Gefitinib
Descripción general
Descripción
Gefitinib, vendido bajo el nombre comercial Iressa, es un medicamento utilizado principalmente para el tratamiento de ciertos tipos de cáncer, incluido el cáncer de pulmón de células no pequeñas y el cáncer de mama. Es un inhibidor selectivo de la tirosina quinasa del receptor del factor de crecimiento epidérmico (EGFR), que juega un papel crucial en la regulación del crecimiento y la supervivencia celular .
Mecanismo De Acción
Gefitinib ejerce sus efectos inhibiendo la tirosina quinasa del EGFR. Se une al sitio de unión del trifosfato de adenosina (ATP) de la enzima, evitando la fosforilación y activación de las vías de señalización aguas abajo. Esta inhibición conduce a la supresión de la proliferación celular y la inducción de apoptosis en células cancerosas con EGFR sobreexpresado o mutado .
Análisis Bioquímico
Biochemical Properties
Gefitinib plays a significant role in biochemical reactions. It interacts with various enzymes, proteins, and other biomolecules. This compound is an inhibitor of the epidermal growth factor receptor (EGFR) tyrosine kinase that binds to the adenosine triphosphate (ATP)-binding site of the enzyme . EGFR is often overexpressed in certain human carcinoma cells, such as lung and breast cancer cells .
Cellular Effects
This compound has profound effects on various types of cells and cellular processes. It influences cell function, including impacts on cell signaling pathways, gene expression, and cellular metabolism. In vitro cytotoxicity studies revealed that this compound enhanced the inhibition of cell proliferation and apoptosis in A549 and H1299 cells compared to free this compound .
Molecular Mechanism
This compound exerts its effects at the molecular level through several mechanisms. It binds to the ATP-binding site of the EGFR tyrosine kinase enzyme, inhibiting its activity . This interaction leads to changes in gene expression and cellular functions .
Métodos De Preparación
Rutas sintéticas y condiciones de reacción: Gefitinib se sintetiza a través de un proceso de varios pasos que comienza con el ácido 4,5-dimetoxi-2-nitrobenzoico. La síntesis involucra varios pasos clave, incluyendo desmetilación, esterificación, alquilación de la cadena lateral, reducción, formación de ciclohexilamina, cloración y sustitución de amoníaco .
Métodos de producción industrial: En entornos industriales, el this compound se produce utilizando rutas sintéticas optimizadas para garantizar un alto rendimiento y pureza. El proceso implica un control estricto de las condiciones de reacción, como la temperatura, el pH y la selección de solventes, para lograr la calidad del producto deseada .
Análisis De Reacciones Químicas
Tipos de reacciones: Gefitinib experimenta varias reacciones químicas, que incluyen:
Oxidación: this compound puede oxidarse para formar diferentes metabolitos.
Reducción: Las reacciones de reducción pueden modificar la estructura del anillo de quinazolina.
Sustitución: Las reacciones de sustitución pueden ocurrir en el anillo aromático, lo que lleva a la formación de derivados.
Reactivos y condiciones comunes:
Oxidación: Los agentes oxidantes comunes incluyen el peróxido de hidrógeno y el permanganato de potasio.
Reducción: Se utilizan agentes reductores como el borohidruro de sodio y el hidruro de aluminio y litio.
Sustitución: Los agentes halogenantes como el cloro y el bromo se emplean a menudo.
Productos principales: Los principales productos formados a partir de estas reacciones incluyen varios derivados de this compound con propiedades farmacológicas alteradas .
Aplicaciones Científicas De Investigación
Non-Small Cell Lung Cancer (NSCLC)
- First-Line Treatment : Gefitinib is approved for use as a first-line treatment in patients with advanced NSCLC harboring sensitive EGFR mutations. Clinical trials have shown that this compound significantly improves progression-free survival compared to standard chemotherapy in this patient population .
- Second-Line Treatment : In cases where patients have previously received chemotherapy, this compound remains an effective option. Studies indicate that it offers better tolerability and quality of life compared to traditional chemotherapy regimens .
- Brain Metastases : One of the notable advantages of this compound is its ability to penetrate the blood-brain barrier effectively. This property makes it a viable option for treating patients with brain metastases from NSCLC, where other therapies may fail to achieve therapeutic concentrations .
Other Cancers
While this compound's primary application is in NSCLC, research is ongoing into its efficacy against other malignancies:
- Head and Neck Cancers : Some studies suggest potential benefits in head and neck squamous cell carcinoma, particularly in tumors expressing high levels of EGFR .
- Combination Therapies : Recent investigations have explored the synergistic effects of this compound when combined with other agents like anlotinib, showing enhanced efficacy against resistant NSCLC cell lines .
Case Study 1: Efficacy in Asian Populations
A significant body of research indicates that this compound is particularly effective among Asian populations with adenocarcinoma histology and those who have never smoked. The IRESSA Pan-Asia Study (IPASS) revealed an overall response rate exceeding 80% in patients with EGFR mutation-positive tumors . This study emphasizes the importance of genetic profiling in optimizing treatment strategies.
Case Study 2: Long-Term Outcomes
A longitudinal study involving patients treated with this compound over several years demonstrated sustained efficacy and manageable side effects. Patients reported improved quality of life metrics compared to those receiving standard chemotherapy regimens .
Adverse Effects
While this compound is generally well-tolerated, it can cause side effects such as skin rash, diarrhea, and liver enzyme elevation. Monitoring and management strategies are essential to mitigate these effects during treatment .
Comparación Con Compuestos Similares
Gefitinib se compara a menudo con otros inhibidores de EGFR, como erlotinib y afatinib. Si bien los tres compuestos se dirigen a la tirosina quinasa del EGFR, difieren en sus propiedades farmacocinéticas y eficacia clínica:
Erlotinib: Mecanismo de acción similar pero tiene un perfil de efectos secundarios y régimen de dosificación diferente.
Afatinib: Inhibidor irreversible de EGFR, que ofrece un espectro de actividad más amplio contra mutaciones de EGFR
Singularidad: this compound es único en su inhibición selectiva de la tirosina quinasa del EGFR y su capacidad para dirigirse a mutaciones específicas en células cancerosas, lo que lo convierte en una herramienta valiosa en la terapia contra el cáncer personalizada .
Compuestos similares:
- Erlotinib
- Afatinib
- Lapatinib
- Osimertinib
Actividad Biológica
Gefitinib, an epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, is primarily used in the treatment of non-small cell lung cancer (NSCLC) with specific EGFR mutations. This article explores the biological activity of this compound, focusing on its mechanisms of action, efficacy in clinical trials, and emerging research findings.
This compound selectively binds to the ATP-binding site of the EGFR tyrosine kinase domain, inhibiting its phosphorylation and subsequent activation of downstream signaling pathways. This results in decreased cell proliferation and increased apoptosis in cancer cells expressing mutant forms of EGFR. The compound has shown significant effects on various cellular processes, including:
- Inhibition of Cell Proliferation : this compound suppresses the growth of cancer cells by blocking EGFR-mediated signaling pathways.
- Induction of Apoptosis : It promotes programmed cell death through alterations in mitochondrial function and expression levels of Bcl-2 family proteins, such as Bcl-xL and Bax .
- Impact on Mitochondrial Activity : Recent studies indicate that this compound enhances mitochondrial functions, such as succinate-tetrazolium reductase (STR) activity, particularly in high-density cell cultures .
Efficacy in Clinical Trials
This compound has been evaluated extensively in clinical trials for its efficacy and safety profile. Key findings from several studies are summarized below:
Case Studies
- Mitochondrial Activity Enhancement : In a study involving HCC827 cells (EGFR mutation positive), this compound was shown to enhance mitochondrial membrane potential and STR activity, indicating its role as a mitochondrial protector during combination therapy with doxorubicin .
- Resistance Development : Despite its initial effectiveness, resistance to this compound often develops within one to two years due to various mechanisms including secondary mutations in the EGFR gene or activation of alternative signaling pathways . A case study highlighted a patient who initially responded well but later exhibited resistance due to a T790M mutation.
- Urothelial Carcinoma : In vitro studies demonstrated this compound's inhibitory effects on growth and invasion in urothelial carcinoma cell lines, suggesting potential applications beyond NSCLC .
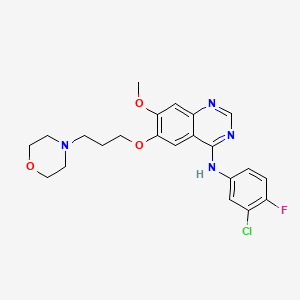
Propiedades
IUPAC Name |
N-(3-chloro-4-fluorophenyl)-7-methoxy-6-(3-morpholin-4-ylpropoxy)quinazolin-4-amine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H24ClFN4O3/c1-29-20-13-19-16(12-21(20)31-8-2-5-28-6-9-30-10-7-28)22(26-14-25-19)27-15-3-4-18(24)17(23)11-15/h3-4,11-14H,2,5-10H2,1H3,(H,25,26,27) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XGALLCVXEZPNRQ-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
COC1=C(C=C2C(=C1)N=CN=C2NC3=CC(=C(C=C3)F)Cl)OCCCN4CCOCC4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H24ClFN4O3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID8041034 | |
| Record name | Gefitinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8041034 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
446.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Gefitinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014462 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Sparingly soluble (|
|
|
| Record name | Gefitinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00317 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Gefitinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014462 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Gefitinib is an inhibitor of the epidermal growth factor receptor (EGFR) tyrosine kinase that binds to the adenosine triphosphate (ATP)-binding site of the enzyme. EGFR is often shown to be overexpressed in certain human carcinoma cells, such as lung and breast cancer cells. Overexpression leads to enhanced activation of the anti-apoptotic Ras signal transduction cascades, subsequently resulting in increased survival of cancer cells and uncontrolled cell proliferation. Gefitinib is the first selective inhibitor of the EGFR tyrosine kinase which is also referred to as Her1 or ErbB-1. By inhibiting EGFR tyrosine kinase, the downstream signaling cascades are also inhibited, resulting in inhibited malignant cell proliferation. | |
| Record name | Gefitinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00317 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
184475-35-2 | |
| Record name | Gefitinib | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=184475-35-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Gefitinib [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0184475352 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Gefitinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00317 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Gefitinib | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759856 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Gefitinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8041034 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Gefitinib | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | GEFITINIB | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/S65743JHBS | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Gefitinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014462 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details







Synthesis routes and methods II
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Descargo de responsabilidad e información sobre productos de investigación in vitro
Tenga en cuenta que todos los artículos e información de productos presentados en BenchChem están destinados únicamente con fines informativos. Los productos disponibles para la compra en BenchChem están diseñados específicamente para estudios in vitro, que se realizan fuera de organismos vivos. Los estudios in vitro, derivados del término latino "in vidrio", involucran experimentos realizados en entornos de laboratorio controlados utilizando células o tejidos. Es importante tener en cuenta que estos productos no se clasifican como medicamentos y no han recibido la aprobación de la FDA para la prevención, tratamiento o cura de ninguna condición médica, dolencia o enfermedad. Debemos enfatizar que cualquier forma de introducción corporal de estos productos en humanos o animales está estrictamente prohibida por ley. Es esencial adherirse a estas pautas para garantizar el cumplimiento de los estándares legales y éticos en la investigación y experimentación.













