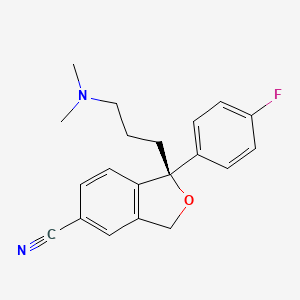
エシタロプラム
概要
説明
エシタロプラムは、主に抗うつ薬として使用される選択的セロトニン再取り込み阻害薬です。これは、シタロプラムのS-エナンチオマーであり、分子の2つの鏡像異性体のうちの1つであることを意味します。 エシタロプラムは、セロトニントランスポーターを阻害する高い選択性と効力を有することが知られており、主要なうつ病および汎化性不安障害の治療に効果的です .
作用機序
エシタロプラムは、神経伝達物質であるセロトニンのシナプス前ニューロンへの再取り込みを阻害することで作用します。この阻害により、シナプス間隙で利用可能なセロトニンのレベルが増加し、セロトニン作動性神経伝達の強化につながります。 主要な分子標的は、セロトニントランスポーター(SERT)であり、エシタロプラムは、セロトニンの再取り込みを阻害する、オルトステリック部位に結合します .
科学的研究の応用
Escitalopram has a wide range of scientific research applications:
Chemistry: It is used as a model compound in the study of chiral separation techniques and enantiomeric purity.
Biology: Research on escitalopram includes its effects on neurotransmitter systems and its role in neuroplasticity.
Medicine: Escitalopram is extensively studied for its efficacy in treating depression, anxiety, and other psychiatric disorders. .
生化学分析
Biochemical Properties
The primary molecular target for Escitalopram is the serotonin transporter (SERT), which is responsible for serotonin (also called 5-hydroxytryptamine [5-HT]) reuptake at the terminals and cell bodies of serotonergic neurons . This leads to antidepressant effects by increasing extracellular 5-HT levels which enhance 5-HT neurotransmission .
Cellular Effects
Escitalopram exerts its effects by restoring serotonergic function in the treatment of depression and anxiety . It is approximately 150 times more potent than citalopram’s R-enantiomer and is responsible for the vast majority of citalopram’s clinical activity .
Molecular Mechanism
Escitalopram exerts its effects at the molecular level through binding interactions with the serotonin transporter (SERT). It has been shown that through allosteric binding, Escitalopram decreases its own dissociation rate from the orthosteric site on the SERT . This may be the mechanism responsible for its observed superior efficacy and faster onset compared to other SSRIs .
Metabolic Pathways
Escitalopram is involved in the serotonin reuptake metabolic pathway. It interacts with the serotonin transporter (SERT), affecting the reuptake of serotonin at the terminals and cell bodies of serotonergic neurons .
準備方法
合成経路と反応条件
エシタロプラムの合成は、前駆体であるシタロプラムから始まり、いくつかのステップを含みます。一般的な方法の1つは、キラルクロマトグラフィーを使用して、シタロプラムのラセミ混合物をそのエナンチオマーに分割することです。 その後、S-エナンチオマーを単離および精製します .
別の方法は、ジオール化合物を環化し、続いて得られた化合物を分割することです。 ジオール化合物は、多くの場合、シュウ酸塩として調製され、分割プロセスを促進します .
工業生産方法
エシタロプラムの工業生産は、通常、同じ合成経路に従いますが、より大規模に行われます。 キラルパックADまたはキラルセルODなどのキラル固定相の使用は、エナンチオマーのクロマトグラフィー分離において一般的です . このプロセスは、最終製品の高い収率と純度を確保するために最適化されています。
化学反応の分析
反応の種類
エシタロプラムは、次のようなさまざまな種類の化学反応を起こします。
酸化: エシタロプラムは、酸化されてさまざまな代謝産物を生成することができます。
還元: 還元反応はあまり一般的ではありませんが、特定の条件下で発生する可能性があります。
一般的な試薬と条件
酸化: 一般的な酸化剤には、過マンガン酸カリウムと過酸化水素が含まれます。
還元: 水素化リチウムアルミニウムなどの還元剤を使用できます。
主要な製品
これらの反応から生成される主要な製品には、エシタロプラムのさまざまな代謝産物と類似体があり、これらは多くの場合、薬理学的特性について研究されています .
科学研究への応用
エシタロプラムは、幅広い科学研究への応用があります。
化学: キラル分離技術とエナンチオマー純度の研究におけるモデル化合物として使用されます。
生物学: エシタロプラムの研究には、神経伝達物質系への影響と神経可塑性における役割が含まれます。
医学: エシタロプラムは、うつ病、不安、およびその他の精神障害の治療における有効性について広く研究されています。 .
産業: 製薬業界では、エシタロプラムを、新しい選択的セロトニン再取り込み阻害薬を開発するためのベンチマークとして使用しています.
類似化合物との比較
類似化合物
シタロプラム: エシタロプラムが由来するラセミ混合物。
フルオキセチン: 化学構造の異なる別の選択的セロトニン再取り込み阻害薬。
独自性
エシタロプラムは、セロトニントランスポーターに対する高い選択性と効力によって特徴付けられます。 いくつかの臨床試験では、ラセミ混合物であるシタロプラムや他の選択的セロトニン再取り込み阻害薬よりも効果的で、忍容性が高いことが示されています . セロトニントランスポーターの異種調節も、他の選択的セロトニン再取り込み阻害薬と比較して、優れた効力とより速い作用発現に貢献しています .
特性
IUPAC Name |
(1S)-1-[3-(dimethylamino)propyl]-1-(4-fluorophenyl)-3H-2-benzofuran-5-carbonitrile | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C20H21FN2O/c1-23(2)11-3-10-20(17-5-7-18(21)8-6-17)19-9-4-15(13-22)12-16(19)14-24-20/h4-9,12H,3,10-11,14H2,1-2H3/t20-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
WSEQXVZVJXJVFP-FQEVSTJZSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CN(C)CCCC1(C2=C(CO1)C=C(C=C2)C#N)C3=CC=C(C=C3)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CN(C)CCC[C@@]1(C2=C(CO1)C=C(C=C2)C#N)C3=CC=C(C=C3)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C20H21FN2O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID8048440 | |
| Record name | Escitalopram | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8048440 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
324.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Escitalopram | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005028 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Sparingly soluble | |
| Record name | Escitalopram | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01175 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Escitalopram, like other selective serotonin re-uptake inhibitors, enhances serotonergic activity by binding to the orthosteric (i.e. primary) binding site on the serotonin transporter (SERT), the same site to which endogenous 5-HT binds, and thus prevents the re-uptake of serotonin into the presynaptic neuron. Escitalopram, along with [paroxetine], is also considered an allosteric serotonin re-uptake inhibitor - it binds to a secondary allosteric site on the SERT molecule to more strongly inhibit 5-HT re-uptake. Its combination of orthosteric and allosteric activity on SERT allows for greater extracellular 5-HT levels, a faster onset of action, and greater efficacy as compared to other SSRIs. The sustained elevation of synaptic 5-HT eventually causes desensitization of 5-HT1A auto-receptors, which normally shut down endogenous 5-HT release in the presence of excess 5-HT - this desensitization may be necessary for the full clinical effect of SSRIs and may be responsible for their typically prolonged onset of action. Escitalopram has shown little-to-no binding affinity at a number of other receptors, such as histamine and muscarinic receptors, and minor activity at these off-targets may explain some of its adverse effects., The mechanism of antidepressant action of escitalopram, the S-enantiomer of racemic citalopram, is presumed to be linked to potentiation of serotonergic activity in the central nervous system (CNS) resulting from its inhibition of CNS neuronal reuptake of serotonin (5-HT). | |
| Record name | Escitalopram | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01175 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Escitalopram | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8410 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
128196-01-0 | |
| Record name | (+)-Citalopram | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=128196-01-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Escitalopram [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0128196010 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Escitalopram | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01175 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Escitalopram | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8048440 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (1S)-1-[3-(dimethylamino)propyl]-1-(4-fluorophenyl)-1,3-dihydro-2-benzofuran-5-carbonitrile | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.244.188 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | ESCITALOPRAM | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/4O4S742ANY | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Escitalopram | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8410 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Escitalopram | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005028 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
147-152C | |
| Record name | Escitalopram | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01175 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details








Synthesis routes and methods III
Procedure details







Synthesis routes and methods IV
Procedure details










Synthesis routes and methods V
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Q1: What is the primary mechanism of action of escitalopram?
A: Escitalopram is a selective serotonin reuptake inhibitor (SSRI). It exerts its therapeutic effect by blocking the serotonin transporter (SERT) in the central nervous system (CNS). [, , , ] This inhibition increases the extracellular concentration of serotonin in the synaptic cleft, enhancing serotonergic neurotransmission.
Q2: What is the molecular formula and weight of escitalopram?
A: Escitalopram's molecular formula is C20H21FN2O, and its molecular weight is 324.4 g/mol. []
Q3: Is there any spectroscopic data available for escitalopram?
A: While specific spectroscopic data wasn't provided in these papers, analytical techniques like HPLC-MS/MS are commonly employed for its characterization and quantification. [, ]
Q4: Is there information available regarding the material compatibility and stability of escitalopram under various conditions?
A4: The provided research papers primarily focus on the pharmacological aspects of escitalopram rather than its material properties. Therefore, information on material compatibility and stability is limited within these sources.
Q5: Does escitalopram possess any catalytic properties or find applications in catalysis?
A5: Escitalopram is primarily recognized for its pharmacological activity as an antidepressant. The provided papers do not suggest any catalytic properties or applications for this compound.
Q6: Have computational chemistry methods been employed to study escitalopram?
A: While not extensively discussed in these papers, computational chemistry techniques, including QSAR modeling, can be valuable for exploring the structure-activity relationships of escitalopram and designing potential analogs. []
Q7: What is the significance of escitalopram being the S-enantiomer of citalopram?
A: Escitalopram is the active S-enantiomer of the racemic drug citalopram. Research has shown that the R-enantiomer of citalopram may counteract the therapeutic effects of the S-enantiomer. [] Therefore, escitalopram, as a single enantiomer, demonstrates enhanced efficacy and potentially fewer side effects compared to the racemate. [, , ]
Q8: What is the primary route of metabolism for escitalopram?
A: Escitalopram is primarily metabolized in the liver by the cytochrome P450 (CYP) enzyme system, specifically CYP2C19. [, ] Polymorphisms in the CYP2C19 gene can significantly influence escitalopram plasma concentrations and potentially affect individual treatment response. [, , ]
Q9: How does the co-administration of fluvoxamine, a CYP2C19 inhibitor, impact escitalopram levels?
A: Co-administration of fluvoxamine significantly increases plasma concentrations of escitalopram by inhibiting its metabolism through CYP2C19. [] This interaction highlights the importance of considering potential drug interactions when prescribing escitalopram.
Q10: Are there any known drug transporter interactions associated with escitalopram?
A: While specific drug transporter interactions are not extensively covered in these papers, it's important to be aware that drug transporters play a crucial role in drug absorption, distribution, and elimination. [] Further research may provide more insights into potential transporter interactions with escitalopram.
Q11: Has the efficacy of escitalopram been demonstrated in preclinical models of depression?
A: Yes, studies using the chronic mild stress (CMS) model in rats have shown that escitalopram effectively alleviates depressive-like behaviors. [] This model is commonly used to assess the efficacy of potential antidepressant compounds.
Q12: What do clinical trials indicate about the efficacy of escitalopram in treating major depressive disorder (MDD)?
A: Numerous clinical trials have consistently demonstrated the efficacy of escitalopram in treating MDD. [, , , ] Meta-analyses comparing escitalopram to other SSRIs and serotonin-norepinephrine reuptake inhibitors (SNRIs) have confirmed its superior efficacy in improving depressive symptoms and achieving remission. [, ]
Q13: Does escitalopram demonstrate efficacy in treating conditions other than MDD?
A: Besides MDD, clinical trials have supported the efficacy of escitalopram in treating generalized anxiety disorder (GAD). [] Additionally, some research suggests its potential benefit in managing other conditions, such as obsessive-compulsive disorder (OCD). []
Q14: Is there evidence of resistance developing to escitalopram treatment?
A: While the development of resistance to escitalopram is not extensively discussed in these papers, it's a phenomenon observed with many antidepressant medications. Further research is needed to understand the mechanisms underlying potential resistance to escitalopram. []
Q15: What are the common adverse effects associated with escitalopram?
A15: As this Q&A focuses on the scientific aspects and not clinical guidelines, we'll refrain from listing specific side effects. Please refer to the drug information leaflet or consult a healthcare professional for comprehensive information on escitalopram's adverse effects.
Q16: Are there any long-term safety concerns associated with escitalopram use?
A16: We recommend referring to the drug information leaflet or consulting a healthcare professional for information on the long-term safety of escitalopram.
Q17: Are there any specific drug delivery systems or targeting strategies being investigated for escitalopram?
A17: The provided research primarily focuses on the pharmacological effects of escitalopram, and these papers do not delve into specific drug delivery systems or targeting strategies being explored for this compound.
Q18: Are there any known biomarkers associated with escitalopram efficacy or adverse effects?
A: While specific biomarkers for escitalopram response are not extensively discussed in these papers, research on identifying potential biomarkers for antidepressant response is ongoing. [] Such biomarkers could potentially guide treatment selection and personalize therapy.
Q19: What analytical techniques are commonly used to quantify escitalopram in biological samples?
A: High-performance liquid chromatography coupled with tandem mass spectrometry (HPLC-MS/MS) is a widely used method for the sensitive and specific quantification of escitalopram in plasma samples. [, , ] This technique enables accurate measurement of drug concentrations for pharmacokinetic studies and therapeutic drug monitoring.
Q20: Is there information available regarding the environmental impact and degradation of escitalopram?
A20: The provided research focuses on the pharmacological and clinical aspects of escitalopram. Therefore, information on its environmental impact and degradation pathways is limited within these sources.
Q21: Are there established protocols for validating analytical methods used to measure escitalopram concentrations?
A: Yes, rigorous analytical method validation is crucial for ensuring the accuracy, precision, and reliability of drug concentration measurements. [] Regulatory agencies, such as the FDA and EMA, provide guidelines for validating analytical methods used in pharmaceutical analysis.
Q22: What quality control measures are implemented during the manufacturing and distribution of escitalopram to ensure its quality, safety, and efficacy?
A: Stringent quality control and assurance measures are implemented throughout the entire lifecycle of escitalopram, from development to manufacturing and distribution. [] These measures ensure that the drug product consistently meets predefined quality standards and regulatory requirements.
Q23: Is there evidence indicating that escitalopram induces significant immunogenicity or immunological responses?
A23: The provided research papers do not focus on the immunogenic potential of escitalopram. Therefore, specific information on its immunogenicity and potential immunological responses is limited within these sources.
Q24: Does escitalopram induce or inhibit drug-metabolizing enzymes, potentially leading to drug interactions?
A: While escitalopram is primarily metabolized by CYP2C19, its potential to induce or inhibit other drug-metabolizing enzymes is not extensively discussed in these papers. [, ] It's generally advisable to consult drug interaction databases and resources for a comprehensive assessment of potential interactions with escitalopram.
Q25: Is there information on the biocompatibility and biodegradability of escitalopram?
A: The research papers provided primarily focus on the pharmacological and clinical aspects of escitalopram. As a result, specific details regarding its biocompatibility and biodegradability are not extensively covered in these sources. []
Q26: What are some alternative antidepressants available, and how does escitalopram compare in terms of efficacy, safety, and cost?
A: Several other SSRIs, SNRIs, and other classes of antidepressants are available as alternative treatment options. [, , ] The choice of antidepressant is typically individualized based on factors such as patient characteristics, medical history, potential drug interactions, and cost.
Q27: Are there specific guidelines or recommendations for the recycling and waste management of escitalopram?
A: Information regarding the recycling and waste management of escitalopram is not directly addressed in the provided research papers. Proper disposal of unused medications, often through take-back programs or following local regulations, is essential to minimize environmental risks. []
Q28: What research infrastructure and resources are essential for advancing our understanding of escitalopram and developing new antidepressants?
A: A robust research infrastructure, including well-equipped laboratories, access to clinical trial networks, and collaborations between academia and industry, is crucial for advancing antidepressant research. [] Additionally, funding agencies play a vital role in supporting research efforts.
Q29: What are some key historical milestones in the development and understanding of escitalopram and SSRIs in general?
A: The discovery and development of SSRIs, including escitalopram, represent significant milestones in the treatment of depression and anxiety disorders. [] These medications offered a more favorable safety and tolerability profile compared to older classes of antidepressants.
Q30: How has cross-disciplinary collaboration contributed to research on escitalopram and the development of new antidepressants?
A: Cross-disciplinary collaboration, involving pharmacologists, chemists, psychiatrists, neuroscientists, and other experts, has been essential for advancing antidepressant research. [] Such collaborations facilitate a more comprehensive understanding of depression, its underlying mechanisms, and the development of innovative treatment strategies.
試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。