Vicriviroc
描述
维克瑞维洛克是一种嘧啶类化合物,作为 HIV-1 的 CCR5 进入抑制剂。 它由制药公司先灵葆雅开发,并已对其在管理 HIV-1 感染方面的潜力进行了研究 . 维克瑞维洛克抑制 HIV-1 与 CCR5 受体的相互作用,阻止病毒进入宿主细胞 .
准备方法
合成路线和反应条件
维克瑞维洛克的合成涉及多个步骤,从关键中间体的制备开始。 具体反应条件,如温度、溶剂和催化剂,经过优化以确保高产率和纯度 .
工业生产方法
维克瑞维洛克的工业生产遵循类似的合成路线,但已扩展规模以满足商业需求。该过程涉及严格的质量控制措施,以确保一致性和符合监管标准。 先进技术,例如连续流动化学和自动化合成,可能被用来提高效率并降低生产成本 .
化学反应分析
反应类型
维克瑞维洛克经历各种化学反应,包括:
氧化: 维克瑞维洛克可以被氧化形成维克瑞维洛克 N-氧化物,这是一种主要的代谢物.
还原: 还原反应可用于修饰分子中的特定官能团。
常用试剂和条件
这些反应中使用的常用试剂包括用于氧化的细胞色素 P450 酶等氧化剂、用于还原的还原剂和用于取代反应的亲核试剂。 反应条件,如温度、pH 和溶剂选择,经过仔细控制以实现预期的结果 .
形成的主要产品
科学研究应用
Phase 2 Study (ACTG A5211)
- Objective : To assess the safety and virologic activity of vicriviroc in treatment-experienced subjects.
- Design : Double-blind, randomized trial involving 118 subjects who received either this compound (5 mg, 10 mg, or 15 mg) or placebo over 24 weeks.
- Results :
- Significant reductions in HIV-1 RNA levels were observed in all this compound groups compared to placebo.
- The mean change in HIV-1 RNA at week 24 was greater for this compound groups (−1.51 to −1.86 log copies/mL) compared to placebo (+0.06 log copies/mL) with statistical significance () .
- Virologic failure rates were lower in the this compound groups, with time to failure significantly longer for those receiving this compound compared to placebo .
VICTOR-E1 Study
- Objective : To evaluate the molecular basis for resistance mutations in subjects treated with this compound.
- Findings : Among 79 this compound-treated subjects, a subset developed resistance mutations leading to virologic failure. Genetic analysis revealed specific amino acid changes in the viral envelope that were associated with reduced susceptibility to this compound .
Efficacy and Safety Profile
This compound has demonstrated a favorable safety profile across multiple studies. Adverse events were comparable between this compound and placebo groups, with no significant increase in severe adverse events reported . Long-term follow-up indicated sustained virologic suppression in approximately 49% of subjects who achieved viral load suppression below 50 copies/mL after three years .
Resistance Patterns
Resistance to this compound has been documented, primarily among treatment-experienced patients. Studies indicate that while some patients develop resistance mutations, only a small proportion of treatment failures were directly associated with these mutations . The mapping of resistance mutations has revealed diverse genetic alterations that contribute to reduced drug efficacy .
Data Summary Table
| Study Name | Population | Dosage | Key Findings |
|---|---|---|---|
| ACTG A5211 | Treatment-experienced | 5 mg, 10 mg, 15 mg | Significant reduction in HIV-1 RNA levels; longer time to virologic failure |
| VICTOR-E1 | Treatment-experienced | 20 mg, 30 mg | Resistance mutations identified; specific amino acid changes linked to reduced susceptibility |
Conclusions
This compound represents a promising therapeutic option for HIV-1-infected individuals who are treatment-experienced. Its ability to suppress viral load effectively while maintaining a manageable safety profile underscores its potential role in antiretroviral therapy. However, ongoing research is necessary to further elucidate its resistance patterns and optimize its use within combination therapy regimens.
作用机制
维克瑞维洛克作为 CCR5 受体的非竞争性变构拮抗剂。它与 CCR5 受体跨膜螺旋之间靠近细胞外表面的疏水口袋结合。这种结合会导致受体发生构象变化,阻止 HIV-1 gp120 蛋白与 CCR5 结合。 结果,HIV-1 进入宿主细胞被抑制,阻断病毒生命周期的初始阶段 .
相似化合物的比较
维克瑞维洛克属于一类称为 CCR5 抑制剂的化合物。类似的化合物包括:
马拉维洛克: 另一种被批准用于临床治疗 HIV-1 感染的 CCR5 抑制剂.
阿普拉维洛克: 由于肝毒性问题而停止使用的一种 CCR5 抑制剂.
INCB009471: 正在进行临床开发的一种 CCR5 抑制剂.
TBR 652: 正在研究的另一种 CCR5 抑制剂.
维克瑞维洛克因其对多种 HIV 亚型的强大体外活性及其良好的药代动力学和药效学特性而具有独特性 . 它在临床试验中显示出希望,特别是在治疗经验丰富的患者中 .
生物活性
Vicriviroc (VCV), a CCR5 antagonist, is an investigational compound primarily studied for its efficacy in treating HIV-1 infection. This article delves into the biological activity of this compound, summarizing key research findings, clinical trial outcomes, and resistance patterns.
This compound functions by selectively inhibiting the CCR5 coreceptor on CD4+ T cells, which is crucial for the entry of R5-tropic HIV-1 strains. By blocking this receptor, this compound prevents the virus from infecting host cells. This mechanism positions it as a valuable option in antiretroviral therapy, particularly for patients with CCR5-tropic HIV-1.
Phase 2 Studies
Several phase 2 studies have demonstrated this compound's potential in achieving virologic suppression among treatment-experienced patients:
- Study Design : In a randomized, double-blind study involving 118 subjects, participants received either this compound (at doses of 5 mg, 10 mg, or 15 mg) or placebo. The primary endpoint was the change in plasma HIV-1 RNA levels after 14 days of treatment.
- Results :
Long-Term Outcomes
In extended follow-up studies:
- Patients receiving 30 mg of this compound achieved viral loads below 50 copies/mL at rates significantly higher than those on placebo (59% vs. 25%) .
- Over a period of up to 192 weeks, average CD4 counts increased steadily among this compound recipients, indicating sustained immunologic benefit .
Safety Profile
The safety profile of this compound has been generally favorable:
- Adverse events were comparable across treatment groups, with a similar incidence of grade 3/4 adverse events reported .
- However, concerns arose regarding malignancies; six cases were reported among this compound recipients compared to two in the placebo group .
Resistance Patterns
Resistance to this compound has been documented in some patients:
- In the VICTOR-E1 study, five subjects developed VCV-resistant HIV variants after virologic failure .
- Resistance mutations were linked to changes in the V3 loop of the gp160 envelope protein, although no consistent mutation pattern was observed across subjects .
Summary of Key Findings
| Study | Sample Size | Doses | Viral Load Reduction (log copies/mL) | Response Rate (<50 copies/mL) | Adverse Events |
|---|---|---|---|---|---|
| Phase 2 Study | 118 | 5 mg | -1.51 | Not specified | Similar across groups |
| 10 mg | -1.86 | Not specified | Similar across groups | ||
| 15 mg | -1.68 | Not specified | Similar across groups | ||
| VICTOR-E1 | Varied | 20/30 mg | >2 | 59% (30 mg) | Malignancies noted |
常见问题
Basic Research Questions
Q. What experimental models are most appropriate for evaluating Vicriviroc’s mechanism of action as a CCR5 antagonist?
this compound’s mechanism involves blocking HIV-1 entry via CCR5 co-receptor antagonism. In vitro models using CCR5-tropic HIV-1 isolates in peripheral blood mononuclear cells (PBMCs) or cell lines like HEK293/CCR5 are standard for assessing antiviral activity . In vivo models include humanized mice or non-human primates infected with CCR5-tropic HIV-1. Dose-response curves and IC50 values should be calculated using validated assays (e.g., p24 antigen reduction assays) .
Q. How do researchers reconcile discrepancies between tropism assay results (original vs. enhanced-sensitivity) in this compound clinical trials?
Original tropism assays may misclassify dual/mixed (DM) tropic viruses as CCR5-tropic (R5), leading to suboptimal virologic responses. Enhanced-sensitivity assays (e.g., population-based sequencing with deep sequencing) improve detection of minority X4/DM variants. Researchers should retest baseline samples with enhanced assays and stratify outcomes based on updated tropism classifications .
Q. What pharmacokinetic (PK) parameters are critical for optimizing this compound dosing in clinical studies?
Key PK parameters include:
- Ctrough : Maintain concentrations above the protein-binding-adjusted IC90 (e.g., 30 mg daily achieves ~50 ng/mL trough levels) .
- Half-life : this compound’s half-life (~24–30 hours) supports once-daily dosing .
- Drug-drug interactions : Co-administration with ritonavir-boosted protease inhibitors (PI/r) increases this compound exposure by ~2-fold, necessitating dose adjustments .
Advanced Research Questions
Q. How should clinical trials be designed to address this compound’s variable efficacy across doses (e.g., 5 mg vs. 15 mg) in treatment-experienced patients?
Phase II trials (e.g., ACTG A5211) used a double-blind, randomized design with dose escalation (5–30 mg) and optimized background regimens (OBR). Key considerations:
- Primary endpoint : Change in HIV-1 RNA at day 14 (short-term activity) .
- Secondary endpoints : CD4 count changes, safety/tolerability at 24–48 weeks .
- Dose selection : Higher doses (15–30 mg) showed sustained virologic suppression, while lower doses (5 mg) had higher failure rates due to emergent X4 tropism .
| Dose (mg) | Mean HIV-1 RNA Reduction (log10 copies/mL) at 24 Weeks | Virologic Failure Rate (%) |
|---|---|---|
| 5 | -1.51 | 40 |
| 10 | -1.86 | 27 |
| 15 | -1.68 | 33 |
| Data from ACTG A5211 trial |
Q. What methodologies identify this compound resistance in patients with virologic failure despite CCR5-tropic virus?
Resistance mechanisms include:
- Phenotypic assays : Reduced maximal percentage inhibition (MPI) in viral entry assays .
- Genotypic analysis : Env gp160 mutations (e.g., V3 loop changes like G316E/K), though no consistent pattern across subjects .
- Coreceptor switching : Longitudinal monitoring for X4/DM variants via enhanced tropism assays .
Q. How do researchers interpret conflicting data on this compound’s long-term safety, including malignancy risks?
In ACTG A5211, malignancies occurred in 6 this compound recipients vs. 2 placebo recipients, but causality was unclear due to small sample sizes and confounding factors (e.g., advanced HIV disease) . Mitigation strategies:
- Extended follow-up : Monitor malignancy rates in open-label extensions (median follow-up >40 weeks) .
- Comparative safety trials : Use matched cohorts in Phase III studies (e.g., VICTOR-E1) to assess risk-benefit ratios .
Q. Data Analysis & Contradictions
Q. How should researchers address contradictions in this compound’s dose-response relationships across trials?
Conflicting efficacy data (e.g., 15 mg underperforming vs. 10 mg in some studies) may stem from:
- OBR variability : Differential OBR potency across cohorts .
- Tropism assay sensitivity : Enhanced assays reclassify responders/non-responders .
- PK variability : Suboptimal trough levels in certain populations (e.g., CYP3A4 fast metabolizers) . Solutions include stratified analysis by OBR quality and PK-guided dosing.
Q. What statistical approaches are recommended for analyzing this compound’s efficacy in heterogeneous patient populations?
Use mixed-effects models to account for:
属性
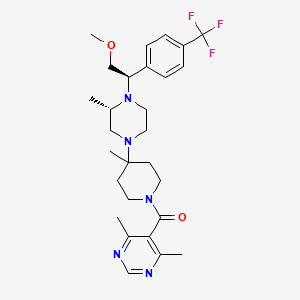
IUPAC Name |
(4,6-dimethylpyrimidin-5-yl)-[4-[(3S)-4-[(1R)-2-methoxy-1-[4-(trifluoromethyl)phenyl]ethyl]-3-methylpiperazin-1-yl]-4-methylpiperidin-1-yl]methanone | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C28H38F3N5O2/c1-19-16-35(14-15-36(19)24(17-38-5)22-6-8-23(9-7-22)28(29,30)31)27(4)10-12-34(13-11-27)26(37)25-20(2)32-18-33-21(25)3/h6-9,18-19,24H,10-17H2,1-5H3/t19-,24-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
CNPVJJQCETWNEU-CYFREDJKSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1CN(CCN1C(COC)C2=CC=C(C=C2)C(F)(F)F)C3(CCN(CC3)C(=O)C4=C(N=CN=C4C)C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@H]1CN(CCN1[C@@H](COC)C2=CC=C(C=C2)C(F)(F)F)C3(CCN(CC3)C(=O)C4=C(N=CN=C4C)C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C28H38F3N5O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID40897719 | |
| Record name | Vicriviroc | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID40897719 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
533.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
Vicriviroc is a once daily oral inhibitor of CCR5. It noncompetitively binds to a hydrophobic pocket between transmembrance helices by the extracellular side of CCR5. This allosteric antagonism causes a conformational change in the protein preventing binding of gp120 to CCR5. This prevents the entry of HIV into the cell. | |
| Record name | Vicriviroc | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06652 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
306296-47-9, 394730-30-4 | |
| Record name | Vicriviroc | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=306296-47-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Vicriviroc [INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0306296479 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | SCH-D (Old RN) | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0394730304 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Vicriviroc | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06652 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Vicriviroc | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID40897719 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | VICRIVIROC | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/TL515DW4QS | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
体外研究产品的免责声明和信息
请注意,BenchChem 上展示的所有文章和产品信息仅供信息参考。 BenchChem 上可购买的产品专为体外研究设计,这些研究在生物体外进行。体外研究,源自拉丁语 "in glass",涉及在受控实验室环境中使用细胞或组织进行的实验。重要的是要注意,这些产品没有被归类为药物或药品,他们没有得到 FDA 的批准,用于预防、治疗或治愈任何医疗状况、疾病或疾病。我们必须强调,将这些产品以任何形式引入人类或动物的身体都是法律严格禁止的。遵守这些指南对确保研究和实验的法律和道德标准的符合性至关重要。















