Nystatin
Overview
Description
Nystatin is a polyene antifungal drug that exhibits broad-spectrum fungicidal and fungistatic activity against a variety of yeasts and fungi, most notably Candida species . It was discovered in 1950 by Rachel Fuller Brown and Elizabeth Lee Hazen and is produced by the bacterium Streptomyces noursei . This compound is used to treat cutaneous, mucocutaneous, and gastrointestinal mycotic infections .
Preparation Methods
Nystatin is typically extracted from the fermentation broth of Streptomyces noursei using a water-soluble organic solvent. The compound is then purified and crystallized . The industrial production of this compound involves a series of steps including fermentation, extraction, purification, and crystallization to achieve high-yield and high-purity this compound crystals .
Chemical Reactions Analysis
Nystatin undergoes various chemical reactions, including oxidation and reduction. The compound is amphoteric due to the presence of carboxyl and amino groups . It is practically insoluble in water but slightly soluble in polar organic solvents . The solubility of this compound increases in aqueous-organic systems, with the greatest solubility achieved in solutions containing 30-40% water .
Scientific Research Applications
Treatment of Oral Candidiasis
Nystatin is commonly used for treating oral candidiasis, especially in immunocompromised patients. A systematic review indicated that this compound pastilles demonstrated a higher clinical cure rate (87%) compared to this compound suspension (80%) in patients with denture stomatitis .
Efficacy Comparison Table:
| Treatment Type | Clinical Cure Rate (%) | Mycological Cure Rate (%) |
|---|---|---|
| This compound Pastilles | 87 | Not specified |
| This compound Suspension | 80 | Not specified |
| Fluconazole | 88 | Not specified |
Prophylaxis in Immunocompromised Patients
This compound has been investigated for prophylactic use in severely immunodepressed patients, such as those undergoing chemotherapy or organ transplantation. However, studies have shown that its efficacy is often inferior to fluconazole, with this compound demonstrating no significant advantage over placebo in preventing fungal colonization .
Prophylaxis Effectiveness Table:
| Study Group | This compound Efficacy | Fluconazole Efficacy | Placebo Efficacy |
|---|---|---|---|
| Acute leukemia patients | No significant difference | Superior | No significant difference |
| Solid cancer patients | No significant difference | Superior | No significant difference |
| Bone marrow transplant recipients | No significant difference | Superior | No significant difference |
Treatment of Cutaneous Infections
This compound is also effective against cutaneous candidiasis and other superficial fungal infections. In cases where patients exhibit hypersensitivity to fungi, this compound has shown superior efficacy compared to placebo in reducing localized and systemic symptoms .
Case Studies
-
Oral Candidiasis in Cancer Patients
A study involving hospitalized cancer patients found that the combination of this compound suspension and pastilles resulted in clinical and mycological cures in 87.5% and 66% of cases, respectively . This highlights this compound's effectiveness in vulnerable populations. -
Fungal Hypersensitivity
In a randomized trial with polysymptomatic patients suspected of having fungus hypersensitivity, this compound significantly reduced overall symptom scores compared to placebo (P < 0.003). This suggests potential benefits beyond traditional antifungal applications .
Safety and Side Effects
While this compound is generally well-tolerated, some common side effects include gastrointestinal discomfort and a poor taste associated with oral formulations. These adverse effects can impact patient adherence to treatment regimens .
Mechanism of Action
Nystatin exerts its antifungal effects by binding to ergosterol, a key component of fungal cell membranes . This binding causes the formation of pores in the membrane, leading to the leakage of potassium ions and other cellular contents, ultimately resulting in cell death . This compound’s mechanism of action is complex and depends on the presence and type of membrane sterols . The formation and stabilization of this compound-induced pores are influenced by the biophysical properties and lipid composition of the membrane .
Comparison with Similar Compounds
Nystatin is often compared with other antifungal agents such as fluconazole and amphotericin B. While this compound is effective for treating localized fungal infections, it is not suitable for systemic use due to its significant toxicity when administered parenterally . Fluconazole, on the other hand, is effective for systemic fungal infections but may have side effects such as headache, nausea, and abdominal pain . Amphotericin B is closely related to this compound in structure and has a broader antifungal activity, but it also has a higher toxicity profile . Other similar compounds include newly synthesized thiazole derivatives, which have shown promising antifungal activity against Candida species .
This compound’s unique ability to bind to ergosterol and disrupt fungal cell membranes makes it a valuable antifungal agent, particularly for treating localized infections .
Biological Activity
Nystatin is a polyene antifungal agent primarily used to treat fungal infections, particularly those caused by Candida species. Its biological activity is characterized by its mechanism of action, efficacy in clinical applications, and safety profile. This article delves into these aspects, supported by research findings, data tables, and case studies.
This compound exerts its antifungal effects through a unique mechanism involving the binding to ergosterol, a key component of fungal cell membranes. This interaction leads to the formation of pores in the membrane, disrupting its integrity and causing leakage of intracellular contents. The selectivity for fungal cells over mammalian cells is attributed to this compound's higher affinity for ergosterol compared to cholesterol found in human cells .
Key Findings on Mechanism
- Pore Formation : this compound forms membrane-spanning channels that increase permeability, allowing ions and small molecules to leak out, which disrupts essential cellular functions .
- Killing Kinetics : Studies show that this compound exhibits rapid killing kinetics against Candida albicans, with significant reductions in colony-forming units (CFUs) observed within hours at effective concentrations .
Efficacy in Clinical Applications
This compound is widely used for treating various fungal infections, particularly oral candidiasis and cutaneous candidiasis. Its formulations include suspensions and pastilles, each demonstrating varying degrees of efficacy.
Clinical Studies Overview
- Oral Candidiasis : A systematic review indicated that this compound pastilles were significantly more effective than placebo for denture stomatitis. However, this compound suspension showed inferior efficacy compared to fluconazole in treating oral candidiasis in vulnerable populations such as infants and HIV/AIDS patients .
- Cutaneous Candidiasis : Historical studies have shown this compound ointment to be effective against skin lesions caused by Candida, with significant clinical response rates compared to placebo .
Efficacy Data Table
| Formulation | Condition | Efficacy (%) | Comparison Drug |
|---|---|---|---|
| This compound Pastille | Denture Stomatitis | 79.6% - 87.5% | Placebo |
| This compound Suspension | Oral Candidiasis | 9% - 63.5% | Fluconazole |
| This compound Ointment | Cutaneous Candidiasis | 78% (clearing rate) | Placebo |
Safety Profile
This compound is generally considered safe with minimal systemic absorption when administered orally or topically. Common adverse effects include gastrointestinal disturbances and a poor taste associated with oral formulations . Notably, high concentrations can lead to cytotoxicity in mammalian cells; however, formulations like this compound-Intralipid have shown reduced toxicity while maintaining antifungal activity .
Case Studies
Several case studies illustrate the effectiveness of this compound in treating specific patient populations:
- Infants with Oral Thrush : In cases involving infants with oral thrush, this compound was administered as a swish-and-swallow treatment, showing significant clinical improvements by day four post-treatment .
- Cancer Patients : A study involving cancer patients demonstrated a clinical cure rate of 87.5% when using a combination of this compound suspension and pastilles for oral candidiasis .
Q & A
Basic Research Questions
Q. How can researchers design experiments to study Nystatin’s pore-forming activity in lipid membranes?
- Methodological Answer : Use giant unilamellar vesicles (GUVs) as model membranes to observe pore formation dynamics. Incorporate fluorescently labeled this compound and ion-sensitive probes (e.g., pH or calcium sensors) to track real-time interactions . Measure osmotic effects via changes in vesicle volume under controlled osmotic gradients. For reproducibility, document line tension and pressure differentials using numerical models (Supplementary File S1) .
Q. What experimental conditions optimize this compound’s antifungal activity in Candida bioassays?
- Methodological Answer : Prepare stock solutions in DMSO (>30.5 mg/mL) and warm to 37°C with sonication to ensure solubility . Use human buccal epithelial cells co-cultured with Candida species to mimic oral infections. Validate antifungal efficacy via inhibition zone assays, comparing gel formulations to commercial creams. Apply ANOVA with Tukey-Kramer post-tests for statistical rigor .
Q. How should this compound’s pharmacokinetics be assessed in non-absorbable formulations?
- Methodological Answer : Measure salivary concentrations post-administration (e.g., 400,000-unit lozenges) using HPLC. Since systemic absorption is negligible, focus on fecal excretion profiles to confirm lack of bioavailability . For topical applications, use Franz diffusion cells to quantify permeation through epithelial barriers .
Advanced Research Questions
Q. How can researchers resolve contradictions in this compound’s clinical efficacy data for immunocompromised patients?
- Methodological Answer : Conduct meta-analyses of randomized trials (e.g., Cochrane reviews) comparing this compound with fluconazole. Use random-effects models to account for heterogeneity in study populations (e.g., leukemia vs. transplant patients). Prioritize endpoints like fungal colonization (RR 0.85 vs. placebo) and invasive infection rates (RR 0.37 for fluconazole superiority) . Perform sensitivity analyses to exclude non-cancer cohorts if confounding exists .
Q. What strategies mitigate this compound’s low solubility and toxicity in systemic delivery?
- Methodological Answer : Develop nanotechnology-based carriers (e.g., liposomes) to enhance solubility and reduce nephrotoxicity. Characterize encapsulation efficiency using dynamic light scattering and in vitro release studies. Compare engineered derivatives like BSG005 for improved safety profiles . Validate stability under physiological conditions via accelerated degradation studies .
Q. How can mathematical modeling improve dose-response analysis for this compound’s antiviral applications?
- Methodological Answer : Define dose-dependent viral inhibition functions (e.g., ) to model SARS-CoV-2 suppression in gastrointestinal cells. Use EC50/EC90 calculations from dose-response curves (e.g., 62.5 µg/mL effective against Wuhan and British variants) . Apply Levene tests to ensure variance homogeneity in inhibition zone data .
Q. What experimental frameworks validate this compound’s synergy with other antifungals?
- Methodological Answer : Employ checkerboard assays to quantify fractional inhibitory concentration indices (FICI). Test combinations with azoles (e.g., fluconazole) against resistant Candida strains. Use time-kill curves to differentiate additive vs. synergistic effects. Ensure statistical power via a priori sample size calculations .
Q. Methodological and Analytical Considerations
Q. How should researchers address variability in this compound’s inhibition zone assays?
- Methodological Answer : Standardize agar diffusion protocols per NCCLS guidelines. Control variables like agar thickness (4 mm), inoculum density (1–5 × 10⁸ CFU/mL), and incubation time (24–48 hours). Use parallel-line symmetrical models for potency determination, validated via ANOVA and Pearson correlations (e.g., between permeation and inhibition) .
Q. What criteria ensure robust structural characterization of this compound derivatives?
- Methodological Answer : For novel compounds (e.g., this compound A3), combine NMR, mass spectrometry, and X-ray crystallography. Assign ionization constants (pK₁ = 5.72, pK₂ = 8.64) via potentiometric titration in methanol-water systems. Confirm zwitterionic behavior at the isoelectric point (pH 7.18) .
Q. How can researchers optimize in vitro models for this compound’s mechanism of action studies?
- Methodological Answer : Use polarized epithelial monolayers (e.g., Caco-2) to simulate intestinal barriers. Quantify membrane depolarization via transepithelial electrical resistance (TEER) measurements. Correlate with sterol-binding assays to confirm target engagement .
Q. Contradictions and Knowledge Gaps
- Key Contradiction : this compound’s prophylactic efficacy in immunocompromised patients remains disputed, with meta-analyses showing no mortality benefit over placebo (RR 0.85) and inferiority to fluconazole (RR 0.37) .
- Knowledge Gap : Limited data exist on this compound’s antiviral mechanisms beyond sterol binding. Multidisciplinary studies integrating virology, pharmacokinetics, and nanotechnology are needed .
Properties
IUPAC Name |
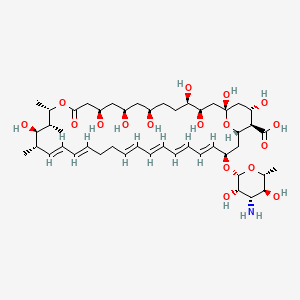
(1S,3R,4R,7R,9R,11R,15S,16R,17R,18S,19E,21E,25E,27E,29E,31E,33R,35S,36R,37S)-33-[(2R,3S,4S,5S,6R)-4-amino-3,5-dihydroxy-6-methyloxan-2-yl]oxy-1,3,4,7,9,11,17,37-octahydroxy-15,16,18-trimethyl-13-oxo-14,39-dioxabicyclo[33.3.1]nonatriaconta-19,21,25,27,29,31-hexaene-36-carboxylic acid | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C47H75NO17/c1-27-17-15-13-11-9-7-5-6-8-10-12-14-16-18-34(64-46-44(58)41(48)43(57)30(4)63-46)24-38-40(45(59)60)37(54)26-47(61,65-38)25-36(53)35(52)20-19-31(49)21-32(50)22-33(51)23-39(55)62-29(3)28(2)42(27)56/h5-6,8,10-18,27-38,40-44,46,49-54,56-58,61H,7,9,19-26,48H2,1-4H3,(H,59,60)/b6-5+,10-8+,13-11+,14-12+,17-15+,18-16+/t27-,28-,29-,30+,31+,32+,33+,34-,35+,36+,37-,38-,40+,41-,42+,43+,44-,46-,47+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
VQOXZBDYSJBXMA-NQTDYLQESA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1C=CC=CCCC=CC=CC=CC=CC(CC2C(C(CC(O2)(CC(C(CCC(CC(CC(CC(=O)OC(C(C1O)C)C)O)O)O)O)O)O)O)C(=O)O)OC3C(C(C(C(O3)C)O)N)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@H]1/C=C/C=C/CC/C=C/C=C/C=C/C=C/[C@@H](C[C@H]2[C@@H]([C@H](C[C@](O2)(C[C@H]([C@@H](CC[C@H](C[C@H](C[C@H](CC(=O)O[C@H]([C@@H]([C@@H]1O)C)C)O)O)O)O)O)O)O)C(=O)O)O[C@H]3[C@H]([C@H]([C@@H]([C@H](O3)C)O)N)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C47H75NO17 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID80872323 | |
| Record name | (7R,10R)-8,9-Dideoxy-28,29-dihydro-7,10-dihydroxyamphotericin B | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80872323 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
926.1 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
MW: 926.12 /Form not specified/, Mg/ml at about 28 °C: methanol 11.2, ethanol 1.2, chloroform 0.48, carbon tetrachloride 1.23, benzene 0.28, toluene 0.285, acetone 0.390, ethyl acetate 0.75, ethylene glycol 8.75, Insol in ether, In water, 3.60X10+2 mg/L at 24 °C | |
| Record name | NYSTATIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3138 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
Nystatin exerts its antifungal activity by binding to sterols in the fungal cell membrane. The drug is not active against organisms (e.g., bacteria) that do not contain sterols in their cell membrane. As a result of this binding, the membrane is no longer able to function as a selective barrier, and potassium and other cellular constituents are lost., ... /Antimicrobial/ agents that act directly on the cell membrane of the microorganism, affecting permeability and leading to leakage of intracellular compounds; these include ... the polyene antifungal agents nystatin ... which bind to cell-wall sterols ... | |
| Record name | NYSTATIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3138 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Light yellow powder, Yellow to tan powder | |
CAS No. |
34786-70-4, 1400-61-9 | |
| Record name | Nystatin A1 | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=34786-70-4 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Nystatin A1 | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0034786704 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | (7R,10R)-8,9-Dideoxy-28,29-dihydro-7,10-dihydroxyamphotericin B | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80872323 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Nystatin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.014.317 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | NYSTATIN A1 | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/W1LX4T91WI | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | NYSTATIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3138 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Melting Point |
Gradually decomp above 160 °C without melting by 250 °C | |
| Record name | NYSTATIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3138 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















