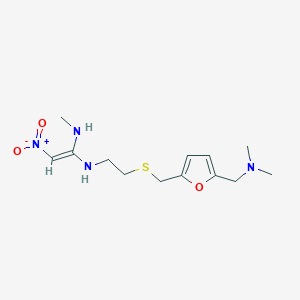
Ranitidine
Overview
Description
Ranitidine is a histamine H2-receptor antagonist that was widely used to decrease stomach acid production. It was commonly prescribed for the treatment of peptic ulcer disease, gastroesophageal reflux disease, and Zollinger-Ellison syndrome. This compound was discovered in England in 1976 and came into commercial use in 1981. It was marketed under the brand name Zantac, among others .
Preparation Methods
Synthetic Routes and Reaction Conditions: Ranitidine can be synthesized through multiple pathways. One common method involves the intermediate 5-(dimethylamino)furfurylthioethylamine. The synthesis begins with furfuryl alcohol, which undergoes a series of reactions to form the intermediate. This intermediate is then reacted with l-methylthio-l-(N-methylamino)-2-nitroethylene to produce this compound .
Industrial Production Methods: Industrial production of this compound typically involves the use of organic solvents and moderate reaction conditions. For instance, the compound can be synthesized by treating an intermediate with N,N-dimethylaminotriphenylphosphonium salts and dimethylamine at around 90°C in organic solvents like dimethylformamide .
Chemical Reactions Analysis
Types of Reactions: Ranitidine undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized under certain conditions, leading to the formation of different by-products.
Common Reagents and Conditions:
Oxidation: Common oxidizing agents can be used to oxidize this compound.
Photolysis: Photolysis reactions typically require exposure to light and can be influenced by the presence of natural organic matter.
Major Products Formed:
Oxidation: Various oxidation products can be formed, depending on the specific conditions and reagents used.
Photolysis: Photolysis of this compound can lead to the formation of multiple degradation products.
Scientific Research Applications
Gastrointestinal Disorders
1.1 Peptic Ulcer Disease
Ranitidine is primarily indicated for the treatment of peptic ulcers. Clinical studies have shown that it is effective in promoting healing of both gastric and duodenal ulcers. A meta-analysis indicated that this compound, at a dosage of 300 mg daily, is at least as effective as cimetidine in ulcer healing rates .
1.2 Gastroesophageal Reflux Disease (GERD)
In patients with GERD, this compound has demonstrated significant efficacy in reducing heartburn frequency and severity. A clinical trial involving 284 patients found that this compound treatment resulted in a marked decrease in heartburn symptoms compared to placebo, with improved endoscopic findings in patients with erosive esophagitis .
1.3 Zollinger-Ellison Syndrome
this compound is also utilized in the management of Zollinger-Ellison syndrome, a condition characterized by excessive gastric acid production. Studies confirm that this compound effectively reduces gastric acid secretion in these patients, thereby alleviating symptoms and preventing complications .
Surgical Applications
2.1 Prevention of Aspiration Pneumonitis
this compound has been used preoperatively to reduce the risk of aspiration pneumonitis during anesthesia induction. It is particularly administered to pregnant women and patients undergoing surgery to minimize gastric acidity and volume .
2.2 Stress Ulcer Prophylaxis
In critically ill patients, this compound is often prescribed for stress ulcer prophylaxis to prevent gastrointestinal bleeding due to stress-induced mucosal disease. Its role in this context remains under investigation but shows promise based on existing data .
Pediatric Use
This compound has been found safe for use in pediatric populations when dosed appropriately. It has been effective in treating conditions such as gastroesophageal reflux and peptic ulcers in children, with studies supporting its safety profile .
Analytical Studies and Validation
This compound's pharmacokinetics and efficacy have been extensively studied, leading to the establishment of validated analytical methods for its measurement in biological samples. These methods ensure accurate dosing and therapeutic monitoring .
Safety Considerations and Controversies
Recent concerns regarding the presence of N-nitrosodimethylamine (NDMA), a potential carcinogen, have led to increased scrutiny of this compound products. Regulatory agencies have issued recalls and advisories, prompting ongoing research into the long-term safety of this compound use .
Comparative Studies
A multinational cohort study investigated the cancer risk associated with this compound compared to other H2 receptor antagonists. The findings suggested no significant increase in cancer incidence among long-term users of this compound compared to those using alternatives like famotidine and lafutidine .
Data Summary Table
Mechanism of Action
Ranitidine works by blocking histamine H2 receptors in the stomach lining. Histamine, released from enterochromaffin-like cells, binds to these receptors and stimulates the secretion of gastric acid. By blocking these receptors, this compound reduces the production of gastric acid, thereby alleviating symptoms associated with excess stomach acid .
Comparison with Similar Compounds
Cimetidine: The first H2-receptor antagonist discovered. It has a similar mechanism of action but a different chemical structure.
Famotidine: Another H2-receptor antagonist with a longer duration of action and fewer side effects compared to ranitidine.
Uniqueness of this compound: this compound was preferred over cimetidine due to its improved side effect profile and potency. concerns about the presence of N-nitrosodimethylamine in this compound products have led to its withdrawal from many markets .
Biological Activity
Ranitidine, a histamine H2 receptor antagonist, has been widely used for the treatment of various gastrointestinal disorders, including duodenal ulcers, gastric ulcers, gastroesophageal reflux disease (GERD), and Zollinger-Ellison syndrome. Its primary mechanism of action involves the reversible binding to H2 receptors on gastric parietal cells, leading to a reduction in gastric acid secretion. This article explores the biological activity of this compound, focusing on its pharmacological effects, potential risks associated with its use, and recent research findings.
This compound functions by blocking the action of histamine at the H2 receptors located on the gastric parietal cells. This interaction inhibits the secretion of gastric acid, providing relief from conditions associated with excess stomach acid. The onset of action typically occurs within 60 minutes after administration, with effects lasting up to 12 hours .
Pharmacological Effects
- Acid Secretion Inhibition : this compound significantly reduces gastric acid secretion, which is beneficial in treating acid-related disorders.
- Gastrin Levels : Prolonged use of this compound can lead to increased plasma gastrin levels due to feedback mechanisms triggered by reduced acidity .
- Neuronal Effects : Recent studies suggest this compound may have effects beyond acid inhibition, such as alleviating anxiety-like behaviors in animal models by enhancing exploratory locomotion and reducing anxiety-related behaviors .
Cancer Risk Studies
The safety profile of this compound has come under scrutiny due to concerns regarding its potential association with cancer risk:
- A multinational cohort study involving over 1.1 million individuals found no significant increase in cancer risk among new users of this compound compared to other H2 receptor antagonists (H2RAs). The crude incidence rates were similar, suggesting that this compound does not pose a higher risk for cancer than its counterparts .
- Conversely, another study indicated a statistically significant association between this compound use and an increased risk for certain gastrointestinal cancers (stomach and esophageal), with proportionate reporting ratios suggesting elevated risks for various cancers .
NDMA Contamination Concerns
One of the major concerns regarding this compound is its potential to form N-nitrosodimethylamine (NDMA), a probable human carcinogen:
- In vitro studies demonstrated that NDMA formation could occur under specific conditions involving high levels of nitrites in gastric fluid. However, rigorous clinical trials have shown no evidence of elevated NDMA levels in urine after administering this compound at standard doses .
- The FDA's studies indicated that this compound would only produce NDMA in simulated conditions far exceeding normal physiological levels of nitrite .
Case Studies
Several case studies have provided insights into the biological activity and safety profile of this compound:
- Study on Anxiety Reduction : In an experimental model using cysteamine HCl-treated mice, those treated with this compound exhibited significantly increased exploratory behavior compared to control groups. This suggests potential neuroactive properties beyond its gastrointestinal applications .
- Long-term Treatment Effects : A study conducted over 106 weeks in rats revealed that prolonged this compound treatment was associated with hyperplasia of gastric ECL cells and carcinoid tumors, raising concerns about long-term use in humans .
Summary Table: Key Findings on this compound
Q & A
Basic Research Questions
Q. How should researchers design initial experiments to investigate ranitidine degradation pathways in environmental matrices?
- Methodological Guidance: Utilize advanced oxidation processes (AOPs) such as UV/ultrasound or ozonation paired with LC-Q-ToF-MS for identifying transformation products. Experimental parameters (e.g., pH, oxidant concentration) must be systematically varied using factorial designs to isolate degradation mechanisms . For replication, document storage conditions (e.g., temperature) to account for NDMA formation variability .
Q. What statistical approaches are appropriate for analyzing this compound's pharmacodynamic effects in preclinical models?
- Methodological Guidance: Apply two-way repeated-measures ANOVA to compare dose-response curves (e.g., muscle strip contractions) and non-parametric tests (Wilcoxon rank test) for pharmacokinetic variables (Cmax, AUC). Ensure sample sizes are powered to detect clinically relevant effect sizes .
Q. How can researchers structure literature reviews to address this compound's carcinogenicity controversies?
- Methodological Guidance: Conduct a systematic review using PRISMA guidelines, prioritizing studies with rigorous designs (e.g., long-term cohort studies with NDMA exposure quantification). Critically assess confounding factors (e.g., storage conditions, prescription vs. OTC use) and heterogeneity across databases .
Advanced Research Questions
Q. What experimental designs optimize the detection of this compound's interaction with organic cation transporters (OCTs) in vitro?
- Methodological Guidance: Use HEK293 cells overexpressing OCT1/OCT3 to measure time- and concentration-dependent uptake. Include control cells (empty vector) to isolate transporter-specific effects. Validate findings with genetic polymorphism data (e.g., OCT1 SNPs) to assess clinical relevance .
Q. How can response surface methodology (RSM) improve this compound synthesis optimization?
- Methodological Guidance: Implement a two-stage RSM approach:
Screening experiments via fractional factorial designs (e.g., 2<sup>6-2</sup> IV) to identify critical factors (e.g., solvent ratio, temperature).
Central composite designs to model non-linear relationships and predict optimal conditions. Transform responses (e.g., ln(CEF)) to address heteroscedasticity .
Q. What strategies resolve contradictory findings in this compound-cancer epidemiologic studies?
- Methodological Guidance:
- Perform meta-analyses with sensitivity analyses to exclude underpowered studies.
- Leverage federated network cohorts (e.g., Observational Health Data Sciences and Informatics) to harmonize multi-national EHR data, ensuring standardized NDMA exposure metrics and sufficient follow-up duration .
- Apply causal inference methods (e.g., propensity score stratification) to address confounding by indication .
Q. How can researchers validate NDMA generation from this compound under simulated gastric conditions?
- Methodological Guidance: Replicate Sloan Kettering’s protocol by incubating this compound in gastric fluid analogs (pH 1–3) at physiological temperatures. Quantify NDMA via GC-MS/MS and correlate with this compound batch variability. Include positive controls (e.g., nitrite-spiked samples) to confirm assay validity .
Q. Data Management & Reproducibility
Q. What metadata standards are critical for sharing this compound-related research data?
- Methodological Guidance: Adhere to FAIR principles:
- F indable: Assign DOIs via repositories (e.g., Zenodo).
- A ccessible: Describe experimental protocols using MIAME or similar frameworks.
- I nteroperable: Use standardized ontologies (e.g., ChEBI for chemical identifiers).
- R eusable: Provide raw LC-MS/MS spectra and analysis scripts (R/Python) .
Q. How can researchers ensure reproducibility in this compound pharmacokinetic studies?
- Methodological Guidance:
- Publish full chemical characterization (e.g., purity, storage conditions) and species-specific OCT expression profiles.
- Pre-register animal/human protocols on platforms like OSF, detailing inclusion/exclusion criteria and statistical plans .
Q. Ethical & Translational Considerations
Q. What ethical frameworks apply to retrospective studies on this compound's carcinogenicity?
Properties
IUPAC Name |
(E)-1-N'-[2-[[5-[(dimethylamino)methyl]furan-2-yl]methylsulfanyl]ethyl]-1-N-methyl-2-nitroethene-1,1-diamine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C13H22N4O3S/c1-14-13(9-17(18)19)15-6-7-21-10-12-5-4-11(20-12)8-16(2)3/h4-5,9,14-15H,6-8,10H2,1-3H3/b13-9+ | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
VMXUWOKSQNHOCA-UKTHLTGXSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CNC(=C[N+](=O)[O-])NCCSCC1=CC=C(O1)CN(C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CN/C(=C\[N+](=O)[O-])/NCCSCC1=CC=C(O1)CN(C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C13H22N4O3S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID101112063 | |
| Record name | (1E)-N-[2-[[[5-[(Dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-N′-methyl-2-nitro-1,1-ethenediamine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID101112063 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
314.41 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Ranitidine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001930 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Water soluble | |
| Record name | RANITIDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3925 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
H2 antagonists inhibit gastric acid secretion elicited by histamine and other H2 agonists in a dose dependent, competitive manner; the degree of inhibition parallels the concentration of the drug in plasma over a wide range. The H2 antagonists also inhibit acid secretion elicited by gastrin and, to a lesser extent, by muscarinic agonists. Importantly, these drugs inhibit basal (fasting) and nocturnal acid secretion and that stimulated by food, sham feeding, fundic distention, and various pharmacological agents; this property reflects the vital role of histamine in mediating the effects of diverse stimuli. /H2 Receptor Antagonists/, ... /H2 Antagonists/ measurably inhibit effects on the cardiovascular and other systems that are elicited through H2 receptors by exogenous or endogenous histamine. /H2 Receptor Antagonists/, ...IS A COMPETITIVE ANTAGONIST OF HISTAMINE-INDUCED GASTRIC ACID SECRETION... INHIBITS BOTH THE VOLUME AND CONCENTRATION OF GASTRIC ACID INDUCED NOCTURNALLY AND BY FOOD BUT DOES NOT AFFECT GASTRIC MUCUS OR ITS PRODUCTION. ...DOES NOT AFFECT LOWER ESOPHAGEAL SPHINCTER PRESSURE... | |
| Record name | RANITIDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3925 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
SOLID | |
CAS No. |
82530-72-1, 66357-35-5 | |
| Record name | (1E)-N-[2-[[[5-[(Dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-N′-methyl-2-nitro-1,1-ethenediamine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=82530-72-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | ranitidine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757851 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | (1E)-N-[2-[[[5-[(Dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-N′-methyl-2-nitro-1,1-ethenediamine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID101112063 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Ranitidine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.060.283 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | RANITIDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3925 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Ranitidine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001930 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
69-70 °C, MP: 133-134 °C /RATINIDINE HYDROCHLORIDE/ | |
| Record name | RANITIDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3925 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















