Atazanavir
Overview
Description
Atazanavir is an antiretroviral medication used primarily to treat HIV/AIDS. It belongs to the protease inhibitor class and works by inhibiting the HIV protease enzyme, which is crucial for the maturation of infectious viral particles. This compound is often used in combination with other antiretroviral agents to enhance its efficacy .
Preparation Methods
Synthetic Routes and Reaction Conditions
The synthesis of atazanavir involves multiple steps, including the use of condensing agents like DEPBT (Diethylphosphoryl cyanide) to facilitate the reaction between 1-[4-(pyridine-2-yl)-phenyl]-4(S)-hydroxy-5(S)-2,5-diamino-6-phenyl-2-azahexane and N-methoxycarbonyl-L-tertiary leucine in an organic solvent . This method is preferred due to its high yield and environmentally friendly nature.
Industrial Production Methods
In industrial settings, this compound is produced using a similar synthetic route but on a larger scale. The process involves the use of automated reactors and stringent quality control measures to ensure the purity and efficacy of the final product .
Chemical Reactions Analysis
Structural Characteristics and Reactivity of Convolvine
Convolvine (8-azabicyclo[3.2.1]octan-3-yl 3,4-dimethoxybenzoate) is a tropane alkaloid isolated from Convolvulus species such as C. pluricaulis and C. subhirsutus . Its structure features a bicyclic nortropane core esterified with 3,4-dimethoxybenzoic acid, conferring reactivity at three key sites:
- Nortropane nitrogen : Susceptible to alkylation and acylation.
- Ester group : Hydrolyzable under acidic/basic conditions.
- Aromatic methoxy groups : Subject to demethylation or electrophilic substitution .
Alkylation Reactions
Alkylation of the nortropane nitrogen produces derivatives with altered bioactivity. Recent studies used alkyl halides (R-X) under basic conditions (K₂CO₃/DMF) :
| Alkyl Group (R) | Reaction Conditions | Yield (%) | Bioactivity Highlight |
|---|---|---|---|
| Methyl | 60°C, 12 hr | 78 | Enhanced antihypoxic |
| Benzyl | 80°C, 24 hr | 65 | Immunomodulatory |
| Ethyl | 60°C, 18 hr | 72 | Anti-inflammatory |
Derivatives showed improved solubility and target specificity compared to the parent compound .
Acylation Reactions
Acylation with anhydrides or acyl chlorides generates esters with modified pharmacokinetics. Key findings include :
| Acylating Agent | Product | Yield (%) | Activity Profile |
|---|---|---|---|
| Acetic anhydride | Acetyl-convolvine | 82 | Antihypoxic (ED₅₀: 12 mg/kg) |
| Maleic anhydride | Maleoyl-convolvine | 68 | Neuroprotective |
| Phthalic anhydride | Phthaloyl-convolvine | 75 | Anti-proliferative |
The acetyl derivative demonstrated 3x higher blood-brain barrier permeability than convolvine .
Palladium-Catalyzed Coupling Reactions
N-Aryl derivatives synthesized via Buchwald-Hartwig amination showed anti-cancer potential :
| Substituent | IC₅₀ (μM) – HeLa Cells | Selectivity Index (vs. HEK293) |
|---|---|---|
| 4-Fluorophenyl | 1.2 ± 0.3 | 18.7 |
| 2-Pyridyl | 2.1 ± 0.5 | 12.4 |
| 3,5-Dimethoxyphenyl | 0.9 ± 0.2 | 22.1 |
This reaction class enables precise tuning of electronic properties for target engagement .
Metabolic and Enzymatic Transformations
In vivo studies in murine models revealed two primary metabolic pathways :
- Ester hydrolysis : Produces 3,4-dimethoxybenzoic acid (t₁/₂ = 45 min).
- N-Demethylation : Forms norconvolvine (CYP3A4-mediated, 22% of metabolites).
A secondary O-demethylation pathway generates catechol intermediates that undergo glucuronidation (detected in urine) .
Stability Under Physicochemical Conditions
| Condition | Degradation Pathway | Half-Life |
|---|---|---|
| pH 1.2 (simulated gastric fluid) | Ester hydrolysis | 28 min |
| pH 7.4 (phosphate buffer) | Oxidative N-demethylation | 6.3 hr |
| UV light (254 nm) | Photooxidation of aromatic rings | 15 min |
Stabilization strategies include microencapsulation (e.g., PLGA nanoparticles extend t₁/₂ to 9.1 hr in plasma) .
Emerging Research Directions
- Electrochemical functionalization : Controlled N-oxidation achieved at 1.2 V vs Ag/AgCl, enabling redox-active derivatives .
- Microwave-assisted synthesis : Reduced reaction times by 78% in acylation steps (45 W, 80°C) .
- Isotope effects : Deuteration at C-2 position lowers activation energy (ΔEₐ = 3.1 kJ/mol) for ester hydrolysis .
These advances position convolvine as a versatile scaffold for medicinal and catalytic chemistry .Key challenges remain in minimizing off-target interactions while preserving the nortropane core’s reactivity profile. Ongoing structure-activity relationship (SAR) studies aim to decouple bioactivity from adrenergic side effects .
Scientific Research Applications
HIV Treatment
Mechanism of Action
Atazanavir is classified as a protease inhibitor, specifically targeting the HIV protease enzyme. By inhibiting this enzyme, ATV prevents the maturation of viral particles, thereby reducing viral load and improving immune function in patients. It is often used in combination with other antiretroviral agents to enhance therapeutic efficacy and reduce resistance development .
Clinical Efficacy
Several studies have demonstrated the effectiveness of ATV in increasing CD4 cell counts and decreasing HIV viral load. A systematic review noted that among 975 children and adolescents treated with ATV, there was a significant improvement in these parameters over 6 to 12 months . The pooled incidence of treatment discontinuation was reported at 19%, highlighting its overall tolerability compared to other treatments like darunavir .
Safety Profile
this compound is associated with fewer lipid abnormalities than other protease inhibitors, making it a favorable option for long-term management of HIV . However, transient hyperbilirubinemia is a common side effect, occurring in approximately half of patients treated with ATV .
Potential Against SARS-CoV-2
Recent research has indicated that this compound may possess antiviral activity against SARS-CoV-2. A study identified ATV as a competitive inhibitor of the SARS-CoV-2 main protease (Mpro), which is crucial for viral replication. In vitro and in vivo experiments showed that ATV could impair the replication of various SARS-CoV-2 variants and reduce virus-induced inflammation and lung damage in infected mice models .
Preclinical Findings
The study highlighted that ATV effectively reached therapeutic concentrations in both plasma and lung tissues, suggesting its potential utility as a treatment option for COVID-19. Further clinical trials are warranted to explore this application more comprehensively .
Drug Repurposing Studies
This compound has also been investigated for its ability to enhance the pharmacokinetics of other drugs. For instance, a recent study explored how ATV could increase the exposure of tizoxanide, an active metabolite used for treating certain infections. This effect arises from ATV's role as a UGT enzyme inhibitor, potentially preventing the rapid metabolism of tizoxanide and enhancing its therapeutic effects .
Case Studies and Clinical Trials
Numerous clinical trials have assessed this compound's effectiveness and safety across different populations:
- Pediatric Populations : A study involving children demonstrated significant improvements in immune function with ATV treatment, reinforcing its use in younger patients .
- Adult Populations : Clinical trials have shown that ATV can maintain viral suppression effectively while minimizing adverse effects related to lipid metabolism .
- COVID-19 Patients : Preliminary studies suggest that ATV may reduce morbidity associated with COVID-19; however, further randomized controlled trials are essential to validate these findings .
Summary Table of this compound Applications
Mechanism of Action
Atazanavir selectively inhibits the virus-specific processing of viral Gag and Gag-Pol polyproteins in HIV-1 infected cells by binding to the active site of HIV-1 protease. This inhibition prevents the formation of mature virions, thereby reducing the viral load in the patient’s body . This compound is not active against HIV-2 .
Comparison with Similar Compounds
Similar Compounds
Darunavir: Another protease inhibitor used in the treatment of HIV/AIDS.
Lopinavir: Often used in combination with ritonavir for enhanced efficacy.
Ritonavir: Used to boost the effectiveness of other protease inhibitors.
Uniqueness
Atazanavir is unique among protease inhibitors due to its once-daily dosing and lesser impact on lipid profiles, making it a preferred choice for patients with concerns about metabolic side effects . Additionally, this compound has shown effectiveness in patients with certain drug-resistant strains of HIV .
Biological Activity
Atazanavir (ATV) is a potent azapeptide protease inhibitor used primarily in the treatment of HIV-1 infection. Its biological activity is characterized by its mechanism of action, pharmacokinetics, efficacy in clinical settings, and safety profile. This article synthesizes findings from various studies to provide a comprehensive overview of this compound's biological activity.
This compound exerts its antiviral effects by selectively inhibiting the HIV-1 protease enzyme, which is crucial for the processing of viral Gag and Gag-Pol polyproteins. By binding to the active site of this enzyme, this compound prevents the maturation of viral particles, thereby inhibiting viral replication . This mechanism is fundamental to its role in antiretroviral therapy.
Pharmacokinetics
This compound is extensively metabolized in the liver, primarily via cytochrome P450 enzymes CYP3A4 and CYP3A5. Genetic polymorphisms affecting these enzymes can influence this compound metabolism and efficacy . The drug also interacts with various drug transporters, which can affect its absorption and distribution in tissues .
Key Pharmacokinetic Parameters
| Parameter | Value |
|---|---|
| Half-life | 7-11 hours |
| Peak plasma concentration (Cmax) | 1-2 hours post-dose |
| Bioavailability | 60% (with food) |
| Metabolism | Hepatic (CYP3A4/5) |
Efficacy in Clinical Studies
This compound has been evaluated in numerous clinical trials, demonstrating its effectiveness in increasing CD4 cell counts and reducing viral loads among HIV-infected patients.
Case Studies and Trial Findings
- Safety and Effectiveness in Children : A study involving 975 children reported a pooled treatment discontinuation rate of 19% at 12 months. This compound was associated with increased CD4 counts and decreased viral loads over time .
- Comparison with Other Regimens : In the ATLAS-M trial, patients on this compound/ritonavir plus lamivudine showed a treatment success rate of 77.4% compared to 65.4% for those on a standard regimen with two NRTIs, indicating superior efficacy for the simplified regimen .
- Long-term Outcomes : The SALT trial confirmed that this compound/ritonavir maintained non-inferior efficacy over longer durations (96 weeks), with no significant benefits in tolerability compared to traditional triple therapy regimens .
Safety Profile
While generally well-tolerated, this compound can cause side effects such as hyperbilirubinemia, gastrointestinal disturbances, and potential renal issues. Notably, transient hyperbilirubinemia occurred in about half of the patients treated with this compound .
Adverse Effects Summary
| Adverse Effect | Incidence (%) |
|---|---|
| Hyperbilirubinemia | ~50% |
| Gastrointestinal issues | Varies by individual |
| Renal impairment | Rare but documented |
Properties
IUPAC Name |
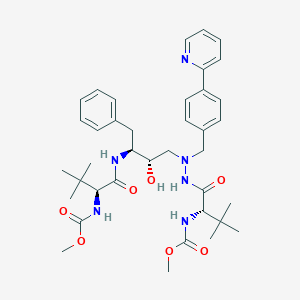
methyl N-[(2S)-1-[2-[(2S,3S)-2-hydroxy-3-[[(2S)-2-(methoxycarbonylamino)-3,3-dimethylbutanoyl]amino]-4-phenylbutyl]-2-[(4-pyridin-2-ylphenyl)methyl]hydrazinyl]-3,3-dimethyl-1-oxobutan-2-yl]carbamate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C38H52N6O7/c1-37(2,3)31(41-35(48)50-7)33(46)40-29(22-25-14-10-9-11-15-25)30(45)24-44(43-34(47)32(38(4,5)6)42-36(49)51-8)23-26-17-19-27(20-18-26)28-16-12-13-21-39-28/h9-21,29-32,45H,22-24H2,1-8H3,(H,40,46)(H,41,48)(H,42,49)(H,43,47)/t29-,30-,31+,32+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
AXRYRYVKAWYZBR-GASGPIRDSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)(C)C(C(=O)NC(CC1=CC=CC=C1)C(CN(CC2=CC=C(C=C2)C3=CC=CC=N3)NC(=O)C(C(C)(C)C)NC(=O)OC)O)NC(=O)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC(C)(C)[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)[C@H](CN(CC2=CC=C(C=C2)C3=CC=CC=N3)NC(=O)[C@H](C(C)(C)C)NC(=O)OC)O)NC(=O)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C38H52N6O7 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID9048691 | |
| Record name | Atazanavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9048691 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
704.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Atazanavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015205 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Free base slightly soluble (4-5 mg/mL), 3.27e-03 g/L | |
| Record name | Atazanavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01072 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Atazanavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015205 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Atazanavir selectively inhibits the virus-specific processing of viral Gag and Gag-Pol polyproteins in HIV-1 infected cells by binding to the active site of HIV-1 protease, thus preventing the formation of mature virions. Atazanavir is not active against HIV-2., Atazanavir is an azapeptide HIV-1 protease inhibitor. The compound selectively inhibits the virus-specific processing of viral Gag and Gag-Pol polyproteins in HIV-1 infected cells, thus preventing formation of mature virions., BMS-232632 is an azapeptide human immunodeficiency virus type 1 (HIV-1) protease (Prt) inhibitor that exhibits potent anti-HIV activity with a 50% effective concentration (EC(50)) of 2.6 to 5.3 nM and an EC(90) of 9 to 15 nM in cell culture. Proof-of-principle studies indicate that BMS-232632 blocks the cleavage of viral precursor proteins in HIV-infected cells, proving that it functions as an HIV Prt inhibitor. Comparative studies showed that BMS-232632 is generally more potent than the five currently approved HIV-1 Prt inhibitors. Furthermore, BMS-232632 is highly selective for HIV-1 Prt and exhibits cytotoxicity only at concentrations 6,500- to 23, 000-fold higher than that required for anti-HIV activity. To assess the potential of this inhibitor when used in combination with other antiretrovirals, BMS-232632 was evaluated for anti-HIV activity in two-drug combination studies. Combinations of BMS-232632 with either stavudine, didanosine, lamivudine, zidovudine, nelfinavir, indinavir, ritonavir, saquinavir, or amprenavir in HIV-infected peripheral blood mononuclear cells yielded additive to moderately synergistic antiviral effects. Importantly, combinations of drug pairs did not result in antagonistic anti-HIV activity or enhanced cytotoxic effects at the highest concentrations used for antiviral evaluation. Our results suggest that BMS-232632 may be an effective HIV-1 inhibitor that may be utilized in a variety of different drug combinations. | |
| Record name | Atazanavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01072 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | ATAZANAVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7339 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
198904-31-3 | |
| Record name | Atazanavir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=198904-31-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Atazanavir [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0198904313 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Atazanavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01072 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Atazanavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9048691 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | methyl N-[(2S)-1-[2-[(2S,3S)-2-hydroxy-3-[[(2S)-2-(methoxycarbonylamino)-3,3-dimethylbutanoyl]amino]-4-phenylbutyl]-2-[(4-pyridin-2-ylphenyl)methyl]hydrazinyl]-3,3-dimethyl-1-oxobutan-2-yl]carbamate | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.243.594 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | ATAZANAVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/QZU4H47A3S | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | ATAZANAVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7339 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Atazanavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015205 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















