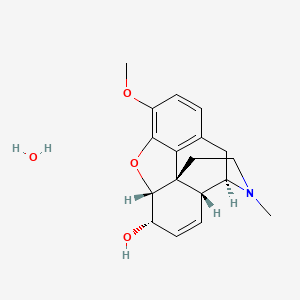
Codeine monohydrate
Overview
Description
Codeine is a naturally occurring alkaloid found in the sap of the opium poppy, Papaver somniferum. It is an opiate and a prodrug of morphine, primarily used to treat pain, coughing, and diarrhea . Codeine is commonly used in medicine due to its analgesic, antitussive, and antidiarrheal properties .
Preparation Methods
Synthetic Routes and Reaction Conditions: This involves reacting morphine with trimethyl phenyl ammonium chloride in the presence of an alkali metal carbonate and a hydrocarbon solvent at temperatures ranging from 40°C to 120°C .
Industrial Production Methods: In industrial settings, codeine is often extracted from opium or synthesized from morphine. The extraction process involves isolating morphine from opium and then converting it to codeine through methylation .
Types of Reactions:
Oxidation: Codeine can undergo oxidation to form codeinone.
Reduction: Codeine can be reduced to dihydrocodeine.
Substitution: Codeine can undergo substitution reactions, such as N-demethylation to form norcodeine.
Common Reagents and Conditions:
Oxidation: Reagents like potassium permanganate or chromium trioxide.
Reduction: Reagents like hydrogen in the presence of a palladium catalyst.
Substitution: Reagents like sodium hydroxide for N-demethylation.
Major Products:
Oxidation: Codeinone.
Reduction: Dihydrocodeine.
Substitution: Norcodeine.
Scientific Research Applications
Pharmacokinetics
Codeine is rapidly absorbed from the gastrointestinal tract, with less than 40% of an orally ingested dose reaching systemic circulation. The peak plasma concentration occurs within 1-2 hours, and its half-life ranges from 2.5 to 3.5 hours . Codeine's solubility and bioavailability are crucial for its effectiveness in various formulations.
Pain Management
Codeine is primarily indicated for the treatment of mild to moderate pain. It is often prescribed in combination with non-opioid analgesics like acetaminophen or non-steroidal anti-inflammatory drugs (NSAIDs) for enhanced efficacy. Despite its common use, recent studies have raised questions about its effectiveness compared to other analgesics .
Cough Suppression
As an antitussive agent, codeine is effective in managing chronic cough associated with various etiologies. Its efficacy can vary based on underlying conditions, and alternatives such as dextromethorphan are sometimes preferred due to superior effectiveness .
Other Indications
- Restless Leg Syndrome : Codeine may be beneficial for refractory cases of restless leg syndrome when other treatments fail.
- Diarrhea : It is occasionally used in palliative care settings to manage persistent diarrhea .
Anticancer Potential
Recent studies have explored the potential anticancer properties of codeine. In vitro studies demonstrated that codeine can reduce cell viability in certain cancer cell lines, suggesting a possible role in cancer prevention or therapy. For instance, codeine showed significant growth inhibition in MCF-7 breast cancer cells and AGS gastric cancer cells when combined with other agents .
Antibacterial Activity
Research has also indicated that codeine complexes exhibit antimicrobial properties against various pathogens, suggesting potential applications beyond traditional uses .
Solubility Studies
Investigations into the solubility of codeine phosphate in supercritical carbon dioxide have been conducted to optimize drug delivery systems. Understanding solubility is essential for enhancing bioavailability and therapeutic effectiveness through micronization techniques .
Pain Management Case Study
A clinical trial involving patients with chronic pain evaluated the efficacy of codeine combined with acetaminophen versus acetaminophen alone. Results indicated that patients receiving the combination experienced greater pain relief without a significant increase in adverse effects.
Cough Management Case Study
A study on patients with chronic cough assessed the effectiveness of codeine syrup compared to dextromethorphan. Findings revealed that while both medications reduced cough frequency, dextromethorphan was preferred by patients due to fewer side effects and better overall satisfaction .
Mechanism of Action
Codeine exerts its effects by binding to the mu-opioid receptors in the brain and spinal cord. This binding results in the inhibition of nociceptive neurotransmitter release, leading to analgesia and increased pain tolerance . Codeine is metabolized in the liver by the enzyme cytochrome P450 2D6 to form morphine, which is responsible for its analgesic effects .
Comparison with Similar Compounds
Morphine: A more potent analgesic with a higher risk of addiction.
Hydrocodone: Similar to codeine but more potent and often used for severe pain.
Oxycodone: Another potent opioid used for managing moderate to severe pain.
Uniqueness of Codeine: Codeine is unique in its relatively mild potency and lower risk of addiction compared to other opioids like morphine and oxycodone. It is often preferred for treating mild to moderate pain and cough due to its favorable safety profile .
Biological Activity
Codeine monohydrate, a naturally occurring alkaloid derived from opium poppy, is primarily used for its analgesic and antitussive properties. Its biological activity is largely attributed to its interaction with the central nervous system through opioid receptors, particularly the mu-opioid receptor. This article delves into the pharmacodynamics, pharmacokinetics, metabolism, and clinical implications of this compound, supported by case studies and research findings.
Pharmacodynamics
Mechanism of Action
Codeine exerts its analgesic effects by binding to mu-opioid receptors in the brain and spinal cord, leading to a decrease in the perception of pain. It also interacts with kappa and delta opioid receptors, albeit to a lesser extent. This action results in hyperpolarization of nociceptive neurons, effectively impairing pain signal transmission .
Antitussive Activity
In addition to its analgesic properties, codeine is recognized for its antitussive effects. It suppresses cough reflexes by acting on the cough center in the medulla oblongata . Clinical trials have demonstrated its efficacy in treating cough associated with conditions such as tuberculosis .
Pharmacokinetics
Absorption and Bioavailability
Codeine is rapidly absorbed from the gastrointestinal tract, with peak plasma concentrations typically reached within 1-2 hours post-administration. The bioavailability of codeine ranges from 30% to 40% due to extensive first-pass metabolism in the liver .
Metabolism
Approximately 70-80% of codeine is metabolized in the liver. Key metabolic pathways include:
- O-Demethylation : Converts codeine into morphine (5-10%).
- N-Demethylation : Produces norcodeine (10%).
- Glucuronidation : Forms codeine-6-glucuronide (C6G), which is pharmacologically active .
The enzyme CYP2D6 plays a crucial role in converting codeine to morphine; genetic polymorphisms affecting CYP2D6 can lead to significant variability in individual responses to codeine .
Elimination
The elimination half-life of codeine ranges from 2.5 to 4 hours, with renal excretion being the primary route for its metabolites .
Distribution
Codeine is widely distributed throughout body tissues and can cross the blood-brain barrier as well as the placenta, which raises considerations for use during pregnancy and breastfeeding . The apparent volume of distribution is approximately 3-6 L/kg, indicating extensive tissue binding .
Clinical Implications
Case Studies
- A case study highlighted myopathy and coordination issues linked to prolonged use of codeine linctus, suggesting potential adverse effects associated with chronic administration .
- Another study discussed interactions between codeine and other medications (e.g., carbamazepine), emphasizing the importance of monitoring for adverse reactions when co-administered with other central nervous system depressants .
Summary Table of this compound Characteristics
| Characteristic | Details |
|---|---|
| Chemical Structure | C18H21NO3 (with monohydrate form) |
| Mechanism of Action | Mu-opioid receptor agonist |
| Bioavailability | 30-40% |
| Peak Plasma Concentration | 1-2 hours after oral administration |
| Half-Life | 2.5 - 4 hours |
| Metabolites | Morphine, norcodeine, C6G |
| Primary Metabolism Enzymes | CYP2D6, UGT2B7, UGT2B4 |
| Excretion Route | Renal (urinary) |
Chemical Reactions Analysis
Dehydration and Hydrate Stability
Codeine monohydrate (C₁₈H₂₁NO₃·H₂O) is efflorescent, readily losing its water of crystallization under ambient or dry conditions to form anhydrous codeine . This dehydration process is reversible, with hydration states affecting its crystallinity and solubility:
-
Hydrate Stability :
Halogenation Reactions
This compound participates in regioselective halogenation at position 1 (C1) or 2 (C2) of its aromatic ring under controlled conditions:
C1 Halogenation
C2 Halogenation
-
2-Chloromorphine Synthesis :
Nitration at C2 followed by reduction and Sandmeyer reaction produces 2-chlorocodeine (37 ) .
Functionalization via Palladium-Catalyzed Coupling
1-Bromocodeine derivatives undergo cross-coupling reactions to introduce diverse functional groups :
| Reaction Type | Substrate | Product | Yield |
|---|---|---|---|
| Heck Coupling | Methyl acrylate | 1-Vinylcodeine (22a ) | 39–85% |
| Stille Coupling | Tetramethyltin | 1-Methylcodeine (25a ) | 50–85% |
| Carbonylation | CO/PdCl₂ | 1-Ethoxycarbonylcodeine (24 ) | 50% |
Acid-Base Reactions
As a tertiary amine, this compound reacts with acids to form salts, critical for pharmaceutical formulations :
-
Salt Formation :
Oxidation and Electrochemical Behavior
Codeine undergoes oxidation at carbon electrodes, enabling electrochemical detection :
| Electrode | Method | LOD | Application |
|---|---|---|---|
| Boron-doped diamond (BDD) | Chronoamperometry | 20,000 nM | Tablet/urine analysis |
| Screen-printed carbon | CYP2D6-modified | 4,900 nM | Biosensing |
The oxidation mechanism involves electron transfer at the phenolic hydroxyl group .
Nitration and Reduction
-
1-Nitrocodeine Synthesis :
Nitration with HNO₃ followed by reduction (SnCl₂/HCl or formamidinesulfinic acid) yields 1-aminocodeine (12 ), a versatile intermediate for azo or thiazole derivatives . -
2-Aminothiazole Formation :
Microwave-assisted reaction with bromine/potassium thiocyanate produces A-ring-fused thiazoles (26 ) .
Stability and Incompatibilities
This compound exhibits reactivity with specific reagents :
Solubility in Supercritical CO₂
Solubility data for codeine phosphate (derived from this compound) in SC-CO₂ informs drug micronization :
| Temperature (K) | Pressure (MPa) | Mole Fraction (×10⁻⁵) |
|---|---|---|
| 308 | 12 | 1.297 |
| 338 | 27 | 6.502 |
Modeled effectively with the Peng-Robinson equation (AARD% < 12.63%) .
Q & A
Basic Research Questions
Q. How can researchers analytically distinguish codeine monohydrate from other hydrate forms (e.g., hemihydrate or anhydrous)?
- Methodology : Use a combination of X-ray diffraction (XRD) to identify crystalline structure and thermogravimetric analysis (TGA) to quantify water content. Spectroscopy (e.g., FTIR or NMR) can confirm functional groups and hydrogen bonding patterns specific to monohydrate forms .
- Key Data : Compare TGA mass loss at ~100°C (indicative of water loss) with theoretical calculations for monohydrate stoichiometry .
Q. What experimental protocols ensure stability of this compound in long-term pharmacological studies?
- Methodology : Conduct accelerated stability studies under controlled humidity (e.g., 40–75% RH) and temperature (25–40°C). Monitor degradation products via HPLC-MS and validate methods per ICH guidelines .
- Critical Step : Use desiccants in storage and verify batch consistency with reference standards (e.g., BP or USP monographs) .
Q. How should researchers design in vitro assays to assess this compound’s opioid receptor binding affinity?
- Methodology : Employ competitive binding assays with radiolabeled ligands (e.g., [³H]-naloxone) in transfected cell lines. Normalize data to control ligands (e.g., morphine) and account for pH-dependent solubility differences in monohydrate formulations .
Advanced Research Questions
Q. What strategies resolve contradictions in this compound’s pharmacokinetic data across preclinical models?
- Methodology : Perform species-specific metabolite profiling (e.g., CYP2D6 activity in humanized mice) and validate using physiologically based pharmacokinetic (PBPK) modeling . Cross-reference human clinical trial data to adjust for interspecies variability .
- Example : Discrepancies in bioavailability may arise from hydration-state-dependent solubility; use biorelevant media (e.g., FaSSIF) to simulate intestinal absorption .
Q. How can synthetic routes for this compound be optimized to minimize diastereomer formation?
- Methodology : Apply asymmetric catalysis (e.g., chiral ligands in phosphorylation) and monitor reaction intermediates via LC-TOF-MS . Optimize crystallization conditions (e.g., solvent polarity, cooling rates) to favor monohydrate nucleation .
- Validation : Compare synthetic yields and purity against reference materials (e.g., CAS 6059-47-8) .
Q. What statistical approaches are recommended for analyzing dose-response relationships in this compound toxicity studies?
- Methodology : Use non-linear regression models (e.g., Hill equation) with bootstrapping to estimate EC₅₀ values. Address batch variability via mixed-effects models and validate assumptions with Q-Q plots .
- Reporting : Follow CONSORT guidelines for preclinical data, including effect sizes and confidence intervals .
Q. Methodological Best Practices
Q. How to ensure reproducibility in this compound research?
- Protocols : Document synthesis and characterization steps in line with Beilstein Journal of Organic Chemistry guidelines. Include raw data (e.g., XRD spectra, chromatograms) in supplementary materials .
- Quality Control : Use certified reference standards (e.g., Mikromol’s MM0442.00) and report purity thresholds (>98%) .
Q. What ethical considerations apply when designing human studies involving this compound?
- Framework : Integrate Cochrane Review principles —systematic evidence synthesis, bias minimization, and consumer perspectives. Use validated questionnaires to assess misuse potential, as demonstrated in GP surveys .
Q. Data Presentation & Compliance
Q. How should researchers present conflicting data on this compound’s hygroscopicity in publications?
- Guidance : Use comparative tables to highlight environmental conditions (RH%, temperature) and analytical methods. Discuss limitations under "Results" and propose validation experiments in "Discussion" .
Q. What are the requirements for depositing this compound’s crystallographic data in public repositories?
Properties
CAS No. |
6059-47-8 |
|---|---|
Molecular Formula |
C18H21NO3 |
Molecular Weight |
299.4 g/mol |
IUPAC Name |
(4R,4aR,7S,7aR,12bS)-9-methoxy-3-methyl-2,4,4a,7,7a,13-hexahydro-1H-4,12-methanobenzofuro[3,2-e]isoquinolin-7-ol |
InChI |
InChI=1S/C18H21NO3/c1-19-8-7-18-11-4-5-13(20)17(18)22-16-14(21-2)6-3-10(15(16)18)9-12(11)19/h3-6,11-13,17,20H,7-9H2,1-2H3/t11-,12+,13-,17-,18-/m0/s1 |
InChI Key |
OROGSEYTTFOCAN-DNJOTXNNSA-N |
SMILES |
CN1CCC23C4C1CC5=C2C(=C(C=C5)OC)OC3C(C=C4)O.O |
Isomeric SMILES |
CN1CC[C@]23[C@@H]4[C@H]1CC5=C2C(=C(C=C5)OC)O[C@H]3[C@H](C=C4)O |
Canonical SMILES |
CN1CCC23C4C1CC5=C2C(=C(C=C5)OC)OC3C(C=C4)O |
boiling_point |
482 °F at 22 mmHg (sublimes) (NTP, 1992) BP: 250 °C at 22 mm Hg |
Color/Form |
Orthorhombic crystals from water, dilute alcohol, ether Colorless or white crystals or powde |
density |
1.32 at 68 °F (NTP, 1992) - Denser than water; will sink |
flash_point |
167 °F (NTP, 1992) |
melting_point |
309 to 313 °F (after drying at 176 °F) (NTP, 1992) 154-156 157.5 °C |
Key on ui other cas no. |
6059-47-8 |
physical_description |
Codeine appears as colorless to white crystalline solid or white powder. Sublimes at 284 °F. Odorless. Bitter taste. pH (saturated aqueous solution) 9.8. (NTP, 1992) Solid |
Pictograms |
Acute Toxic; Irritant; Health Hazard |
Related CAS |
41444-62-6 (PO4) |
solubility |
less than 1 mg/mL at 70 °F (NTP, 1992) soluble in water Soluble in water Slightly soluble in water Soluble in ethyl ether, benzene, chloroform, toluene; very soluble in ethanol; insoluble in petroleum ether Soluble in alcohol and chloroform 0.577 g/L |
Synonyms |
Ardinex Codeine Codeine Phosphate Isocodeine N Methylmorphine N-Methylmorphine |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















