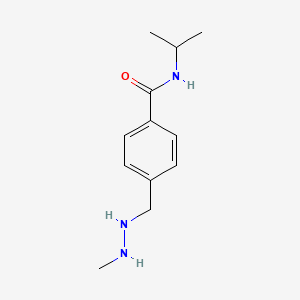
Procarbazine
Descripción general
Descripción
La procarbazina es un medicamento de quimioterapia que se usa principalmente para el tratamiento del linfoma de Hodgkin y ciertos tipos de cáncer cerebral, como el glioblastoma multiforme . Pertenece a la clase de agentes alquilantes, que funcionan agregando grupos alquilo a muchos grupos electronegativos en condiciones presentes en las células . La procarbazina fue aprobada para uso médico en los Estados Unidos en 1969 y está incluida en la Lista de Medicamentos Esenciales de la Organización Mundial de la Salud .
Métodos De Preparación
Rutas sintéticas y condiciones de reacción: La síntesis de procarbazina implica varios pasos que comienzan con el p-toluil aldehído. El proceso incluye la adición de ácido cianúrico dibromo e isopropilamina para obtener toluil isopropilamina. Este intermedio se disuelve luego en un reactivo orgánico, seguido de la adición de N-bromo-succinimida y un iniciador. La mezcla se calienta a reflujo y se elimina el solvente. Se añaden acetonitrilo y un agente acelerador hidrolítico, y la mezcla se calienta a reflujo para formar formoxil benzoil isopropil amina. Finalmente, la formoxil benzoil isopropil amina se hace reaccionar con sulfato de metilhidrazinio y trietilamina, seguida de la adición de cianoborohidruro de sodio, lo que da como resultado la formación de procarbazina .
Métodos de producción industrial: La producción industrial de procarbazina sigue rutas sintéticas similares pero se optimiza para obtener mayores rendimientos y eficiencia. El proceso evita el uso de oxidantes fuertes y ácidos fuertes, lo que lo hace más ecológico. La tasa de recuperación total del método industrial es aproximadamente del 52.9% .
Análisis De Reacciones Químicas
Tipos de reacciones: La procarbazina experimenta diversas reacciones químicas, que incluyen oxidación, reducción y sustitución. Una reacción notable es su autooxidación para formar un derivado azoico, que luego se isomeriza a una hidrazona. Esta hidrazona se somete a hidrólisis para producir un derivado de benzaldehído y metilhidrazina .
Reactivos y condiciones comunes: Los reactivos comunes utilizados en las reacciones que involucran procarbazina incluyen N-bromo-succinimida, acetonitrilo, agentes aceleradores hidrolíticos, sulfato de metilhidrazinio y cianoborohidruro de sodio . Las condiciones de reacción generalmente involucran calentamiento a reflujo y el uso de solventes orgánicos.
Productos principales: Los productos principales formados a partir de las reacciones de procarbazina incluyen derivados de benzaldehído y metilhidrazina .
Aplicaciones Científicas De Investigación
Clinical Applications
Procarbazine is primarily indicated for:
- Hodgkin Lymphoma : Historically used in combination regimens such as MOPP (mechlorethamine, vincristine, this compound, and prednisone) and ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine). Recent studies have reaffirmed its efficacy when combined with newer agents .
- Non-Hodgkin Lymphoma : Used in various treatment protocols, particularly for aggressive forms of the disease .
- Brain Tumors : Effective in treating gliomas and other central nervous system malignancies. This compound is often part of multi-drug regimens aimed at improving patient outcomes .
- Melanoma and Lung Cancer : Although less common, this compound has shown some efficacy in these cancers as well .
Immunosuppressive Properties
Recent studies have highlighted this compound's immunosuppressive effects. It has been shown to inhibit lymphocyte proliferation in experimental models of arthritis, suggesting potential applications in autoimmune diseases . However, this property necessitates careful monitoring due to the risk of increased infections in treated patients.
Comparative Studies and Efficacy
A significant body of research has compared this compound with other chemotherapeutic agents:
- A phase II study comparing this compound with temozolomide (another alkylating agent) demonstrated that temozolomide had superior progression-free survival rates in patients with glioblastoma multiforme . This raises questions about the ongoing role of this compound in modern treatment protocols.
- A recent nested case-control study indicated that this compound use is associated with increased mutation burdens and novel mutational signatures in Hodgkin lymphoma survivors. This finding raises concerns about long-term genomic health and hereditary implications for offspring .
Case Studies
| Study | Cancer Type | Treatment Regimen | Findings |
|---|---|---|---|
| Matche et al. (1963) | Hodgkin Lymphoma | MOPP | Effective response rates observed; established this compound's role in combination therapy. |
| Sertoli et al. (1994) | Non-Hodgkin Lymphoma | ProMACE-MOPP vs MACOP-B | ProMACE-MOPP showed comparable efficacy but was less aggressive than MACOP-B. |
| Yung et al. (2000) | Glioblastoma Multiforme | This compound vs Temozolomide | Temozolomide demonstrated superior progression-free survival compared to this compound. |
Mecanismo De Acción
El mecanismo de acción preciso de la procarbazina no se comprende completamente. se sabe que inhibe la síntesis de proteínas, ARN y ADN al interferir con la transmetilación de los grupos metilo de la metionina en el ARN de transferencia . La procarbazina también funciona como un agente alquilante, metilando la guanina en la posición O-6, lo que lleva a la ruptura del ADN e inhibición de la síntesis de ARN y proteínas .
Comparación Con Compuestos Similares
Compuestos similares: Los compuestos similares a la procarbazina incluyen dacarbazina, bleomicina y nivolumab . Estos compuestos también se utilizan en el tratamiento de diversos cánceres y tienen mecanismos de acción similares como agentes alquilantes o antineoplásicos.
Singularidad: La procarbazina es única en su capacidad para ser utilizada en combinación con otros agentes quimioterapéuticos, como la clormetina, la vincristina y la prednisona, para el tratamiento del linfoma de Hodgkin . También se distingue por su interacción con las nanoestructuras para sistemas de administración de fármacos, lo que aumenta su eficacia en la focalización de las células cancerosas .
Actividad Biológica
Procarbazine is an alkylating agent primarily used in the treatment of certain types of cancer, particularly Hodgkin's lymphoma and brain tumors. Its biological activity is characterized by its mechanisms of action, pharmacokinetics, therapeutic uses, and associated risks. This article delves into these aspects, supported by data tables and relevant case studies.
This compound acts as a cell cycle phase-nonspecific pro-drug and is a derivative of hydrazine. Its precise mechanism remains somewhat ambiguous; however, several key actions have been identified:
- Inhibition of Nucleic Acid Synthesis : this compound inhibits protein, RNA, and DNA synthesis, which is critical for cancer cell proliferation. This inhibition may occur through the disruption of transmethylation processes essential for t-RNA function .
- Free Radical Formation : The drug generates reactive intermediates, including hydrogen peroxide, which can cause direct DNA damage and cross-linking .
- Monoamine Oxidase Inhibition : this compound also exhibits properties as a monoamine oxidase (MAO) inhibitor, which can influence neurotransmitter levels .
Pharmacokinetics
- Absorption : this compound is rapidly absorbed when administered orally, achieving peak plasma concentrations within one hour .
- Metabolism : It undergoes extensive hepatic metabolism to form active metabolites like methylazoxythis compound, which are responsible for its cytotoxic effects. The primary pathway involves oxidation to azothis compound followed by further N-oxidation .
- Excretion : Approximately 25-70% of the drug is excreted in urine within 24 hours, primarily as inactive metabolites .
Therapeutic Uses
This compound is utilized in various cancer treatments:
- Hodgkin's Lymphoma : Often used in combination with other agents (e.g., BCNU and vincristine) for enhanced efficacy .
- Brain Tumors : Effective in treating glioblastoma multiforme when combined with other chemotherapeutic agents like temozolomide .
- Other Cancers : It has shown effectiveness against non-Hodgkin’s lymphoma, melanoma, and multiple myeloma .
Efficacy Comparison with Temozolomide
A notable study compared this compound with temozolomide (TMZ) in patients with recurrent glioblastoma multiforme. The results indicated:
| Treatment | 6-Month PFS Rate | Overall Survival Rate |
|---|---|---|
| This compound | 8% | 44% |
| Temozolomide | 21% | 60% |
The findings highlighted that TMZ significantly outperformed this compound in terms of progression-free survival (PFS) and overall survival rates (OS) .
Risk Assessment in Hodgkin's Lymphoma Survivors
A recent nested case-control study examined the association between this compound dosage and colorectal cancer risk among Hodgkin lymphoma survivors. Key findings included:
- Increased colorectal cancer rates correlated with higher doses of this compound.
- The excess rate ratio (ERR) for colorectal cancer increased linearly with this compound dosage, indicating a potential long-term risk associated with its use .
Safety Profile and Side Effects
While this compound is effective, it is not without risks:
Q & A
Basic Research Questions
Q. What molecular markers are critical for stratifying patients in Procarbazine-based clinical trials for low-grade gliomas (LGG)?
- Answer: Key markers include IDH1/2 mutation status and 1p/19q chromosome codeletion . IDH-mutant, 1p/19q-codeleted tumors show prolonged progression-free survival (PFS) and overall survival (OS) with adjuvant PCV (this compound, lomustine, vincristine) . These markers are assessed via immunohistochemistry (IDH1 R132H) and fluorescence in situ hybridization (FISH) for 1p/19q status. Researchers should stratify trials by these subtypes to avoid confounding results, as IDH-wildtype LGGs have distinct biological behaviors .
Q. How do survival outcomes (PFS/OS) for this compound compare to Temozolomide (TMZ) in LGGs?
- Answer: PCV demonstrates superior PFS (median 124.8 months) and OS (median 120 months) in IDH-mutant, 1p/19q-codeleted LGGs compared to TMZ (median PFS: 31–54 months) . However, TMZ has lower toxicity (grade 3–4 hematologic toxicity: 9% vs. 15% with PCV) . Methodologically, survival data should be analyzed using Kaplan-Meier curves with log-rank tests, adjusting for molecular subtypes .
Q. What are the primary toxicity considerations when designing this compound-based trials?
- Answer: PCV regimens commonly induce hematologic toxicity (e.g., leukopenia, thrombocytopenia) in 15% of patients and gastrointestinal effects (nausea, constipation). Dose reductions or delays are required in 15% of cases . Toxicity monitoring should follow CTCAE guidelines, with CBCs every 2–4 weeks. Comparatively, TMZ trials prioritize neurocognitive and quality-of-life assessments due to its better tolerability .
Advanced Research Questions
Q. How can researchers reconcile conflicting survival data between historical and contemporary this compound studies?
- Answer: Historical studies often lack molecular stratification (e.g., IDH/1p/19q status), leading to heterogenous cohorts. To resolve contradictions, re-analyze legacy data using modern molecular criteria or conduct meta-regressions adjusting for subtype prevalence. For example, IDH-wildtype LGGs (historically grouped with IDH-mutant) have worse outcomes, skewing earlier PCV trial results .
Q. What experimental models elucidate this compound’s mechanism of action and resistance?
- Answer: In vitro models using rat liver microsomes and isolated hepatocytes demonstrate this compound’s metabolism into methyl radicals via cytochrome P450 (CYP2B/IA isoforms), detected via electron spin resonance (ESR) with spin-trapping agents (e.g., 4-POBN) . Resistance mechanisms can be studied in IDH-mutant glioma cell lines exposed to chronic this compound, assessing DNA repair pathways (e.g., MGMT expression) .
Q. What methodological challenges arise in designing prospective trials comparing PCV and TMZ?
- Answer: Key challenges include:
- Molecular heterogeneity : Stratification by IDH/1p/19q status requires large cohorts (n > 500) for adequate power.
- Toxicity vs. efficacy trade-offs : Use composite endpoints (e.g., PFS-adjusted toxicity scores) to balance survival benefits against adverse events .
- Longitudinal follow-up : LGG trials require >10-year follow-up to assess OS, necessitating adaptive trial designs with interim analyses .
Q. How does this compound’s interaction with semicarbazide-sensitive amine oxidase (SSAO) influence its therapeutic index?
- Answer: this compound inhibits SSAO non-competitively via a "suicide" mechanism, disrupting amine metabolism in neural tissues. This interaction may contribute to neurotoxicity. Researchers should measure SSAO activity in patient serum pre-/post-treatment using benzylamine deamination assays and correlate with adverse event profiles .
Q. Data Analysis & Interpretation
Q. What statistical methods are optimal for analyzing this compound trial data with censored survival outcomes?
- Answer: Use Kaplan-Meier estimators for PFS/OS visualization, stratified by molecular subtypes. For multivariate analysis, employ Cox proportional hazards models incorporating covariates like age, resection extent, and MGMT methylation. Competing risks analysis (e.g., Fine-Gray model) is critical for trials where non-cancer deaths are common .
Q. How should researchers address potential hypermutation phenotypes induced by TMZ in comparative studies with this compound?
- Answer: Hypermutation (e.g., from TMZ-induced MGMT silencing) can confound survival outcomes. Pre-screen tumors for mismatch repair (MMR) deficiencies via PCR or sequencing. In analysis, exclude hypermutated cases or adjust for tumor mutational burden as a covariate .
Propiedades
IUPAC Name |
4-[(2-methylhydrazinyl)methyl]-N-propan-2-ylbenzamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C12H19N3O/c1-9(2)15-12(16)11-6-4-10(5-7-11)8-14-13-3/h4-7,9,13-14H,8H2,1-3H3,(H,15,16) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
CPTBDICYNRMXFX-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)NC(=O)C1=CC=C(C=C1)CNNC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C12H19N3O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
366-70-1 (mono-hydrochloride) | |
| Record name | Procarbazine [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000671169 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID4021189 | |
| Record name | Procarbazine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4021189 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
221.30 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Procarbazine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015299 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
In water, 1,400 mg/L @ 25 °C /Estimated/, 2.28e-01 g/L | |
| Record name | Procarbazine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01168 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PROCARBAZINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3250 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Procarbazine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015299 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Vapor Pressure |
8.4X10-7 mm Hg @ 25 °C /Estimated/ | |
| Record name | PROCARBAZINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3250 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
The precise mode of cytotoxic action of procarbazine has not been clearly defined. There is evidence that the drug may act by inhibition of protein, RNA and DNA synthesis. Studies have suggested that procarbazine may inhibit transmethylation of methyl groups of methionine into t-RNA. The absence of functional t-RNA could cause the cessation of protein synthesis and consequently DNA and RNA synthesis. In addition, procarbazine may directly damage DNA. Hydrogen peroxide, formed during the auto-oxidation of the drug, may attack protein sulfhydryl groups contained in residual protein which is tightly bound to DNA., Procarbazine is an alkylating agent. The exact mechanism of antineoplastic action is unknown but is thought to resemble that of the alkylating agents; procarbazine is cell cycle-specific for the S phase of cell division. Procarbazine is thought to inhibit DNA, RNA, and protein synthesis., O-6-Methylguanine was measured in blood leukocyte DNA of seven patients with Hodgkin's or non-Hodgkin's lymphoma during therapeutic exposure to procarbazine involving three daily p.o. doses (50 mg each) for 10 days (corresponding to 2.1 mg/kg/day for a 70-kg human). Adduct accumulation was observed in all seven cases, reaching levels up to 0.28 fmol/microgram of DNA (0.45 umol/mol of guanine). In one individual, maximal levels of adduct were reached after 7 days of exposure, followed by a steady decline, whereas in all other individuals continuous accumulation was observed throughout the exposure period. In four individuals for which data were available for day 11 (12 to 16 hr after the final intake of procarbazine), decreased amounts of O-6-methylguanine were observed relative to the last previous measurements. The accumulation of O-6-methylguanine was linearly correlated with the cumulative dose of procarbazine, with a slope of 0.011 fmol of O-6-methylguanine/microgram of DNA per mg/kg of body weight or 2.68x10-4 fmol of O-6-methylguanine DNA per mg/sq m. Two hr after the administration of single p.o. doses of l to 10 mg/kg of procarbazine to rats, O-6-methylguanine formation in leukocyte DNA was just under half that in liver DNA and showed a linear relationship with dose with a slope of 0.017 fmol/microgram of DNA per mg/kg of body weight or 5.67x10-4 fmol of O-6-methylguanine/microgram of DNA per mg/sq m. A negative correlation between the rate of accumulation of O-6-methylguanine in different individuals and lymphocyte O-6-alkylguanine-DNA alkyltransferase was observed, demonstrating a probable protective effect of O-6-alkylguanine-DNA alkyltransferase against the accumulation of O-6-methylguanine during exposure to methylating agents. This observation supports the suggestion of a possible role of procarbazine-induced O-6-methylguanine in the pathogenesis of acute nonlymphocytic leukemia appearing after treatment with chemotherapeutic protocols which include procarbazine, based on the finding of low lymphocyte O-6-alkylguanine-DNA alkyltransferase levels in patients with such therapy-related neoplastic disease. Lymphocyte O-6-alkylguanine-DNA alkyltransferase levels were mainly in the range of 5 to 10 fmol/micrograms of DNA and showed no consistent variation during procarbazine exposure., Procarbazine causes weak inhibition of monoamine oxidase (MAO). MAO inhibitors prevent the inactivation of tyramine by hepatic and gastrointestinal monoamine oxidase. Tyramine in the bloodstream releases norepinephrine from the sympathetic nerve terminals and produces a sudden increase in blood pressure. | |
| Record name | Procarbazine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01168 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PROCARBAZINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3250 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
671-16-9, 366-70-1 | |
| Record name | Procarbazine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=671-16-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Procarbazine [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000671169 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Procarbazine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01168 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Procarbazine hydrochloride | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759626 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Procarbazine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4021189 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Procarbazine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.010.531 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PROCARBAZINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/35S93Y190K | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | PROCARBAZINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3250 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Procarbazine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015299 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
223 °C | |
| Record name | Procarbazine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01168 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Procarbazine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015299 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Descargo de responsabilidad e información sobre productos de investigación in vitro
Tenga en cuenta que todos los artículos e información de productos presentados en BenchChem están destinados únicamente con fines informativos. Los productos disponibles para la compra en BenchChem están diseñados específicamente para estudios in vitro, que se realizan fuera de organismos vivos. Los estudios in vitro, derivados del término latino "in vidrio", involucran experimentos realizados en entornos de laboratorio controlados utilizando células o tejidos. Es importante tener en cuenta que estos productos no se clasifican como medicamentos y no han recibido la aprobación de la FDA para la prevención, tratamiento o cura de ninguna condición médica, dolencia o enfermedad. Debemos enfatizar que cualquier forma de introducción corporal de estos productos en humanos o animales está estrictamente prohibida por ley. Es esencial adherirse a estas pautas para garantizar el cumplimiento de los estándares legales y éticos en la investigación y experimentación.
















