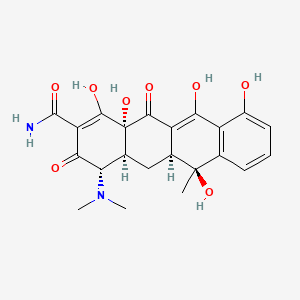
Tetracycline
Overview
Description
Tetracycline is a broad-spectrum antibiotic belonging to the tetracyclines family, which was discovered in the 1940s. It is used to treat a variety of infections caused by bacteria, including acne, cholera, brucellosis, plague, malaria, and syphilis . This compound works by inhibiting protein synthesis in bacteria, making it an effective bacteriostatic agent .
Preparation Methods
Synthetic Routes and Reaction Conditions: Tetracycline can be synthesized through various methods. One common method involves the fermentation of Streptomyces bacteria, which naturally produce this compound. The process includes the extraction and purification of the antibiotic from the bacterial culture .
Industrial Production Methods: In industrial settings, this compound is often produced semi-synthetically. The process begins with the fermentation of Streptomyces bacteria to produce chlorthis compound, which is then chemically modified to produce this compound. This method allows for large-scale production of the antibiotic .
Chemical Reactions Analysis
Types of Reactions: Tetracycline undergoes various chemical reactions, including oxidation, reduction, and substitution reactions. These reactions can alter the chemical structure and properties of this compound, affecting its efficacy and stability .
Common Reagents and Conditions:
Oxidation: this compound can be oxidized using reagents such as hydrogen peroxide or potassium permanganate under acidic conditions.
Reduction: Reduction of this compound can be achieved using reagents like sodium borohydride or lithium aluminum hydride.
Major Products Formed: The major products formed from these reactions include various this compound derivatives, which may have different pharmacological properties and applications .
Scientific Research Applications
Tetracycline has a wide range of scientific research applications:
Chemistry: this compound is used as a model compound in studies of antibiotic synthesis and degradation.
Biology: It is used to study bacterial resistance mechanisms and the effects of antibiotics on microbial communities.
Medicine: this compound is used to treat bacterial infections and is also studied for its potential use in treating other conditions, such as cancer and inflammatory diseases.
Industry: this compound is used in animal husbandry to promote growth and prevent infections in livestock
Mechanism of Action
Tetracycline exerts its effects by binding to the 30S ribosomal subunit of bacteria, preventing the attachment of aminoacyl-tRNA to the ribosome. This inhibits protein synthesis, which is essential for bacterial growth and replication. This compound also binds to the 50S ribosomal subunit to a lesser extent, further disrupting protein synthesis .
Comparison with Similar Compounds
- Doxycycline
- Minocycline
- Tigecycline
- Omadacycline
- Eravacycline
Comparison: Tetracycline and its similar compounds share a common mechanism of action but differ in their pharmacokinetic properties and spectrum of activity. For example, doxycycline has a longer half-life and better absorption compared to this compound, making it more effective for certain infections. Minocycline has better penetration into tissues, while tigecycline is effective against a broader range of resistant bacteria .
This compound remains a valuable antibiotic due to its broad-spectrum activity and relatively low cost. the development of resistance has limited its use, prompting the development of newer this compound derivatives with improved properties .
Biological Activity
Tetracycline is a broad-spectrum antibiotic that has been widely used for the treatment of various bacterial infections. Its biological activity primarily stems from its ability to inhibit protein synthesis in bacteria, making it effective against a wide range of gram-positive and gram-negative organisms. This article delves into the mechanisms of action, resistance patterns, clinical implications, and recent research findings regarding this compound.
This compound exerts its antibacterial effects by binding to the 30S ribosomal subunit of bacterial ribosomes. This binding interferes with the attachment of aminoacyl-tRNA to the ribosome, thereby inhibiting protein synthesis. The specific interaction occurs at a conserved site within the 16S rRNA component of the ribosome, leading to a bacteriostatic effect, although some studies suggest that certain this compound derivatives may exhibit bactericidal properties under specific conditions .
Key Points:
- Binding Site : this compound binds to the 30S ribosomal subunit.
- Mechanism : Inhibits aminoacyl-tRNA binding, blocking protein synthesis.
- Effect : Primarily bacteriostatic, with some bactericidal activity observed.
Biological Activity Spectrum
Tetracyclines are effective against a variety of pathogens, including:
- Gram-positive bacteria : Staphylococcus aureus, Streptococcus pneumoniae
- Gram-negative bacteria : Escherichia coli, Klebsiella pneumoniae
- Atypical organisms : Mycoplasma pneumoniae, Chlamydia trachomatis
The antibiotic's spectrum is significant for treating infections like acne, respiratory tract infections, and certain zoonotic diseases .
Resistance Mechanisms
Resistance to tetracyclines has become a growing concern. The primary mechanisms include:
- Efflux pumps : Bacteria can expel this compound before it exerts its effect.
- Ribosomal protection proteins (RPPs) : These proteins can prevent this compound from binding to the ribosome.
- Enzymatic inactivation : Some bacteria produce enzymes that chemically modify this compound.
Recent studies have highlighted the emergence of resistance genes in clinical isolates, underscoring the need for ongoing surveillance and stewardship efforts .
Case Study: this compound in Treating STIs
A recent pilot trial involving daily doxycycline (a this compound derivative) showed significant reductions in bacterial sexually transmitted infections (STIs) among HIV-negative men who have sex with men (gbMSM). The study reported an odds ratio (OR) of 0.18 for STI incidence in the doxycycline group compared to controls . However, concerns about developing resistance necessitate careful consideration before widespread prophylactic use.
Cohort Study: First Trimester Exposure
A cohort study examined the risks associated with first-trimester exposure to tetracyclines. Findings indicated no significant increase in major congenital malformations (MCMs) among infants exposed to tetracyclines compared to unexposed controls. Although some increased risks were noted for nervous system anomalies, these were not sustained over longer follow-up periods .
Recent Research Findings
Recent studies have expanded our understanding of this compound's biological activity:
- Antitumor Activity : New this compound derivatives are being explored for their potential antitumor effects. For instance, SF2575 has shown promising results in preliminary studies .
- Resistance Patterns : Ongoing research is investigating the molecular basis of resistance mechanisms and their implications for treatment efficacy .
- Structure-Activity Relationships : Novel synthetic derivatives are being developed to enhance antibacterial activity and overcome resistance mechanisms associated with traditional tetracyclines .
Summary Table of this compound Properties
| Property | Description |
|---|---|
| Class | Broad-spectrum antibiotic |
| Mechanism of Action | Inhibits protein synthesis by binding to 30S ribosomal subunit |
| Target Organisms | Gram-positive and negative bacteria; atypical organisms |
| Resistance Mechanisms | Efflux pumps, RPPs, enzymatic inactivation |
| Clinical Uses | Acne treatment, respiratory infections, STIs |
Q & A
Basic Research Questions
Q. What are the primary mechanisms of tetracycline adsorption on engineered materials, and how can adsorption efficiency be optimized experimentally?
Methodological Answer:
Adsorption efficiency depends on factors like pH, adsorbent dose, initial concentration, and contact time. Use Response Surface Methodology (RSM) with a central composite design to model interactions between variables. For example:
- Key Variables : pH (3–7), adsorbent dose (12–40 mg/L), contact time (10–120 min) .
- Statistical Tools : Design-Expert 7.0 or Stat-Ease for ANOVA validation (p < 0.05) .
- Equation : Calculate uptake capacity , where and are initial/equilibrium concentrations (mg/L), is solution volume (L), and is adsorbent mass (g) .
Table 1 : Optimal Adsorption Parameters for Fe-ZIF-8/MWCNTs
| Parameter | Optimal Range | Impact on Efficiency |
|---|---|---|
| pH | 5.0–5.4 | Maximizes π–π interactions |
| Adsorbent Dose | 12–20 mg | Balances active sites |
| Contact Time | 60–120 min | Ensures equilibrium |
Q. How does this compound interact with soil components, and what methods quantify its sorption-desorption dynamics?
Methodological Answer:
this compound binds to Al/Fe hydrous oxides via tricarbonylamide and carbonyl groups, influenced by pH and ionic strength. Use batch equilibrium experiments :
- Steps :
Key Finding : Sorption to Al oxides peaks at pH 7, while Fe oxides show no pH dependency .
Q. What molecular mechanisms underlie bacterial resistance to this compound?
Methodological Answer:
Resistance arises from efflux pumps (e.g., tetA, tetB) or ribosomal protection proteins (e.g., tetM). Use transcriptome sequencing to identify upregulated genes under this compound stress:
- Protocol :
- Culture Lactiplantibacillus plantarum in sub-inhibitory this compound concentrations .
- Extract RNA and perform Illumina sequencing.
- Validate efflux pump activity via RT-qPCR and ethidium bromide accumulation assays .
Table 2 : Transcriptome Data for L. plantarum Under this compound Stress
| Gene | Log2 Fold Change | Function |
|---|---|---|
| tetA | +3.2 | Efflux pump |
| ribosomal rpsL | -1.8 | Target site mutation |
Advanced Research Questions
Q. How can Response Surface Methodology (RSM) resolve contradictions in this compound adsorption studies?
Methodological Answer:
Conflicting results often stem from unoptimized variable interactions. Apply Box-Behnken Design (BBD) with 3–5 factors:
- Case Study : For magnetic nanocomposites, BBD revealed antagonism between pH and contact time, requiring trade-offs for 99.16% removal .
- Validation : Compare experimental vs. predicted values (e.g., in chitosan-graft copolymers) .
Pro Tip : Use Pareto charts to rank factor significance (e.g., pH > adsorbent dose > time) .
Q. How do co-contaminants like glyphosate or MCPA influence this compound’s environmental fate?
Methodological Answer:
Co-contaminants compete for sorption sites or form ternary complexes. Design competitive sorption experiments :
- Steps :
Finding : Phosphate reduces this compound sorption by 40% via ligand competition .
Q. What advanced genetic tools enable this compound-regulated gene expression in eukaryotic systems?
Methodological Answer:
The Tet-Off system allows precise control:
- Protocol :
- Transduce cells with AAV vectors expressing this compound transactivator (tTA) .
- Insert bidirectional promoters with tetO sequences to co-regulate two genes (e.g., reporter + target) .
- Validate with luciferase assays under ±doxycycline conditions.
Application : Used to label hippocampal neurons active during specific behavioral epochs .
Properties
IUPAC Name |
4-(dimethylamino)-1,6,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4,4a,5,5a-tetrahydrotetracene-2-carboxamide | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.10.14) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H24N2O8/c1-21(31)8-5-4-6-11(25)12(8)16(26)13-9(21)7-10-15(24(2)3)17(27)14(20(23)30)19(29)22(10,32)18(13)28/h4-6,9-10,15,25-26,29,31-32H,7H2,1-3H3,(H2,23,30) | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.10.14) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
NWXMGUDVXFXRIG-UHFFFAOYSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.10.14) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1(C2CC3C(C(=O)C(=C(C3(C(=O)C2=C(C4=C1C=CC=C4O)O)O)O)C(=O)N)N(C)C)O | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.10.14) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H24N2O8 | |
| Details | Computed by PubChem 2.2 (PubChem release 2021.10.14) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
444.4 g/mol | |
| Details | Computed by PubChem 2.2 (PubChem release 2021.10.14) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
60-54-8 | |
| Record name | tetracycline | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=108579 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















