechanism of Action | Inhibits protein synthesis by binding to 30S ribosomal subunit |
| Spectrum of Activity | Effective against gram-positive/negative bacteria and anaerobes |
| Pharmacokinetics | Rapid absorption; extensive tissue distribution; prolonged half-life |
| Clinical Uses | Respiratory infections, skin infections, genitourinary infections, periodontal disease |
| Advancements | Nanoparticle formulations enhance efficacy; combination therapies improve outcomes |
Q & A
Basic Research Questions
Q. What analytical methodologies are recommended for quantifying doxycycline in pharmaceutical formulations?
A validated enzymatic technique with high accuracy and repeatability is recommended, employing recovery studies and statistical validation (e.g., mean recovery rates >95%). This method ensures reliable quality control for pharmaceutical batches and bulk samples .
Q. How should researchers design a randomized controlled trial (RCT) to evaluate this compound's efficacy in clinical settings?
Key considerations include:
- Primary endpoints : Predefined clinical outcomes (e.g., infection resolution rate).
- Blinding : Double-blind protocols to minimize bias.
- Inclusion/exclusion criteria : Strictly defined to ensure homogeneity (e.g., age, comorbidities).
- Data management : Use Case Report Forms (CRFs) and source documents (e.g., lab results, adverse event logs) for traceability .
Q. What protocols ensure data integrity in pharmacokinetic studies of this compound?
Maintain detailed source documents, including medical histories, lab records, and signed consent forms. Use CRFs for systematic data entry and audit trails to track modifications .
Q. How can researchers address potential biases in this compound clinical trials?
Implement randomization (e.g., stratified by risk factors), blinding (participant/investigator), and predefined statistical analysis plans to mitigate selection and measurement biases .
Advanced Research Questions
Q. How can Box-Behnken Design optimize this compound adsorption in environmental samples?
Experimental parameters should be modeled using response surface methodology. For MCM-41 adsorbents, optimal conditions include pH 7.3, 0.02 g/L adsorbent dose, and 20-minute contact time, achieving 99% removal efficiency .
Q. What methodologies determine tissue depletion kinetics of this compound in food-producing animals?
Conduct withdrawal studies with HPLC/MS analysis of edible tissues (e.g., muscle, liver) post-administration. In broilers, a 25% this compound formulation showed tissue-specific clearance rates, requiring withdrawal periods validated via residue depletion curves .
Q. How is this compound applied in tetracycline-controlled gene expression systems?
In "tet-on" systems, this compound (0–1 µg/mL) activates transgene expression via a tetracycline-controlled transactivator (tTA). Critical parameters include concentration-dependent regulation (5-log dynamic range) and reversibility upon withdrawal .
Q. What computational approaches validate this compound's specificity in transcriptional riboswitch design?
Use molecular docking and negative controls (e.g., this compound vs. tetracycline) to confirm aptamer specificity. In silico-designed riboswitches should show no activation with this compound, ensuring system reliability .
Q. How can conflicting data on this compound's pediatric safety profile be resolved methodologically?
Conduct longitudinal cohort studies with enamel hypoplasia assessments and meta-analyses of historical data. Recent CDC studies suggest minimal tooth staining risk in children under 8, contradicting prior hypotheses .
Q. What ethical guidelines govern human studies involving this compound?
Obtain informed consent with clear descriptions of risks/benefits. Follow protocols for adverse event reporting and ensure transparency in drug administration (e.g., dosage, off-label use). Reference institutional review board (IRB) standards and GCP guidelines .
Properties
IUPAC Name |
(4S,4aR,5S,5aR,6R,12aR)-4-(dimethylamino)-1,5,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4a,5,5a,6-tetrahydro-4H-tetracene-2-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H24N2O8/c1-7-8-5-4-6-9(25)11(8)16(26)12-10(7)17(27)14-15(24(2)3)18(28)13(21(23)31)20(30)22(14,32)19(12)29/h4-7,10,14-15,17,25-27,30,32H,1-3H3,(H2,23,31)/t7-,10+,14+,15-,17-,22-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
SGKRLCUYIXIAHR-AKNGSSGZSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1C2C(C3C(C(=O)C(=C(C3(C(=O)C2=C(C4=C1C=CC=C4O)O)O)O)C(=O)N)N(C)C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@@H]1[C@H]2[C@@H]([C@H]3[C@@H](C(=O)C(=C([C@]3(C(=O)C2=C(C4=C1C=CC=C4O)O)O)O)C(=O)N)N(C)C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H24N2O8 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
17086-28-1 (mono-hydrate), 41411-66-9 (6-epimer, mono-hydrochloride), 69935-17-7 (mono-hydrochloride, di-hydrate), 94088-85-4 (calcium salt (1:2)) | |
| Record name | Doxycycline [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000564250 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID0037653, DTXSID80992212 | |
| Record name | Doxycycline | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0037653 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 4-(Dimethylamino)-1,5,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-3,4,4a,5,5a,6,12,12a-octahydrotetracene-2-carboximidic acid | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80992212 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
444.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
SOL IN WATER /DOXYCYCLINE HYDROCHLORIDE/, VERY SLIGHTLY SOL IN WATER; SPARINGLY SOL IN ALC; FREELY SOL IN DIL ACID & ALKALI HYDROXIDE SOLN; PRACTICALLY INSOL IN CHLOROFORM & ETHER. | |
| Record name | Doxycycline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00254 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | DOXYCYCLINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3071 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
Protein synthesis is essential for survival and functioning of cells, including bacteria. Doxycycline inhibits bacterial protein synthesis by allosterically binding to the 30S prokaryotic ribosomal subunit. The drug blocks the association charged aminoacyl-tRNA (aa-tRNA) with the ribosomal A site, which is the acceptor site on the mRNA-ribosome complex. Doxycycline ultimately impedes the elongation phase of protein synthesis and halts the production of essential proteins for bacterial survival and functioning. Doxycycline mediates anti-inflammatory actions by preventing calcium-dependent microtubular assembly and lymphocytic proliferation, thereby inhibiting leukocyte movement during inflammation. It also inhibits nitric oxide synthase, which is an enzyme that produces nitric oxide, an inflammatory signaling molecule., TETRACYCLINES INHIBIT BACTERIAL PROTEIN SYNTHESIS. ... ONCE TETRACYCLINES GAIN ACCESS TO THE BACTERIAL CELL, THEY BIND PRINCIPALLY TO 30 S SUBUNITS OF BACTERIAL RIBOSOMES. THEY APPEAR TO PREVENT ACCESS OF AMINOACYL T-RNA TO M-RNA-RIBOSOME COMPLEX. ... ONLY SMALL PORTION OF DRUG IS IRREVERSIBLY BOUND, & INHIBITORY EFFECTS OF THE TETRACYCLINESARE REVERSIBLE WHEN THE DRUG IS REMOVED. /TETRACYCLINES/ | |
| Record name | Doxycycline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00254 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | DOXYCYCLINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3071 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
YELLOW, CRYSTALLINE POWDER | |
CAS No. |
564-25-0, 7164-70-7, 24390-14-5 | |
| Record name | Doxycycline [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000564250 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Doxycycline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00254 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Doxycycline | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0037653 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 4-(Dimethylamino)-1,5,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-3,4,4a,5,5a,6,12,12a-octahydrotetracene-2-carboximidic acid | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80992212 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Doxycycline | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.008.429 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | 2-Naphthacenecarboxamide, 4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-, hydrochloride, (4S,4aR,5S,5aR,6R,12aS)-, compd. with ethanol, hydrate (2:2:1:1) | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.106.555 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Doxycycline Hyclate | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | DOXYCYCLINE ANHYDROUS | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/334895S862 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | DOXYCYCLINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3071 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















