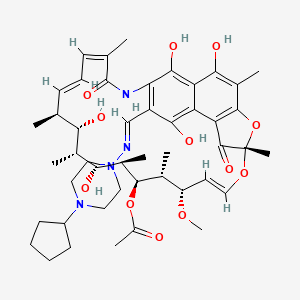
Rifapentine
- Click on QUICK INQUIRY to receive a quote from our team of experts.
- With the quality product at a COMPETITIVE price, you can focus more on your research.
Overview
Description
Rifapentine, sold under the brand name Priftin, is an antibiotic used primarily in the treatment of tuberculosis. It belongs to the rifamycin class of antibiotics and is known for its ability to inhibit bacterial RNA synthesis by targeting DNA-dependent RNA polymerase . This compound is particularly effective against Mycobacterium tuberculosis, the bacterium responsible for tuberculosis .
Preparation Methods
Synthetic Routes and Reaction Conditions: Rifapentine is synthesized starting from rifamycin S. The process involves several key steps:
Conversion to Intermediate: Rifamycin S reacts with dihydroxy methyl tert-butyl amine in an organic solvent to form an intermediate called rifaoxazine.
Hydrolysis and Ring Opening: The intermediate undergoes hydrolysis and ring opening in the presence of a catalyst and a reductant in a second organic solvent.
Condensation: The hydrolysis product is then condensed with 1-amino-4-cyclopentyl piperazine to form this compound.
Industrial Production Methods: The industrial production of this compound involves scaling up the laboratory synthesis process. Key challenges include maintaining the purity and uniformity of the product, optimizing reaction conditions, and ensuring efficient crystallization and drying processes .
Chemical Reactions Analysis
Structural Basis for Reactivity
Rifapentine shares a core ansamycin structure with rifampin but incorporates a cyclopentyl ring substitution at the C3 position. This modification enhances its lipophilicity and influences interactions with bacterial RNA polymerase, where it binds the β-subunit to inhibit transcription . The molecule contains multiple reactive sites, including hydroxyl, amine, and quinone groups, which govern its degradation and metabolic pathways.
Table 1: Synthetic Pathway Optimization
| Parameter | Original Method | Improved Method (Patent CN111018886A) |
|---|---|---|
| Cyclization Environment | Alkaline | Acidic (pH 4–5) |
| Impurity Control | ≤1% | ≤0.1% |
| Solvent Residues | n-butanol: ≤2% | n-butanol: ≤0.5% |
Degradation Pathways
This compound undergoes hydrolysis and oxidative degradation:
-
Hydrolysis : The ester bond at C25 hydrolyzes to form 25-desacetyl this compound, a less active metabolite .
-
Maillard Reaction : Reacts with amino-containing excipients (e.g., isoniazid) under acidic conditions, producing isonicotinyl hydrazine derivatives .
-
Oxidation : Quinone groups in the ansa chain oxidize under high humidity or light, reducing potency .
Table 2: Stability Under Environmental Stressors
| Condition | Degradation Rate (24h) | Major Degradation Products |
|---|---|---|
| High Humidity (85% RH) | 12% | 25-desacetyl this compound |
| Acidic pH (pH 3) | 18% | Isonicotinyl hydrazine derivatives |
| Light Exposure (UV) | 15% | Oxidized ansa-chain byproducts |
Interaction with Co-Administered Drugs
This compound’s reactivity complicates combination therapies:
-
Isoniazid (INH) : Forms hydrazine adducts via nucleophilic substitution, requiring formulation separation to prevent interaction .
-
Pyrazinamide : No direct reaction, but this compound’s hepatic enzyme induction (4.5-fold vs. rifampin’s 3-fold) alters pyrazinamide metabolism .
Analytical Characterization
Key methods for monitoring reactions include:
-
Reverse-Phase HPLC : Resolves this compound from impurities (e.g., 25-desacetyl metabolite) with a C18 column and acetonitrile-phosphate buffer .
-
FTIR/NMR : Confirms structural integrity during synthesis, particularly cyclopentyl substitution .
Stabilization Strategies
-
Excipient Selection : Avoid amino acids and acidic stabilizers to prevent Maillard reactions .
-
Packaging : Use light-resistant, desiccant-containing containers to mitigate hydrolysis and oxidation .
This compound’s chemical profile underscores the need for precise synthesis and storage protocols to maintain efficacy. Ongoing research focuses on stabilizing its reactive sites while enhancing bioavailability through formulation innovations .
Scientific Research Applications
Treatment of Tuberculosis
Active Tuberculosis
Rifapentine is effective in treating drug-susceptible TB. It is often used in combination with other antitubercular agents such as isoniazid. The efficacy of this compound in short-course regimens has been demonstrated in clinical trials, showing comparable results to standard treatments while reducing treatment duration from six months to four months .
Latent Tuberculosis Infection
this compound has gained prominence in treating latent TB infection, particularly among high-risk populations. A regimen combining this compound and isoniazid (3HP) has been shown to be highly effective, allowing for a more manageable treatment course that enhances patient adherence . This combination therapy is especially beneficial for individuals with diabetes, who are at increased risk for progression to active TB .
Pharmacokinetics and Dosing Strategies
Recent studies have focused on the pharmacokinetics of this compound to optimize dosing strategies. Research indicates that weight-based dosing may not be appropriate for all patients, particularly those with lower body weights or those co-infected with HIV. Higher doses may be necessary to achieve therapeutic levels .
Table 1: Pharmacokinetic Characteristics of this compound
| Parameter | Value |
|---|---|
| Bioavailability | Decreases by 27% with HIV infection |
| Clearance Increase | Up to 72% after 21 days |
| Impact of Diet | Increased by 49% with high-fat meals |
| Recommended Dosing | Flat dosing suggested over weight-based |
Case Studies and Clinical Trials
Several clinical trials have explored the safety and efficacy of this compound in various populations:
- Study on Age Impact : A study examined how age affects treatment outcomes with this compound-based weekly therapy. Older patients exhibited different systemic drug reactions compared to younger cohorts, highlighting the need for age-specific treatment protocols .
- Combination Therapy : In trials assessing the combination of this compound with newer anti-TB drugs like SQ109, results indicated that while this compound was safe, its interaction with SQ109 did not enhance bacteriological outcomes significantly .
- Adverse Effects : A rare case of disseminated intravascular coagulation induced by rifampicin therapy was documented, underscoring the importance of monitoring patients for severe adverse effects during treatment .
Immunomodulatory Effects
Emerging research suggests that this compound may have immunomodulatory properties. It has been shown to influence immune pathways, potentially benefiting patients with inflammatory conditions . This aspect opens new avenues for research into its use beyond infectious diseases.
Mechanism of Action
Rifapentine exerts its effects by inhibiting DNA-dependent RNA polymerase in susceptible strains of Mycobacterium tuberculosis . This inhibition leads to the suppression of RNA synthesis, ultimately causing cell death . The molecular target of this compound is the β-subunit of the bacterial RNA polymerase .
Comparison with Similar Compounds
Rifampin: Commonly used in combination therapy for tuberculosis.
Rifabutin: Used primarily in the treatment of Mycobacterium avium complex infections.
Rifapentine’s unique pharmacokinetic properties and its efficacy in shorter treatment regimens make it a valuable addition to the arsenal against tuberculosis .
Q & A
Basic Research Questions
Q. How should researchers design a Phase 2 clinical trial to evaluate rifapentine's efficacy in tuberculosis treatment?
- Methodological Answer : Use a Simon 2-stage adaptive design to balance efficiency and rigor. In the first stage, enroll a small cohort (e.g., 15 participants/arm) and predefine efficacy thresholds (e.g., ≥7/11 participants achieving culture negativity). If met, expand to a second stage (total ~38 participants/arm) for validation. Incorporate a concurrent control arm (e.g., rifampin) for comparative safety and efficacy analysis. This design minimizes resource waste while allowing early termination of underperforming regimens .
Q. What experimental methods are suitable for assessing this compound's cytotoxicity in vitro?
- Methodological Answer : Seed adipose-derived stem cells (ASCs) in 96-well plates (5,000 cells/well) and expose them to this compound concentrations (e.g., 0–30 µg/mL). Use a CCK-8 assay to quantify proliferation and Annexin V/PI staining with flow cytometry to measure apoptosis. Normalize results to untreated controls and validate with triplicate replicates. This approach identifies dose-dependent cytotoxicity thresholds and apoptotic mechanisms .
Q. How do food patterns influence this compound pharmacokinetics in clinical studies?
- Methodological Answer : Conduct a crossover study where participants receive a single 900 mg dose under varying meal conditions (high-fat, low-fat, high-fluid). Collect plasma samples over 72 hours and quantify this compound via HPLC. Use nonlinear mixed-effects modeling (e.g., NONMEM) to estimate bioavailability changes. High-fat meals increase bioavailability by ~49%, while fasting reduces it by 28% .
Q. What statistical methods are appropriate for analyzing time-to-culture conversion in this compound trials?
- Methodological Answer : Apply survival analysis with log-rank tests to compare Kaplan-Meier curves between treatment arms. Use Wilcoxon rank-sum tests for pairwise comparisons of median conversion times. Adjust for covariates (e.g., cavitary lesions, HIV status) via Cox proportional hazards models to isolate this compound's effect .
Advanced Research Questions
Q. How can population pharmacokinetic (PK) models address this compound dosing controversies in HIV-positive patients?
- Methodological Answer : Pool individual PK data from multiple studies (n > 800 participants) and fit a one-compartment model with autoinduction parameters. Incorporate covariates: HIV infection reduces bioavailability by 27%, necessitating a 30% dose increase. Use simulations to compare flat dosing (e.g., 600 mg daily) vs. weight-based regimens, showing flat dosing improves exposure consistency across weight strata .
Q. How do genetic polymorphisms (e.g., AA genotype) affect this compound clearance, and how should dosing be optimized?
- Methodological Answer : Genotype participants for polymorphisms (e.g., SLCO1B1) and measure this compound plasma concentrations. Use multivariate regression to quantify clearance reductions (e.g., 20–30% in AA carriers). Adjust dosing algorithms using Bayesian forecasting to maintain AUC/MIC targets, particularly in populations with high polymorphism prevalence .
Q. What methodologies reconcile conflicting efficacy data between this compound and rifampin in clinical trials?
- Methodological Answer : Conduct dose-ranging trials (e.g., 450–1500 mg/day) with PK/pharmacodynamic (PD) modeling to establish exposure-response relationships. Compare this compound's AUC/MIC ratios to rifampin's, accounting for protein binding differences (95% vs. 80%). Higher this compound doses (e.g., 1200 mg) may overcome lower free drug concentrations, aligning efficacy with rifampin .
Q. How can researchers model this compound's auto-induction of clearance during prolonged therapy?
- Methodological Answer : Develop a time-dependent PK model where clearance increases by 72% over 21 days due to CYP3A4 induction. Use maximum likelihood estimation to fit nonlinear mixed-effects models to longitudinal concentration data. Validate with external datasets to ensure predictive accuracy across diverse cohorts .
Q. What strategies improve in vitro-to-in vivo extrapolation (IVIVE) of this compound's antimycobacterial activity?
- Methodological Answer : Integrate hollow-fiber infection model (HFIM) data with PK/PD models. Expose M. tuberculosis to dynamic this compound concentrations mimicking human plasma profiles. Measure bactericidal activity and resistance emergence. Calibrate models using clinical trial outcomes to refine optimal dosing thresholds .
Q. How should researchers analyze safety endpoints in trials combining this compound with moxifloxacin?
- Methodological Answer : Use competing risks regression to distinguish adverse events (AEs) leading to discontinuation from microbiological failures. Compare AE rates (e.g., grade ≥3 AEs) between arms via Fisher’s exact tests. Adjust for drug-drug interactions (e.g., this compound-induced moxifloxacin clearance) using PK-guided dose adjustments .
Q. Contradiction Analysis
- Lower Efficacy in HIV Patients : Early trials reported reduced this compound exposure in HIV-positive patients due to decreased bioavailability . However, later PK models attribute this to unadjusted dosing and recommend 30% dose escalation, which improves efficacy parity .
- This compound vs. Rifampin : While this compound initially underperformed rifampin in weekly regimens , daily high-dose regimens (600–1200 mg) show comparable or superior efficacy when optimized for exposure .
Properties
Key on ui mechanism of action |
Rifapentine has shown higher bacteriostatic and bactericidal activities especially against intracellular bacteria growing in human monocyte-derived macrophages. Rifapentine inhibits DNA-dependent RNA polymerase in susceptible strains of M. tuberculosis. Rifapentine acts via the inhibition of DNA-dependent RNA polymerase, leading to a suppression of RNA synthesis and cell death. |
|---|---|
CAS No. |
61379-65-5 |
Molecular Formula |
C47H64N4O12 |
Molecular Weight |
877.0 g/mol |
IUPAC Name |
[(7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-26-[(4-cyclopentylpiperazin-1-yl)iminomethyl]-2,15,17,27,29-pentahydroxy-11-methoxy-3,7,12,14,16,18,22-heptamethyl-6,23-dioxo-8,30-dioxa-24-azatetracyclo[23.3.1.14,7.05,28]triaconta-1(29),2,4,9,19,21,25,27-octaen-13-yl] acetate |
InChI |
InChI=1S/C47H64N4O12/c1-24-13-12-14-25(2)46(59)49-37-32(23-48-51-20-18-50(19-21-51)31-15-10-11-16-31)41(56)34-35(42(37)57)40(55)29(6)44-36(34)45(58)47(8,63-44)61-22-17-33(60-9)26(3)43(62-30(7)52)28(5)39(54)27(4)38(24)53/h12-14,17,22-24,26-28,31,33,38-39,43,53-57H,10-11,15-16,18-21H2,1-9H3,(H,49,59)/b13-12+,22-17+,25-14-,48-23?/t24-,26+,27+,28+,33-,38-,39+,43+,47-/m0/s1 |
InChI Key |
WDZCUPBHRAEYDL-LYDPARFQSA-N |
SMILES |
CC1C=CC=C(C(=O)NC2=C(C(=C3C(=C2O)C(=C(C4=C3C(=O)C(O4)(OC=CC(C(C(C(C(C(C1O)C)O)C)OC(=O)C)C)OC)C)C)O)O)C=NN5CCN(CC5)C6CCCC6)C |
Isomeric SMILES |
C[C@H]1/C=C/C=C(\C(=O)NC2=C(C(=C3C(=C2O)C(=C(C4=C3C(=O)[C@](O4)(O/C=C/[C@@H]([C@H]([C@H]([C@@H]([C@@H]([C@@H]([C@H]1O)C)O)C)OC(=O)C)C)OC)C)C)O)O)C=NN5CCN(CC5)C6CCCC6)/C |
Canonical SMILES |
CC1C=CC=C(C(=O)NC2=C(C(=C3C(=C2O)C(=C(C4=C3C(=O)C(O4)(OC=CC(C(C(C(C(C(C1O)C)O)C)OC(=O)C)C)OC)C)C)O)O)C=NN5CCN(CC5)C6CCCC6)C |
Appearance |
Solid powder |
Purity |
>98% (or refer to the Certificate of Analysis) |
shelf_life |
>3 years if stored properly |
solubility |
Soluble in DMSO |
storage |
Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
Synonyms |
Rifapentine; DL 473; DL-473; DL473; R 773; R-773; R773; |
Origin of Product |
United States |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















