Prednisone
Vue d'ensemble
Description
La prednisone est un glucocorticoïde synthétique principalement utilisé pour ses propriétés anti-inflammatoires et immunosuppressives . Il s'agit d'un promédicament qui est métabolisé dans le foie en sa forme active, la prednisolone . La this compound est largement utilisée dans le traitement de diverses affections telles que l'asthme, la bronchopneumopathie chronique obstructive, les maladies rhumatismales et certains types de cancer .
Méthodes De Préparation
Voies synthétiques et conditions réactionnelles : La préparation de la prednisone implique généralement plusieurs étapes à partir d'un oxyde de moisissure. Le procédé comprend une réaction d'oxydation de Platts, suivie d'une réaction d'addition de brome, d'un retrait de brome et d'une réaction d'addition d'iode en 21 bits . La dernière étape implique une réaction de remplacement en 21 bits pour obtenir de la this compound .
Méthodes de production industrielle : La production industrielle de this compound implique souvent l'utilisation d'acétate d'hydrocortisone comme matière première . Le processus comprend des étapes telles que l'oxydation, la réduction et l'estérification pour produire de la this compound à grande échelle . Les méthodes sont conçues pour minimiser les déchets et réduire l'impact environnemental en recyclant le chrome et l'acide acétique .
Analyse Des Réactions Chimiques
Types de réactions : La prednisone subit diverses réactions chimiques, notamment l'oxydation, la réduction et la substitution .
Réactifs et conditions courants :
Oxydation : Réaction d'oxydation de Platts utilisant de l'oxyde de moisissure.
Substitution : Les réactions d'addition et de retrait du brome sont courantes dans la synthèse de la this compound.
Produits majeurs : Le principal produit formé par ces réactions est la this compound elle-même, qui est ensuite transformée en sa forme active, la prednisolone .
4. Applications de la recherche scientifique
La this compound a un large éventail d'applications dans la recherche scientifique :
Chimie : Utilisé comme composé de référence dans l'étude des glucocorticoïdes et de leurs dérivés.
Biologie : Étudié pour ses effets sur les processus cellulaires et l'expression des gènes.
5. Mécanisme d'action
La this compound exerce ses effets en étant convertie en prednisolone dans le foie . La prednisolone se lie ensuite aux récepteurs des glucocorticoïdes, les activant et déclenchant des changements dans l'expression des gènes . Cela conduit à la suppression de la migration des leucocytes polymorphonucléaires et à l'inversion de l'augmentation de la perméabilité capillaire, réduisant ainsi l'inflammation . Il supprime également le système immunitaire en réduisant l'activité et le volume du système immunitaire .
Composés similaires :
- Dexaméthasone
- Méthotrexate
- Mycophénolate
- Mercaptopurine
- Azathioprine
- Leflunomide
Comparaison : La this compound est unique en son genre en ce qu'elle peut être convertie en une forme active, la prednisolone, qui a de puissants effets anti-inflammatoires et immunosuppresseurs . Comparée à la dexaméthasone, la this compound a une demi-vie plus courte et est moins puissante . Le méthotrexate et le mycophénolate sont principalement utilisés comme immunosuppresseurs, mais n'ont pas les mêmes propriétés anti-inflammatoires que la this compound . La mercaptopurine, l'azathioprine et le leflunomide sont également utilisés comme immunosuppresseurs, mais ils ont des mécanismes d'action différents et sont utilisés dans des contextes cliniques différents .
Applications De Recherche Scientifique
Indications for Use
Prednisone is indicated for numerous conditions, categorized as follows:
- Autoimmune Disorders : Such as lupus and rheumatoid arthritis.
- Respiratory Conditions : Including asthma and chronic obstructive pulmonary disease.
- Dermatological Conditions : Such as severe skin allergies and dermatitis.
- Gastrointestinal Disorders : Including inflammatory bowel disease.
- Endocrine Disorders : Such as adrenal insufficiency.
- Neoplastic Diseases : As part of chemotherapy regimens.
- Organ Transplantation : To prevent rejection .
Rheumatoid Arthritis
This compound is commonly used to manage symptoms of rheumatoid arthritis. A study indicated that patients receiving modified-release this compound experienced significant improvements in morning stiffness compared to placebo .
| Outcome Measure | This compound Group (n=100) | Placebo Group (n=100) | Risk Ratio (95% CI) |
|---|---|---|---|
| Morning stiffness reduction | 75% | 30% | 2.5 (1.8–3.4) |
Asthma Exacerbations
In patients with acute asthma exacerbations, this compound has been shown to reduce hospital admissions significantly. A recent study reported treatment failure rates of 42% in the this compound group compared to 34.5% in the placebo group, indicating a relative risk increase but also highlighting its role in managing severe cases .
| Outcome Measure | This compound Group (n=88) | Placebo Group (n=87) | Risk Ratio (95% CI) |
|---|---|---|---|
| Treatment failure at 8 weeks | 42% | 34.5% | 1.22 (0.87–1.69) |
Kidney Disorders
This compound is also utilized in treating nephrotic syndrome and IgA vasculitis nephritis, where it helps reduce proteinuria and delay renal function decline. Clinical guidelines recommend its use for patients with persistent proteinuria .
Case Studies
Case Study 1: Management of Lupus Nephritis
A cohort study involving patients with lupus nephritis showed that those treated with this compound had improved renal function and reduced proteinuria over six months compared to those receiving standard care without corticosteroids.
Case Study 2: Organ Transplantation
In a clinical trial involving kidney transplant recipients, the use of this compound as part of the immunosuppressive regimen significantly decreased the incidence of acute rejection episodes compared to controls not receiving corticosteroids.
Safety Profile
While this compound is effective, it is associated with potential side effects such as hyperglycemia, osteoporosis, and increased infection risk. Long-term use requires careful monitoring to mitigate these risks .
Mécanisme D'action
Prednisone exerts its effects by being converted to prednisolone in the liver . Prednisolone then binds to glucocorticoid receptors, activating them and triggering changes in gene expression . This leads to the suppression of the migration of polymorphonuclear leukocytes and reversal of increased capillary permeability, thereby reducing inflammation . It also suppresses the immune system by reducing the activity and volume of the immune system .
Comparaison Avec Des Composés Similaires
- Dexamethasone
- Methotrexate
- Mycophenolate
- Mercaptopurine
- Azathioprine
- Leflunomide
Comparison: Prednisone is unique in its ability to be converted to an active form, prednisolone, which has potent anti-inflammatory and immunosuppressive effects . Compared to dexamethasone, this compound has a shorter half-life and is less potent . Methotrexate and mycophenolate are primarily used as immunosuppressants but do not have the same anti-inflammatory properties as this compound . Mercaptopurine, azathioprine, and leflunomide are also used as immunosuppressants but have different mechanisms of action and are used in different clinical contexts .
Activité Biologique
Prednisone is a synthetic corticosteroid widely used for its anti-inflammatory and immunosuppressive properties. Its biological activity primarily stems from its conversion to prednisolone, the active metabolite, which exerts significant effects on various physiological processes. This article explores the biological activity of this compound, including its mechanism of action, clinical implications, and relevant research findings.
This compound is biologically inert and requires metabolic conversion in the liver to prednisolone, which binds to glucocorticoid receptors (GRs). This binding initiates a cascade of genomic and non-genomic effects that modulate inflammation and immune responses:
- Genomic Effects : The GR-ligand complex translocates to the nucleus, where it influences gene transcription. This process involves:
- Non-Genomic Effects : These include rapid signaling pathways that affect cellular functions such as ion transport and enzyme activity .
Pharmacokinetics
The pharmacokinetics of this compound involves its absorption, distribution, metabolism, and excretion (ADME):
- Absorption : this compound is well absorbed orally.
- Metabolism : Primarily occurs in the liver, where it converts to prednisolone. Factors such as liver disease can impair this conversion, affecting the drug's efficacy .
- Half-Life : The half-life of this compound is approximately 2-3 hours, with effects lasting longer due to the prolonged action of prednisolone .
Clinical Applications
This compound is utilized in various clinical settings, including:
- Autoimmune Disorders : Such as rheumatoid arthritis and lupus.
- Allergic Reactions : Effective in managing severe allergies and asthma exacerbations.
- Cancer Therapy : Used in combination with other agents to manage certain cancers.
Study on Liver Disease Impact
A study assessed the conversion efficiency of this compound in patients with liver disease compared to healthy controls. It found that patients with active liver disease had significantly higher plasma levels of prednisolone after this compound administration due to impaired conversion processes. This suggests that dosing adjustments may be necessary for this patient population .
Dystrophin Muscle Content Study
In a clinical trial involving patients with Duchenne muscular dystrophy (DMD), this compound was compared with other corticosteroids regarding its effect on muscle function and dystrophin production. Results indicated that while many patients showed minimal increases in dystrophin content, those treated with this compound exhibited varied responses based on individual metabolic differences .
Summary of Biological Activities
The following table summarizes key biological activities associated with this compound:
| Biological Activity | Description |
|---|---|
| Anti-inflammatory | Reduces inflammation by inhibiting pro-inflammatory mediators. |
| Immunosuppression | Decreases immune response, beneficial in autoimmune diseases. |
| Gene Expression Modulation | Alters transcription of genes involved in inflammation and immune response. |
| Metabolic Effects | Influences glucose metabolism and can lead to side effects such as hyperglycemia. |
Q & A
Basic Research Questions
Q. How should researchers formulate a hypothesis-driven research question for studying Prednisone’s anti-inflammatory mechanisms?
- Methodological Answer : Begin by identifying gaps in existing literature (e.g., unresolved pathways like NF-κB modulation). Use the FINER criteria (Feasible, Interesting, Novel, Ethical, Relevant) to evaluate the question’s scope. For example: “Does this compound inhibit interleukin-6 (IL-6) synthesis in macrophages via glucocorticoid receptor dimerization, and how does this vary across immune cell subtypes?” Align the question with preclinical models (e.g., murine macrophage assays) to ensure testability .
Q. What experimental design principles are critical for establishing causality in this compound efficacy studies?
- Methodological Answer : Implement randomized controlled trials (RCTs) with double-blinding to minimize bias. Define inclusion/exclusion criteria rigorously (e.g., excluding patients with recent steroid use). Use stratified randomization to account for covariates like age or comorbidities. For preclinical studies, adhere to NIH guidelines for animal models, including sample size calculations and ethical oversight .
Q. How can researchers conduct a systematic literature review to contextualize this compound’s pharmacokinetics?
- Methodological Answer : Use databases like PubMed and Embase with Boolean terms (e.g., “this compound AND pharmacokinetics NOT asthma”). Apply PRISMA guidelines for screening and data extraction. Critically evaluate studies for biases (e.g., industry-funded trials) and prioritize peer-reviewed, high-impact journals. Synthesize findings into a meta-analysis if heterogeneity permits .
Advanced Research Questions
Q. How can researchers enhance reproducibility in this compound metabolite quantification studies?
- Methodological Answer : Standardize LC-MS/MS protocols across labs (e.g., identical column temperatures and ionization sources). Share raw data via repositories like MetaboLights. Perform inter-laboratory validation with blinded samples .
Key Takeaways :
Propriétés
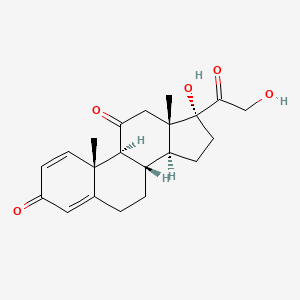
IUPAC Name |
(8S,9S,10R,13S,14S,17R)-17-hydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-6,7,8,9,12,14,15,16-octahydrocyclopenta[a]phenanthrene-3,11-dione | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C21H26O5/c1-19-7-5-13(23)9-12(19)3-4-14-15-6-8-21(26,17(25)11-22)20(15,2)10-16(24)18(14)19/h5,7,9,14-15,18,22,26H,3-4,6,8,10-11H2,1-2H3/t14-,15-,18+,19-,20-,21-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XOFYZVNMUHMLCC-ZPOLXVRWSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC12CC(=O)C3C(C1CCC2(C(=O)CO)O)CCC4=CC(=O)C=CC34C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@]12CC(=O)[C@H]3[C@H]([C@@H]1CC[C@@]2(C(=O)CO)O)CCC4=CC(=O)C=C[C@]34C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C21H26O5 | |
| Record name | PREDNISONE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20949 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID4021185 | |
| Record name | Prednisone | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4021185 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
358.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Prednisone is an odorless white crystalline powder. (NTP, 1992), Solid | |
| Record name | PREDNISONE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20949 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Prednisone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014773 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Very slightly soluble (NTP, 1992), Very slightly soluble, Very slightly soluble in water; 1 g soluble in 150 mL alcohol, in 200 mL chloroform; slightly soluble in methanol, 1.11e-01 g/L | |
| Record name | PREDNISONE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20949 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Prednisone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00635 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PREDNISONE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3168 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Prednisone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014773 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Prednisone is first metabolized in the liver to its active form, prednisolone, a glucocorticoid agonist corticosteroid. The short term effects of corticosteroids are decreased vasodilation and permeability of capillaries, as well as decreased leukocyte migration to sites of inflammation. Corticosteroids binding to the glucocorticoid receptor mediates changes in gene expression that lead to multiple downstream effects over hours to days. Glucocorticoids inhibit neutrophil apoptosis and demargination; they inhibit phospholipase A2, which decreases the formation of arachidonic acid derivatives; they inhibit NF-Kappa B and other inflammatory transcription factors; they promote anti-inflammatory genes like interleukin-10. Lower doses of corticosteroids provide an anti-inflammatory effect, while higher doses are immunosuppressive. High doses of glucocorticoids for an extended period bind to the mineralocorticoid receptor, raising sodium levels and decreasing potassium levels., In physiologic doses, corticosteroids are administered to replace deficient endogenous hormones. In larger (pharmacologic) doses, glucocorticoids decrease inflammation by stabilizing leukocyte lysosomal membranes, preventing release of destructive acid hydrolases from leukocytes; inhibiting macrophage accumulation in inflamed areas; reducing leukocyte adhesion to capillary endothelium; reducing capillary wall permeability and edema formation; decreasing complement components; antagonizing histamine activity and release of kinin from substrates; reducing fibroblast proliferation, collagen deposition, and subsequent scar tissue formation; and possibly by other mechanisms as yet unknown. The drugs suppress the immune response by reducing activity and volume of the lymphatic system, producing lymphocytopenia, decreasing immunoglobulin and complement concentrations, decreasing passage of immune complexes through basement membranes, and possibly by depressing reactivity of tissue to antigen-antibody interactions. Glucocorticoids stimulate erythroid cells of bone marrow, prolong survival time of erythrocytes and platelets, and produce neutrophilia and eosinopenia. Glucocorticoids promote gluconeogenesis, redistribution of fat from peripheral to central areas of the body, and protein catabolism, which results in negative nitrogen balance. They reduce intestinal absorption and increase renal excretion of calcium. /Corticosteroids/, Glucocorticoids are capable of suppressing the inflammatory process through numerous pathways. They interact with specific intracellular receptor proteins in target tissues to alter the expression of corticosteroid-responsive genes. Glucocorticoid-specific receptors in the cell cytoplasm bind with steroid ligands to form hormone-receptor complexes that eventually translocate to the cell nucleus. There these complexes bind to specific DNA sequences and alter their expression. The complexes may induce the transcription of mRNA leading to synthesis of new proteins. Such proteins include lipocortin, a protein known to inhibit PLA2a and thereby block the synthesis of prostaglandins, leukotrienes, and PAF. Glucocorticoids also inhibit the production of other mediators including AA metabolites such as COX, cytokines, the interleukins, adhesion molecules, and enzymes such as collagenase. /Glucocorticoids/ | |
| Record name | Prednisone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00635 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PREDNISONE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3168 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals, White to practically white, crystalline powder | |
CAS No. |
53-03-2 | |
| Record name | PREDNISONE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20949 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Prednisone | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=53-03-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Prednisone [USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000053032 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Prednisone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00635 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | prednisone | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=10023 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Pregna-1,4-diene-3,11,20-trione, 17,21-dihydroxy- | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | Prednisone | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4021185 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Prednisone | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.147 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PREDNISONE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/VB0R961HZT | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | PREDNISONE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3168 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Prednisone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014773 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
451 to 455 °F (DEC) (NTP, 1992), 234 °C (decomposes), 233 - 235 °C | |
| Record name | PREDNISONE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20949 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Prednisone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00635 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PREDNISONE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3168 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Prednisone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014773 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Avertissement et informations sur les produits de recherche in vitro
Veuillez noter que tous les articles et informations sur les produits présentés sur BenchChem sont destinés uniquement à des fins informatives. Les produits disponibles à l'achat sur BenchChem sont spécifiquement conçus pour des études in vitro, qui sont réalisées en dehors des organismes vivants. Les études in vitro, dérivées du terme latin "in verre", impliquent des expériences réalisées dans des environnements de laboratoire contrôlés à l'aide de cellules ou de tissus. Il est important de noter que ces produits ne sont pas classés comme médicaments et n'ont pas reçu l'approbation de la FDA pour la prévention, le traitement ou la guérison de toute condition médicale, affection ou maladie. Nous devons souligner que toute forme d'introduction corporelle de ces produits chez les humains ou les animaux est strictement interdite par la loi. Il est essentiel de respecter ces directives pour assurer la conformité aux normes légales et éthiques en matière de recherche et d'expérimentation.
















