Bendamustine
Vue d'ensemble
Description
Bendamustine est un médicament de chimiothérapie principalement utilisé dans le traitement de la leucémie lymphoïde chronique, du myélome multiple et du lymphome non hodgkinien . Il appartient à la classe des agents alkylants et agit en interférant avec la fonction de l’ADN et de l’ARN, ce qui conduit à la mort cellulaire . This compound a été synthétisé pour la première fois au début des années 1960 en République démocratique allemande et a été approuvé pour un usage médical aux États-Unis en 2008 .
Méthodes De Préparation
Voies de synthèse et conditions réactionnelles : Le chlorhydrate de bendamustine peut être synthétisé par un processus en plusieurs étapes. L’intermédiaire clé, le butanoate de 1-méthyl-5-[bis(2-chloroéthyl)amino]-1H-benzimidazol-2-yl]lithium, est préparé puis converti en chlorhydrate de this compound . Le processus implique l’utilisation de divers réactifs et solvants, notamment l’acide trifluoroacétique et l’acétonitrile .
Méthodes de production industrielle : La production industrielle du chlorhydrate de this compound vise une pureté élevée (≥ 99 %) et implique des étapes telles que la chromatographie liquide haute performance (HPLC) pour la purification . Le processus est conçu pour être économique et respectueux de l’environnement, évitant l’utilisation de produits chimiques dangereux .
Analyse Des Réactions Chimiques
Hydrolysis Reactions
Bendamustine undergoes rapid hydrolysis, primarily non-enzymatic, via cleavage of its mechlorethamine group (Fig. 1 ):
-
Primary metabolites :
-
Monohydroxy-bendamustine (HP1)
-
Dihydroxy-bendamustine (HP2)
-
-
Conditions : Hydrolysis occurs spontaneously in aqueous solutions, accelerated at physiological pH (7.4) and temperature (37°C) .
-
Activity : HP1 and HP2 exhibit minimal cytotoxicity (<1% potency of parent compound) .
Table 1: Hydrolysis Pathways and Metabolite Characteristics
| Reaction | Enzyme Involved | Metabolite | Cytotoxic Activity | Plasma Concentration Ratio (vs. Parent) |
|---|---|---|---|---|
| Non-enzymatic hydrolysis | None | HP1, HP2 | Negligible | HP1: ~10%, HP2: ~5% |
Oxidative Metabolism
This compound is metabolized by CYP1A2 into two active intermediates:
-
γ-Hydroxythis compound (M3) : Formed via γ-oxidation of the butyric acid side chain.
-
N-Desmethyl-bendamustine (M4) : Produced by demethylation of the benzimidazole ring .
Key Findings:
-
Potency : M3 retains ~100% potency of this compound, while M4 is 5–10-fold less active .
-
Plasma levels : M3 and M4 concentrations are 1/10th and 1/100th of this compound, respectively, limiting their therapeutic impact .
-
Minor pathway : CYP1A2 contributes <5% to total metabolism, with hydrolysis dominating .
Synthetic Routes
The industrial synthesis of this compound hydrochloride involves:
-
Alkylation : Reaction of 4-[1-methyl-5-amino-benzimidazol-2-yl]butyric acid ethyl ester with 2-chloroethanol.
-
Chlorination : Treatment with thionyl chloride (SOCl₂) to replace hydroxyl groups with chlorine atoms.
-
Hydrolysis : Conversion of the ethyl ester to the carboxylic acid using lithium hydroxide (LiOH) .
Table 2: Key Synthetic Steps and Conditions
Degradation and Stability
This compound is highly unstable in aqueous media, degrading into non-therapeutic products:
-
Primary degradation pathways :
-
Stabilization : Supplied as a lyophilized powder to prevent hydrolysis; reconstituted solutions degrade within 1–3 hours .
Table 3: Stability Under Different Conditions
| Condition | Degradation Rate | Major Degradants |
|---|---|---|
| Aqueous solution | Rapid (t₁/₂: 40 min) | HP1, HP2, oxidized species |
| Lyophilized form | Stable (>24 months) | None |
| Acidic pH (pH 4) | Accelerated | HP1, HP2 |
Conjugative Pathways
Limited data suggest involvement of phase II enzymes (e.g., UGTs, GSTs) in minor conjugative metabolism, producing inactive glucuronides or glutathione adducts .
Applications De Recherche Scientifique
Clinical Applications
Bendamustine is primarily utilized in the treatment of:
- Chronic Lymphocytic Leukemia (CLL) : It has shown significant efficacy in patients with relapsed or refractory CLL, particularly when combined with rituximab. Studies indicate an overall response rate exceeding 90% in such cases .
- Non-Hodgkin Lymphoma (NHL) : this compound is effective against various subtypes of NHL, including indolent forms. Clinical trials have demonstrated its ability to induce remissions even in patients resistant to other therapies .
- Multiple Myeloma : Research indicates potential benefits when used in combination with dexamethasone for treating relapsed or refractory multiple myeloma .
- Chronic Cold Agglutinin Disease : this compound combined with rituximab has resulted in high response rates and sustained remissions in patients with this condition .
Efficacy Data
| Condition | Treatment Regimen | Overall Response Rate (%) | Median Duration of Response (months) |
|---|---|---|---|
| Chronic Lymphocytic Leukemia | This compound + Rituximab | >90 | 23-24 |
| Non-Hodgkin Lymphoma | This compound Alone | 75 | 9.2 |
| Multiple Myeloma | This compound + Dexamethasone | 57 (PR or better) | Not specified |
| Chronic Cold Agglutinin Disease | This compound + Rituximab | 71 | Not specified |
Safety Data
This compound is generally well-tolerated, though it is associated with certain adverse effects:
- Hematologic Toxicities : Neutropenia (61%), thrombocytopenia (25%), and anemia (10%) are common .
- Non-Hematologic Adverse Events : Nausea (77%), infections (69%), fatigue (64%), and gastrointestinal symptoms are frequently reported .
Case Studies
-
Indolent B-cell Lymphoma :
A multicenter study involving 100 patients demonstrated a 75% overall response rate with acceptable toxicity levels. The median duration of response was noted at 9.2 months, indicating this compound's effectiveness in this population . -
Chronic Cold Agglutinin Disease :
In a study involving 32 patients treated with this compound and rituximab, 71% responded to treatment, with notable improvements in hemoglobin levels among complete responders . -
Systemic Light-chain Amyloidosis :
A phase II trial assessed this compound combined with dexamethasone in 31 patients, achieving a partial response rate of 57%, highlighting its potential beyond traditional hematological malignancies .
Mécanisme D'action
Bendamustine exerce ses effets par le biais de plusieurs mécanismes :
Alkylation : Il forme des liaisons croisées intra- et inter-brin entre les bases de l’ADN, ce qui conduit à la mort cellulaire.
Activation des réponses aux dommages de l’ADN : This compound active les réponses de stress aux dommages de l’ADN et l’apoptose.
Inhibition des points de contrôle mitotique : Il inhibe les points de contrôle mitotique et induit une catastrophe mitotique.
Cibles moléculaires : Les principales cibles sont les bases de l’ADN, ce qui conduit à la perturbation de la réplication et de la transcription de l’ADN.
Comparaison Avec Des Composés Similaires
Bendamustine est unique en raison de sa nature bifonctionnelle, combinant des propriétés d’agents alkylants et d’analogues de la purine . Les composés similaires comprennent :
Cyclophosphamide : Un autre agent alkylant utilisé en chimiothérapie.
Melphalan : Un agent alkylant utilisé pour le myélome multiple.
Chlorambucil : Utilisé pour la leucémie lymphoïde chronique.
Comparé à ces composés, la this compound a montré une stabilité accrue et une gamme d’activité plus large contre les cellules quiescentes et en division .
Activité Biologique
Bendamustine is a unique alkylating agent that has gained attention for its distinct biological activity and clinical efficacy, particularly in treating hematologic malignancies. This article provides a comprehensive overview of its biological mechanisms, clinical applications, and research findings, supported by data tables and case studies.
This compound exhibits a multifaceted mechanism of action that differentiates it from traditional alkylating agents. Key mechanisms include:
- DNA Damage and Repair : this compound induces DNA damage through alkylation, leading to the activation of DNA-damage stress responses. Unlike conventional alkylators, it activates a base excision repair pathway rather than relying on alkyltransferase mechanisms .
- Apoptosis Induction : The compound promotes apoptosis in cancer cells by activating caspases (e.g., caspase-3 and caspase-8), which are crucial for programmed cell death. This effect has been observed in various cell lines, including multiple myeloma and lymphomas .
- Cell Cycle Arrest : this compound inhibits mitotic checkpoints, leading to mitotic catastrophe. It has been shown to induce G2 phase arrest in several cancer cell lines by inhibiting proteins involved in mitosis such as Polo-like kinase 1 and Aurora kinase A .
Clinical Efficacy
This compound has demonstrated significant clinical efficacy in various malignancies, particularly non-Hodgkin lymphoma (NHL) and chronic lymphocytic leukemia (CLL). Below is a summary of notable clinical studies.
Case Study Data
Pharmacokinetics
This compound is primarily metabolized via hydrolysis, yielding less active metabolites (HP1 and HP2). It is also processed by CYP1A2 enzymes into active metabolites M3 and M4, with M3 having comparable potency to the parent drug while M4 is significantly less potent . The pharmacokinetic profile indicates that the active metabolites reach peak concentrations alongside this compound, contributing to its therapeutic effects.
Safety Profile
This compound is generally well tolerated, with a safety profile that includes common chemotherapy-related adverse effects such as myelosuppression. In clinical trials, dose-limiting toxicities have been primarily related to prolonged myelosuppression rather than severe organ toxicity .
Comparative Studies
Recent studies have compared this compound with other therapeutic agents:
- This compound vs. Ibrutinib : In patients with relapsed or refractory CLL, this compound combined with rituximab showed comparable efficacy to newer agents like ibrutinib but with different toxicity profiles .
- Synergistic Effects : this compound has shown synergistic effects when combined with other chemotherapeutics, enhancing overall efficacy against resistant cancer types .
Q & A
Basic Research Questions
Q. What are the established experimental protocols for evaluating Bendamustine's cytotoxicity in vitro, and how can variability in assay conditions impact reproducibility?
- Methodological Answer: Standard protocols involve cell viability assays (e.g., MTT, CCK-8) with controlled variables like cell density (5,000–10,000 cells/well), drug exposure time (24–72 hours), and dose ranges (0.1–100 µM). Variability arises from differences in cell passage numbers, serum concentrations, or incubation temperatures. To ensure reproducibility, document batch-specific reagent details and use internal controls (e.g., cisplatin as a positive control) .
Q. What mechanistic evidence supports this compound's dual alkylating and antimetabolite activity, and how can this be validated in new cellular models?
- Methodological Answer: this compound induces DNA crosslinking (via alkylation) and inhibits purine synthesis (via purine analog activity). Validate mechanisms using:
- Comet assays to detect DNA damage.
- LC-MS/MS to quantify intracellular metabolites (e.g., dATP depletion).
- siRNA knockdown of repair pathways (e.g., BRCA1) to assess synthetic lethality .
Q. Which pharmacokinetic parameters are critical for designing this compound dosing regimens in preclinical studies?
- Methodological Answer: Key parameters include plasma half-life (t½), volume of distribution (Vd), and clearance (CL). Use non-compartmental analysis (NCA) of plasma concentration-time curves from murine models. Adjust doses based on allometric scaling to human equivalents (e.g., 120 mg/m<sup>2</sup> in mice ≈ 90 mg/m<sup>2</sup> in humans) .
Advanced Research Questions
Q. How can researchers resolve contradictions in this compound efficacy data across heterogeneous cancer subtypes (e.g., CLL vs. lymphoma)?
- Methodological Answer: Apply meta-analysis with subgroup stratification by:
- Genetic biomarkers (e.g., TP53 mutations in CLL).
- Tumor microenvironment (e.g., hypoxia-inducible factor levels).
- Dose-intensity adjustments (e.g., 70 mg/m<sup>2</sup> vs. 100 mg/m<sup>2</sup>). Use Cochran’s Q test to assess heterogeneity and random-effects models for pooled effect sizes .
Q. What experimental design optimizes the study of this compound combination therapies with novel agents (e.g., BTK inhibitors) while avoiding synergistic toxicity?
- Methodological Answer: Implement sequential vs. concurrent dosing in vitro/vivo:
- Isobologram analysis to classify synergy (CI <1) or antagonism (CI >1).
- Toxicity profiling via liver enzyme assays (ALT/AST) and hematopoietic colony-forming unit (CFU) assays.
- Pharmacodynamic biomarkers (e.g., phospho-BTK inhibition) to correlate efficacy with target engagement .
Q. How can researchers address variability in this compound resistance mechanisms when developing predictive in vitro models?
- Methodological Answer: Use CRISPR-Cas9 screens to identify resistance drivers (e.g., ABCC1 efflux pumps). Validate with:
- RNA-seq of resistant clones to map upregulated pathways.
- 3D co-culture models incorporating stromal cells to mimic tumor microenvironment-mediated resistance.
- Machine learning (e.g., LASSO regression) to prioritize biomarkers from multi-omics datasets .
Q. Methodological Guidelines for Data Validation
Q. What analytical techniques are essential for characterizing this compound stability in biological matrices during pharmacokinetic studies?
- Methodological Answer:
- HPLC-UV/LC-MS with deuterated internal standards (e.g., this compound-d4) to account for matrix effects.
- Forced degradation studies (pH, temperature, light) to identify degradation products.
- Stability criteria : ≤15% deviation in peak area ratios under storage conditions (-80°C, 3 freeze-thaw cycles) .
Q. How should researchers design longitudinal studies to assess this compound-induced immunosuppression in murine models?
- Methodological Answer: Track immune subsets via flow cytometry (CD4<sup>+</sup>/CD8<sup>+</sup> T cells, B220<sup>+</sup> B cells) at baseline, 7 days, and 21 days post-treatment. Use mixed-effects models to account for inter-individual variability. Include CFSE dilution assays to measure T-cell proliferation .
Q. Ethical and Reproducibility Considerations
Q. What informed consent elements are critical for secondary use of this compound trial biospecimens in translational research?
- Methodological Answer: Consent forms must specify:
- Future genomic/proteomic analyses .
- Data sharing (e.g., dbGaP repositories).
- Withdrawal rights . Use broad consent frameworks compliant with GDPR/HIPAA and IRB-approved protocols .
Q. How can researchers enhance transparency in reporting this compound study outcomes to mitigate publication bias?
- Methodological Answer: Adhere to ARRIVE 2.0 guidelines for preclinical studies and CONSORT for clinical trials. Pre-register protocols on ClinicalTrials.gov or OSF , and publish negative results in repositories like Figshare . Disclose all conflicts of interest and funding sources .
Tables for Quick Reference
| Assay Type | Key Parameters | References |
|---|---|---|
| Cytotoxicity (MTT) | Cell density, serum %, IC50 | |
| Pharmacokinetics (NCA) | t½, AUC, Cmax | |
| Synergy (Isobologram) | Combination Index (CI), dose reduction index |
Propriétés
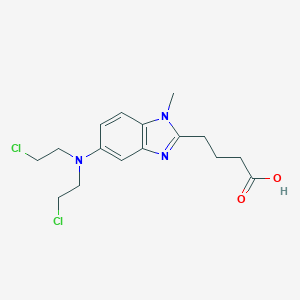
IUPAC Name |
4-[5-[bis(2-chloroethyl)amino]-1-methylbenzimidazol-2-yl]butanoic acid | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C16H21Cl2N3O2/c1-20-14-6-5-12(21(9-7-17)10-8-18)11-13(14)19-15(20)3-2-4-16(22)23/h5-6,11H,2-4,7-10H2,1H3,(H,22,23) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
YTKUWDBFDASYHO-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CN1C2=C(C=C(C=C2)N(CCCl)CCCl)N=C1CCCC(=O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C16H21Cl2N3O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
3543-75-7 (hydrochloride) | |
| Record name | Bendamustine [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0016506277 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID2046888 | |
| Record name | Bendamustine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2046888 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
358.3 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
Bendamustine is a bifunctional mechlorethamine derivative capable of forming electrophilic alkyl groups that covalently bond to other molecules. Through this function as an alkylating agent, bendamustine causes intra- and inter-strand crosslinks between DNA bases resulting in cell death. It is active against both active and quiescent cells, although the exact mechanism of action is unknown., Multiple myeloma is a fatal hematological disease caused by malignant transformation of plasma cells. Bendamustine has been proven to be a potent alternative to melphalan in phase 3 studies, yet its molecular mode of action is still poorly understood. The four-myeloma cell lines NCI-H929, OPM-2, RPMI-8226, and U266 were cultured in vitro. Apoptosis was measured by flow cytometry after annexin V FITC and propidium iodide staining. Cell cycle distribution of cells was determined by DNA staining with propidium iodide. Intracellular levels of (phosphorylated) proteins were determined by western blot. /It was shown/ that bendamustine induces apoptosis with an IC50 of 35-65 mug/ml and with cleavage of caspase 3. Incubation with 10-30 mug/ml results in G2 cell cycle arrest in all four-cell lines. The primary DNA-damage signaling kinases ATM and Chk2, but not ATR and Chk1, are activated. The Chk2 substrate Cdc25A phosphatase is degraded and Cdc2 is inhibited by inhibitory phosphorylation of Tyr15 accompanied by increased cyclin B levels. Additionally, p53 activation occurs as phosphorylation of Ser15, the phosphorylation site for ATM. p53 promotes Cdc2 inhibition by upregulation of p21. Targeting of p38 MAPK by the selective inhibitor SB202190 significantly increases bendamustine induced apoptosis. Additionally, SB202190 completely abrogates G2 cell cycle arrest. Bendamustine induces ATM-Chk2-Cdc2-mediated G2 arrest and p53 mediated apoptosis. Inhibition of p38 MAPK augments apoptosis and abrogates G2 arrest and can be considered as a new therapeutic strategy in combination with bendamustine., Microarray-based gene expression profiling, real-time PCR, immunoblot, cell cycle, and functional DNA damage repair analyses were used to characterize response to bendamustine and compare it with chlorambucil and phosphoramide mustard. Bendamustine displays a distinct pattern of activity unrelated to other DNA-alkylating agents. Its mechanisms of action include activation of DNA-damage stress response and apoptosis, inhibition of mitotic checkpoints, and induction of mitotic catastrophe. In addition, unlike other alkylators, bendamustine activates a base excision DNA repair pathway rather than an alkyltransferase DNA repair mechanism. These results suggest that bendamustine possesses mechanistic features that differentiate it from other alkylating agents and may contribute to its distinct clinical efficacy profile., Bendamustine is a bifunctional mechlorethamine derivative containing a purine-like benzimidazole ring. Mechlorethamine and its derivatives form electrophilic alkyl groups. These groups form covalent bonds with electron-rich nucleophilic moieties, resulting in interstrand DNA crosslinks. The bifunctional covalent linkage can lead to cell death via several pathways. Bendamustine is active against both quiescent and dividing cells. The exact mechanism of action of bendamustine remains unknown. | |
| Record name | Bendamustine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06769 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Bendamustine | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7763 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
16506-27-7 | |
| Record name | Bendamustine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=16506-27-7 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Bendamustine [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0016506277 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Bendamustine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06769 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Bendamustine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2046888 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 16506-27-7 | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | BENDAMUSTINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/9266D9P3PQ | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Bendamustine | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7763 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Avertissement et informations sur les produits de recherche in vitro
Veuillez noter que tous les articles et informations sur les produits présentés sur BenchChem sont destinés uniquement à des fins informatives. Les produits disponibles à l'achat sur BenchChem sont spécifiquement conçus pour des études in vitro, qui sont réalisées en dehors des organismes vivants. Les études in vitro, dérivées du terme latin "in verre", impliquent des expériences réalisées dans des environnements de laboratoire contrôlés à l'aide de cellules ou de tissus. Il est important de noter que ces produits ne sont pas classés comme médicaments et n'ont pas reçu l'approbation de la FDA pour la prévention, le traitement ou la guérison de toute condition médicale, affection ou maladie. Nous devons souligner que toute forme d'introduction corporelle de ces produits chez les humains ou les animaux est strictement interdite par la loi. Il est essentiel de respecter ces directives pour assurer la conformité aux normes légales et éthiques en matière de recherche et d'expérimentation.















