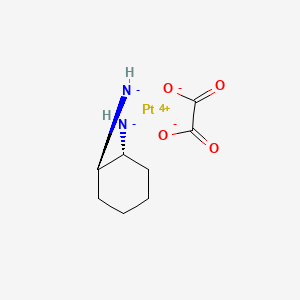
Oxaliplatin
描述
奥沙利铂是一种铂类化疗药物,主要用于治疗结直肠癌。它属于含铂抗肿瘤药物类,因其具有与 DNA 形成交联的能力而闻名,从而抑制 DNA 复制和转录。这种化合物已证明在治疗各种癌症类型方面有效,包括结直肠癌、卵巢癌和胰腺癌 .
准备方法
合成路线和反应条件: 奥沙利铂是通过多步合成过程合成的,该过程涉及铂化合物与特定配体的反应。主要的合成路线涉及顺式-二碘(反式-1,2-二氨基环己烷)铂(II)与草酸的反应。反应条件通常包括控制温度和 pH 值,以确保形成所需的产物 .
工业生产方法: 在工业环境中,奥沙利铂是在大型反应器中生产的,反应条件经过精心控制,以实现高产量和高纯度。该过程涉及使用高纯度起始材料和溶剂,最终产品经过严格的质量控制措施,以确保其有效性和安全性 .
化学反应分析
反应类型: 奥沙利铂会经历几种类型的化学反应,包括取代、氧化和还原。最显著的反应是它与 DNA 形成共价键的能力,导致形成链内和链间交联 .
常用试剂和条件: 涉及奥沙利铂的反应通常需要特定试剂,例如氯离子、水和各种有机溶剂。条件通常包括控制温度和 pH 值,以促进所需的化学转化 .
主要形成的产物: 奥沙利铂反应形成的主要产物是 DNA 加合物,它们是其细胞毒作用的原因。这些加合物阻止 DNA 复制和转录,导致细胞死亡 .
科学研究应用
Colorectal Cancer
Indications:
- Oxaliplatin is primarily indicated for:
Efficacy:
- A phase II trial reported a response rate of 24.3% in patients with previously untreated metastatic colorectal adenocarcinoma, with a median progression-free survival of 126 days .
- Combination therapies have shown improved response rates ranging from 34% to 67% , significantly enhancing survival outcomes compared to treatments without this compound .
Off-Label Uses
This compound has also been explored for off-label indications, including:
- Refractory or relapsed solid tumors in pediatric patients.
- Refractory neuroendocrine tumors and various hematologic malignancies .
- Advanced ovarian cancer and refractory testicular cancer , often in combination with other agents like gemcitabine and paclitaxel .
Kounis Syndrome Induced by this compound
A notable case study documented a 52-year-old woman who experienced Kounis syndrome—a hypersensitivity reaction affecting cardiac tissue—after administration of this compound. This case highlighted the need for awareness regarding hypersensitivity reactions to platinum-based drugs .
Extravasation Events
Another case reported significant tissue damage following the extravasation of this compound during an infusion. Despite the severity of the incident, the patient showed remarkable recovery without surgical intervention after appropriate management .
Side Effects and Toxicity Profile
While this compound is effective, it is associated with several adverse effects:
- Peripheral sensory neuropathy : One of the most common side effects, reported in up to 40% of patients.
- Other toxicities include gastrointestinal disturbances (nausea, vomiting), hematologic issues (neutropenia), and acute dysesthesias .
Comparative Efficacy
The following table summarizes key findings from various clinical trials regarding this compound's efficacy:
| Study Type | Population | Response Rate | Median Survival (Months) | Main Toxicity |
|---|---|---|---|---|
| Phase II Trial | Untreated Metastatic CRC | 24.3% | 216+ days | Peripheral neuropathy |
| Combination Therapy | Previously treated CRC | 34%-67% | 15-19 months | Gastrointestinal |
| Off-label Use | Refractory solid tumors | Variable | N/A | Varies by regimen |
作用机制
奥沙利铂通过与 DNA 形成共价键发挥作用,导致 DNA 交联的形成。这些交联阻止 DNA 复制和转录,最终导致细胞死亡。奥沙利铂的主要分子靶点是 DNA 中的嘌呤碱基,在那里它形成链内和链间交联 .
参与奥沙利铂作用机制的途径包括激活 DNA 损伤应答途径,导致细胞周期停滞和凋亡。该化合物还干扰癌细胞的修复机制,使其更容易受到其细胞毒作用的影响 .
相似化合物的比较
奥沙利铂经常与其他铂类化疗药物(如顺铂和卡铂)进行比较。虽然这三种化合物都具有相似的作用机制,但奥沙利铂具有独特的特性,使其与其他化合物有所区别。
类似化合物:
奥沙利铂的独特性: 奥沙利铂的独特性在于它能够形成更稳定的 DNA 加合物,导致癌细胞的细胞毒性更强。它还具有不同的副作用特征,肾毒性和耳毒性发生率较低,但周围神经病变发生率较高 .
总之,奥沙利铂是一种重要的化疗药物,在科学研究和临床实践中具有广泛的应用。其独特的特性和作用机制使其成为抗癌斗争中宝贵的工具。
生物活性
Oxaliplatin is a platinum-based chemotherapeutic agent primarily used in the treatment of colorectal cancer. Its unique chemical structure and mechanism of action differentiate it from other platinum compounds like cisplatin and carboplatin. This article provides a comprehensive overview of the biological activity of this compound, including its mechanisms, efficacy in clinical studies, and potential side effects.
This compound is characterized by the presence of a diaminocyclohexane (DACH) moiety, which enhances its antitumor activity compared to traditional platinum drugs. The mechanism of action involves the formation of DNA cross-links, which inhibit DNA replication and transcription, leading to cell death. Specifically, this compound forms intrastrand and interstrand cross-links primarily at the N7 position of guanine bases in DNA. This action complicates DNA repair mechanisms, making cancer cells more susceptible to its effects .
Key Mechanisms:
- DNA Cross-Linking: Formation of Pt-DNA adducts that prevent DNA replication.
- Immunogenic Cell Death: this compound can induce immunogenic signals in colon cancer cells, promoting an immune response against tumors .
- Cell Cycle Effects: The cytotoxicity is cell-cycle nonspecific, affecting various phases of the cell cycle .
Efficacy in Clinical Studies
Numerous clinical trials have evaluated the efficacy of this compound, particularly in combination with other agents such as fluorouracil (5-FU) and leucovorin (LV). The following table summarizes key findings from significant studies:
Case Studies
- Phase II Trial on Metastatic Colorectal Cancer :
- MOSAIC Trial :
Pharmacokinetics
The pharmacokinetics of this compound involve rapid distribution and non-enzymatic conversion into active metabolites post-administration. After intravenous infusion, the drug is primarily bound to plasma proteins, with a volume of distribution indicating extensive tissue uptake . The elimination half-life is approximately 30 hours, with renal excretion being a significant route for its metabolites.
Safety Profile and Side Effects
While this compound is effective against various cancers, it is associated with several side effects:
常见问题
Basic Research Questions
Q. What are the pharmacodynamic distinctions between oxaliplatin and cisplatin, and how do these differences influence experimental design?
this compound differs from cisplatin in its DNA adduct formation and resistance mechanisms. Unlike cisplatin, this compound forms bulky platinum-DNA adducts that evade mismatch repair (MMR) detection, contributing to activity in MMR-deficient tumors . Experimental comparisons should include cytotoxicity assays across cell lines with varying MMR status (e.g., HCT116 MMR-proficient vs. MMR-deficient models) and use COMPARE analysis to evaluate differential gene expression profiles .
Q. How should researchers optimize combination therapies involving this compound in preclinical models?
Synergy studies require factorial experimental designs to test this compound with agents like 5-fluorouracil (5-FU) or bevacizumab. For example, in the NSABP C-07 trial, this compound combined with fluorouracil/leucovorin improved 3-year disease-free survival (78.2% vs. 72.9%; HR: 0.77, P=0.002) . Preclinical models should replicate clinical dosing schedules (e.g., FOLFOX regimens) and include endpoints like tumor growth delay and apoptosis markers (e.g., caspase-3 activation) .
Q. What experimental models best recapitulate this compound-induced neuropathy for mechanistic studies?
Rodent models using cumulative this compound doses (4–6 mg/kg/week) mimic chronic neuropathy. Assess mechanical allodynia via von Frey filaments and correlate with histopathological changes in dorsal root ganglia. In vitro models using sensory neurons can evaluate mitochondrial dysfunction and oxidative stress via Seahorse assays .
Advanced Research Questions
Q. How do molecular subtypes of colorectal cancer (CRC) predict this compound benefit?
CRC subtypes (e.g., CMS4/stem-like) show differential responses. In the NSABP C-07 trial, enterocyte-subtype stage III patients had significant this compound benefit (HR: 0.22, P=0.001 in discovery cohort), while stem-like subtypes showed no benefit (HR: 0.99, P=0.96) . Researchers should integrate transcriptomic profiling (e.g., RNA-seq) with clinical outcomes and validate findings using locked algorithms in independent cohorts .
Q. What biomarkers predict this compound resistance, and how can they be functionally validated?
BRAF mutations (e.g., V600E) are prognostic for poor survival (HR: 2.31 for post-recurrence survival, P<0.0001) but not predictive of this compound resistance . Validate candidates via CRISPR/Cas9 knock-in models and assess platinum-DNA adduct repair efficiency using comet assays. Correlate with ATP7A/B transporter expression in patient-derived organoids .
Q. How does this compound reintroduction impact survival in metastatic CRC, and what statistical methods adjust for confounding factors?
In the OPTIMOX1 trial, this compound reintroduction improved OS (HR: 0.56, P=0.009). Use Cox proportional hazards models with time-dependent covariates (e.g., progression events, second-line therapies) and shared frailty models to account for center-specific reintroduction rates .
Q. Methodological Guidance
Q. What statistical approaches are critical for analyzing this compound clinical trial data with heterogeneous subtypes?
- Stratified log-rank tests : Compare survival between treatment arms within molecular subgroups .
- Multivariate Cox models : Adjust for baseline risk factors (e.g., WBC, alkaline phosphatase) .
- Interaction tests : Evaluate treatment-by-subtype effects (e.g., enterocyte vs. stem-like) .
| Subtype | This compound Benefit (HR) | 95% CI | P-Value |
|---|---|---|---|
| Enterocyte | 0.22 | 0.09–0.56 | 0.001 |
| Stem-like | 0.99 | 0.73–1.34 | 0.96 |
| Data from NSABP C-07 trial |
Q. How can network pharmacology models elucidate this compound’s off-target effects?
Integrate transcriptomic data (e.g., microarray profiling of pancreatic cancer cells) with protein interaction networks. Use systems biology tools like Cytoscape to identify hubs (e.g., autophagy-related proteins LC3B/Beclin-1) and validate via siRNA knockdown and functional assays (e.g., autophagosome quantification) .
Q. Contradictions and Validation
- vs. 11 : While molecular subtyping predicts this compound benefit in specific cohorts (e.g., enterocyte), BRAF mutations are prognostic but not predictive. Researchers must validate subtype-specific findings in independent trials and control for confounding variables like microsatellite instability .
- : this compound reintroduction improves survival, but trial designs should predefine reintroduction criteria to avoid selection bias.
属性
Key on ui mechanism of action |
Oxaliplatin undergoes nonenzymatic conversion in physiologic solutions to active derivatives via displacement of the labile oxalate ligand. Several transient reactive species are formed, including monoaquo and diaquo DACH platinum, which covalently bind with macromolecules. Both inter and intrastrand Pt-DNA crosslinks are formed. Crosslinks are formed between the N7 positions of two adjacent guanines (GG), adjacent adenine-guanines (AG), and guanines separated by an intervening nucleotide (GNG). These crosslinks inhibit DNA replication and transcription. Cytotoxicity is cell-cycle nonspecific. |
|---|---|
CAS 编号 |
61825-94-3 |
分子式 |
C8H14N2O4Pt |
分子量 |
397.29 g/mol |
IUPAC 名称 |
[(1R,2R)-2-azanidylcyclohexyl]azanide;oxalic acid;platinum(2+) |
InChI |
InChI=1S/C6H12N2.C2H2O4.Pt/c7-5-3-1-2-4-6(5)8;3-1(4)2(5)6;/h5-8H,1-4H2;(H,3,4)(H,5,6);/q-2;;+2/t5-,6-;;/m1../s1 |
InChI 键 |
DRMCATBEKSVAPL-BNTLRKBRSA-N |
SMILES |
C1CCC(C(C1)[NH-])[NH-].C(=O)(C(=O)[O-])[O-].[Pt+4] |
手性 SMILES |
C1CC[C@H]([C@@H](C1)[NH-])[NH-].C(=O)(C(=O)O)O.[Pt+2] |
规范 SMILES |
C1CCC(C(C1)[NH-])[NH-].C(=O)(C(=O)O)O.[Pt+2] |
外观 |
white solid powder |
沸点 |
100ºC |
Key on ui other cas no. |
63121-00-6 61825-94-3 |
物理描述 |
Solid |
Pictograms |
Irritant; Health Hazard |
纯度 |
>98% (or refer to the Certificate of Analysis) |
相关CAS编号 |
63121-00-6 63121-00-6 (SP-4-2 (trans)) |
保质期 |
>10 years if stored properly |
溶解度 |
Soluble in water at 4 mg/mL and DMSO at 20 mg/mL; slightly soluble in methanol; insoluble in ethanol. |
储存 |
Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
同义词 |
1,2 Diaminocyclohexane Platinum Oxalate 1,2-diaminocyclohexane platinum oxalate 1,2-diamminocyclohexane(trans-1)oxolatoplatinum(II) ACT 078 ACT-078 ACT078 cis-oxalato-(trans-l)-1,2-diaminocyclohexane-platinum(II) Eloxatin Eloxatine L-OHP cpd oxalato-(1,2-cyclohexanediamine)platinum II oxaliplatin oxaliplatin, (SP-4-2-(1R-trans))-isomer oxaliplatin, (SP-4-2-(1S-trans))-isomer oxaliplatin, (SP-4-3-(cis))-isomer oxaliplatine Platinum(2+) ethanedioate (1R,2R)-1,2-cyclohexanediamine (1:1:1) platinum(II)-1,2-cyclohexanediamine oxalate |
产品来源 |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
体外研究产品的免责声明和信息
请注意,BenchChem 上展示的所有文章和产品信息仅供信息参考。 BenchChem 上可购买的产品专为体外研究设计,这些研究在生物体外进行。体外研究,源自拉丁语 "in glass",涉及在受控实验室环境中使用细胞或组织进行的实验。重要的是要注意,这些产品没有被归类为药物或药品,他们没有得到 FDA 的批准,用于预防、治疗或治愈任何医疗状况、疾病或疾病。我们必须强调,将这些产品以任何形式引入人类或动物的身体都是法律严格禁止的。遵守这些指南对确保研究和实验的法律和道德标准的符合性至关重要。















