Nilotinib
概述
描述
尼洛替尼是一种第二代酪氨酸激酶抑制剂,主要用于治疗费城染色体阳性的慢性髓系白血病(CML)。它以商品名 Tasigna 出售。 尼洛替尼通过抑制 BCR-ABL 酪氨酸激酶发挥作用,BCR-ABL 酪氨酸激酶是由费城染色体异常产生的,并负责白血病细胞的无控制增殖 .
准备方法
尼洛替尼可以通过多种方法合成。一种常见的合成路线涉及将式(IV)化合物转化为尼洛替尼的盐酸盐。 该过程包括使用特定试剂和条件来确保最终产品的稳定性和纯度 . 另一种方法涉及通过将尼洛替尼盐酸盐与药学上可接受的赋形剂压制来制备尼洛替尼的干颗粒制剂 . 此外,纳米制备方法涉及使用 N, N-二甲基甲酰胺和聚乙烯吡咯烷酮来制备稳定的纳米药物浆液,然后喷雾干燥以获得最终产品 .
化学反应分析
尼洛替尼经历着各种化学反应,包括氧化、还原和取代。这些反应中常用的试剂包括氧化剂、还原剂和亲核试剂。 例如,尼洛替尼可以通过在水中使用天然化学连接 (NCL) 化学方法进行苯酰胺形成合成 . 该方法涉及苯甲酰基和巯基苯胺片段的偶联,形成芳香酰胺键。这些反应形成的主要产物包括尼洛替尼的各种衍生物,具有潜在的治疗应用。
科学研究应用
Treatment of Chronic Myeloid Leukemia
Clinical Efficacy
Nilotinib has been extensively studied for its efficacy in treating newly diagnosed chronic-phase CML. The ENESTnd study demonstrated that patients receiving this compound achieved significantly higher rates of major molecular response compared to those treated with imatinib. Specifically, at 12 months, the rates were 44% and 43% for this compound doses of 300 mg and 400 mg twice daily, respectively, versus 22% for imatinib (p < 0.001) .
Long-Term Outcomes
In a phase 2 study involving 122 patients treated with this compound (400 mg twice daily), long-term follow-up indicated that 91% achieved a complete cytogenetic response, with significant event-free survival rates at 89% and overall survival rates at 93% after five years .
Table 1: Summary of Clinical Trials for this compound in CML
| Study | Patient Population | This compound Dose | Major Molecular Response Rate | Event-Free Survival (5 years) | Overall Survival (5 years) |
|---|---|---|---|---|---|
| ENESTnd | Newly diagnosed CML | 300 mg & 400 mg BID | 44% (300 mg), 43% (400 mg) | Not specified | Not specified |
| Phase 2 | Newly diagnosed CML | 400 mg BID | 91% | 89% | 93% |
Potential Applications in Neurodegenerative Diseases
Parkinson's Disease
this compound is being investigated for its potential to alleviate symptoms of Parkinson's disease. A phase II trial indicated that while this compound was generally safe and tolerable, it did not demonstrate significant clinical benefits compared to placebo . The study involved multiple sites and assessed safety across different dosages, revealing more adverse events than the placebo group.
Alzheimer's Disease
Research has also explored this compound's neuroprotective properties in Alzheimer's disease. Preclinical studies suggest that this compound can reduce levels of amyloid-β and prevent neuronal degeneration by inhibiting c-Abl signaling pathways . However, clinical trials have yet to yield robust evidence supporting its efficacy in humans.
Table 2: Summary of this compound Studies in Neurodegenerative Diseases
| Study | Condition | Findings | |
|---|---|---|---|
| NILO-PD | Parkinson's Disease | Safe but no significant clinical benefit | Not recommended for use |
| Preclinical | Alzheimer's Disease | Reduces amyloid-β levels; prevents neuron loss | Promising but unproven in humans |
Safety Profile and Adverse Effects
While this compound is effective against CML, its safety profile warrants attention. Common adverse effects include cardiovascular events, rash, elevated bilirubin, and aminotransferases . In studies involving CML patients, treatment discontinuation due to toxicity occurred in approximately 19% of cases.
Table 3: Adverse Effects Reported in Clinical Trials
| Adverse Effect | Percentage of Patients Affected |
|---|---|
| Rash | 55% |
| Elevated Bilirubin | 57% |
| Elevated Aminotransferases | 48% |
| Cardiovascular Events | 10% |
作用机制
尼洛替尼通过选择性抑制 BCR-ABL 酪氨酸激酶、c-KIT 和血小板衍生生长因子受体 (PDGFR) 发挥作用 . 它与 BCR-ABL 的 ATP 结合位点结合,并抑制其酪氨酸激酶活性,从而阻止参与细胞增殖和存活的下游信号通路的磷酸化和激活。 尼洛替尼对对伊马替尼(另一种酪氨酸激酶抑制剂)耐药的慢性髓系白血病病例特别有效 .
相似化合物的比较
生物活性
Nilotinib is a potent, second-generation tyrosine kinase inhibitor (TKI) primarily used in the treatment of chronic myeloid leukemia (CML). It selectively inhibits the BCR-ABL1 fusion protein, which is responsible for the uncontrolled proliferation of myeloid cells in CML. This article explores the biological activity of this compound, focusing on its mechanisms of action, effects on various cell types, clinical efficacy, and associated adverse effects.
This compound works by binding to the ATP-binding site of the BCR-ABL1 protein, inhibiting its kinase activity. This inhibition prevents the phosphorylation of downstream signaling molecules involved in cell proliferation and survival pathways. The specificity of this compound for the BCR-ABL1 kinase also contributes to its effectiveness and reduced off-target effects compared to earlier TKIs like imatinib.
Effects on Endothelial Cells
Recent studies have highlighted this compound's impact on endothelial cell function, which is critical for vascular health. Research utilizing human induced pluripotent stem cells (hiPSCs) has shown that this compound adversely affects endothelial cell proliferation and migration while increasing intracellular nitric oxide levels. Notably, these effects were independent of ABL1 inhibition, suggesting that this compound may exert off-target effects that compromise endothelial function and contribute to this compound-induced arterial disease (NAD) in some patients .
| Endothelial Cell Function | Effect of this compound |
|---|---|
| Proliferation | Decreased |
| Migration | Decreased |
| Intracellular Nitric Oxide | Increased |
| Barrier Function | No effect |
| Lipid Uptake | No effect |
Clinical Efficacy
This compound has demonstrated significant efficacy in treating CML. In the ENEST1st trial, which included 1089 patients with newly diagnosed CML in chronic phase, this compound achieved a major molecular response (MMR) rate of 38.4% at 18 months . Long-term follow-up studies have confirmed sustained efficacy, with 91% of patients achieving a complete cytogenetic response and a significant proportion reaching deep molecular responses (MR4 and MR4.5) .
Case Study: Long-Term Efficacy
A phase 2 study involving 122 patients treated with this compound at 400 mg twice daily reported:
- Event-Free Survival : 89% at 5 years
- Overall Survival : 93% at 5 years
- Sustained MR4.5 : Achieved by 59% of patients beyond 2 years
- Adverse Events : Cardiovascular events occurred in 10% of patients .
Adverse Effects
Despite its efficacy, this compound is associated with several adverse effects, particularly cardiovascular complications. In clinical trials, ischemic cardiovascular events were reported in approximately 6-10% of patients . Other common side effects include:
- Rash: 55%
- Elevated bilirubin: 57%
- Elevated aminotransferases: 48%
These adverse effects necessitate careful monitoring during treatment.
常见问题
Basic Research Questions
Q. What are the primary methodologies for investigating nilotinib’s mechanism of action in preclinical studies?
- Methodological Answer: Preclinical studies should employ kinase inhibition assays (e.g., KINOMEscan® platform) to profile this compound’s selectivity across 442 kinases . Cell culture models (e.g., endothelial angiogenesis assays) and in vivo murine models are critical for validating on-target BCR-ABL inhibition and off-target effects (e.g., EGFR or FRK signaling) . Ensure characterization of pharmacokinetics, including cerebrospinal fluid (CSF) penetration, to assess bioavailability in neurological studies .
Q. How should researchers design phase 3 trials to evaluate this compound’s efficacy in chronic myeloid leukemia (CML)?
- Methodological Answer: Use randomized controlled trials (RCTs) with comparator arms (e.g., imatinib or dasatinib) and predefined endpoints: major cytogenetic response (MCyR), complete cytogenetic response (CCyR), and progression-free survival (PFS). Account for cross-trial variability by standardizing inclusion criteria (e.g., newly diagnosed chronic-phase CML) and adjusting for geographical/ethnic differences in patient responses .
Q. What statistical methods are recommended for analyzing this compound’s safety and tolerability data?
- Methodological Answer: Apply survival analysis (Kaplan-Meier curves) for time-to-event outcomes (e.g., cardiotoxicity incidence). Use multivariate regression to control for confounders like age, prior cardiovascular disease, or genetic polymorphisms linked to KCNH2 channel dysfunction . Report adverse events with Clopper-Pearson confidence intervals for binary outcomes .
Advanced Research Questions
Q. How can conflicting data on this compound’s cardiotoxicity be resolved in meta-analyses?
- Methodological Answer: Conduct systematic reviews with strict inclusion criteria (e.g., 10-year span, peer-reviewed phase 3 trials) . Stratify studies by patient subgroups (e.g., those with preexisting cardiovascular risk factors) and use GRADE criteria to assess evidence quality. Highlight genetic predispositions (e.g., KCNH2 variants) that may explain heterogeneous toxicity profiles .
Q. What experimental strategies differentiate this compound’s efficacy from other BCR-ABL inhibitors in mutation-driven resistance?
- Methodological Answer: Perform in vitro kinase assays to compare this compound’s binding affinity against BCR-ABL mutants (e.g., T315I) with dasatinib or ponatinib . Use structural biology (e.g., X-ray crystallography) to map resistance-conferring mutations. In clinical studies, prioritize patients with confirmed mutation status via Sanger sequencing or ddPCR .
Q. How can researchers address this compound’s off-target effects on angiogenesis in endothelial cell models?
- Methodological Answer: Employ VEGF-induced angiogenesis assays and phosphoproteomics to identify signaling pathways (e.g., VEGFR2 inhibition by ponatinib vs. EGFR activation by this compound) . Validate findings with siRNA knockdown of proposed off-target kinases (e.g., FRK) and correlate with clinical biomarkers (e.g., HVA/DOAPC levels in CSF) .
Q. What are best practices for reconciling cross-trial discrepancies in this compound’s long-term outcomes?
- Methodological Answer: Use individual patient data (IPD) meta-analysis to harmonize endpoints (e.g., PFS definitions) and adjust for trial design heterogeneity (e.g., dosing schedules, follow-up duration) . Apply counterfactual frameworks to estimate outcomes if crossover between trial arms (e.g., imatinib to this compound) had not occurred .
Q. Methodological Frameworks
Q. How to formulate a PICOT research question for this compound’s role in Parkinson’s disease (PD)?
- Population: Patients with PD and CSF-confirmed dopamine deficiency.
- Intervention: this compound 150–300 mg/day.
- Comparison: Placebo or standard PD therapy.
- Outcome: Change in CSF HVA/DOAPC levels at 12 months.
- Time: 24-month follow-up for safety/tolerability .
Q. What guidelines ensure reproducibility in this compound preclinical studies?
- Follow NIH preclinical reporting standards (e.g., ARRIVE guidelines) for animal studies, including valve calcification assays in murine models . For cell-based work, document passage numbers, serum batches, and assay controls (e.g., imatinib as a comparator) .
Q. Data Analysis and Interpretation
Q. How to interpret this compound’s dual role in promoting autophagy and valvular calcification?
属性
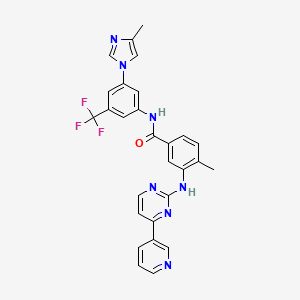
IUPAC Name |
4-methyl-N-[3-(4-methylimidazol-1-yl)-5-(trifluoromethyl)phenyl]-3-[(4-pyridin-3-ylpyrimidin-2-yl)amino]benzamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
HHZIURLSWUIHRB-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=C(C=C(C=C1)C(=O)NC2=CC(=CC(=C2)C(F)(F)F)N3C=C(N=C3)C)NC4=NC=CC(=N4)C5=CN=CC=C5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C28H22F3N7O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID5042663 | |
| Record name | Nilotinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5042663 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
529.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Nilotinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015595 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
The solubility ... in aqueous solutions decreases with increasing pH, 2.01e-03 g/L | |
| Record name | Nilotinib | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7842 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Nilotinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015595 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Chronic myelogenous leukaemia (CML) is caused by the BCR-ABL oncogene. Nilotinib inhibits the tyrosine kinase activity of the BCR-ABL protein. Nilotinib fits into the ATP-binding site of the BCR-ABL protein with higher affinity than imatinib, over-riding resistance caused by mutations. The ability of AMN107 to inhibit TEL-platelet-derived growth factor receptor-beta (TEL-PDGFRbeta), which causes chronic myelomonocytic leukaemia, and FIP1-like-1-PDGFRalpha, which causes hypereosinophilic syndrome, suggests potential use of AMN107 for myeloproliferative diseases characterised by these kinase fusions (Stover et al, 2005; Weisberg et al, 2005). AMN107 also inhibits the c-Kit receptor kinase, including the D816V-mutated variant of KIT, at pharmacologically achievable concentrations, supporting potential utility in the treatment of mastocytosis, and gastrointestinal stromal tumours (Weisberg et al, 2005; von Bubnoff et al, 2005; Gleixner et al, 2006)., Nilotinib, an inhibitor of Bcr-Abl tyrosine kinase, is an antineoplastic agent. Chronic myelogenous leukemia (CML) is a clonal myeloproliferative disorder characterized by the expansion of hematopoietic cells carrying the Philadelphia chromosome (Ph), resulting from a reciprocal translocation of the long arms of chromosomes 9 and 22. A novel fusion gene is formed, Bcr-Abl, which encodes a constitutively active, cytoplasmic form of protein tyrosine kinase. The unregulated activity of the Abl tyrosine kinase in Bcr-Abl is the cause of CML. Nilotinib is an orally active aminopyrimidine-derivative tyrosine kinase inhibitor that functions through competitive inhibition at the ATP-binding site of Bcr-Abl, leading to the inhibition of tyrosine phosphorylation of proteins that are involved in the intracellular signal transduction that Bcr-Abl mediates., Clinical resistance to imatinib in CML has been attributed to several mechanisms, but point mutations in the Bcr-Abl kinase domain appear to the most common, occurring in 30-90% of patients who develop resistance. The ability of nilotinib to overcome imatinib resistance resulting from Bcr-Abl kinase domain mutations has been demonstrated in vitro. In preclinical studies in cell-line models, nilotinib inhibited most (32 of 33) imatinib-resistant Bcr-Abl kinase domain mutant forms., Nilotinib binds to and stabilizes the inactive conformation of the kinase domain of Abl protein. In vitro, nilotinib inhibited Bcr-Abl mediated proliferation of murine leukemic cell lines and human cell lines derived from patients with Ph+ CML. Under the conditions of the assays, nilotinib was able to overcome imatinib resistance resulting from Bcr-Abl kinase mutations, in 32 out of 33 mutations tested. In vivo, nilotinib reduced the tumor size in a murine Bcr-Abl xenograft model. Nilotinib inhibited the autophosphorylation of the following kinases at IC50 values as indicated: Bcr-Abl (20-60 nM), PDGFR (69 nM), c-Kit (210 nM), CSF-1R (125-250 nM) and DDR (3.7 nM)., It is an important challenge to better understand the mechanisms of tyrosine kinase inhibitors-induced apoptosis in CML cells. Thus, /the authors/ have investigated how this apoptosis can be modulated by extracellular factors. Apoptosis induced by imatinib and nilotinib was determined in BCR-ABL expressing cell lines and primary CML CD34+ cells. Both molecules induced apoptosis of BCR-ABL expressing cells. This apoptosis was inhibited by protein synthesis inhibition in both K562 and CML CD34+ cells. In K562, 80% inhibition of the BCR-ABL auto-phosphorylation by either imatinib or nilotinib induced a two fold increase in Bim-EL expression and induction of apoptosis in 48 hr. Bim accumulation preceded apoptosis induction which was completely abolished by depletion in Bim using shRNA. However, the anti-proliferative effect of imatinib was preserved in Bim-depleted cells. When K562 cells were cultured in a cytokine containing medium, the pro-apoptotic effect of nilotinib was decreased by 68% and this was related to a decrease in Bim-EL dephosphorylation and accumulation. Similarly, the presence of a combination of cytokines inhibited 88% of NIL- and 39% of IMA-induced apoptosis in primary CML CD34+ cells. In conclusion, both nilotinib and imatinib induce apoptosis through Bim accumulation independently of cell cycle arrest. However, the pro-apoptotic effect of both molecules can be attenuated by the presence of cytokines and growth factors, particularly concerning nilotinib. Thus BCR-ABL inhibition restores the cytokine dependence but is not sufficient to induce apoptosis when other signaling pathways are activated., For more Mechanism of Action (Complete) data for Nilotinib (7 total), please visit the HSDB record page. | |
| Record name | Nilotinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB04868 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Nilotinib | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7842 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White to slightly yellowish to slightly greenish yellow powder | |
CAS No. |
641571-10-0, 923288-90-8 | |
| Record name | Nilotinib | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=641571-10-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Nilotinib [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0641571100 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Nilotinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB04868 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Nilotinib | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=747599 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Nilotinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5042663 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifluoromethyl)phenyl]-3-{[4-(pyridin-3-yl)pyrimidin-2-yl]amino}benzamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.166.395 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | 4-Methyl-N-(3-(4-methyl-1H-imidazol-1-yl)-5-(trifluoromethyl)-phenyl)-3-((4-(pyridin-3-yl)pyrimidin-2-yl)amino)benzamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | NILOTINIB | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/F41401512X | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Nilotinib | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7842 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Nilotinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015595 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details








Synthesis routes and methods II
Procedure details







Synthesis routes and methods III
Procedure details





Synthesis routes and methods IV
Procedure details











Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
体外研究产品的免责声明和信息
请注意,BenchChem 上展示的所有文章和产品信息仅供信息参考。 BenchChem 上可购买的产品专为体外研究设计,这些研究在生物体外进行。体外研究,源自拉丁语 "in glass",涉及在受控实验室环境中使用细胞或组织进行的实验。重要的是要注意,这些产品没有被归类为药物或药品,他们没有得到 FDA 的批准,用于预防、治疗或治愈任何医疗状况、疾病或疾病。我们必须强调,将这些产品以任何形式引入人类或动物的身体都是法律严格禁止的。遵守这些指南对确保研究和实验的法律和道德标准的符合性至关重要。













