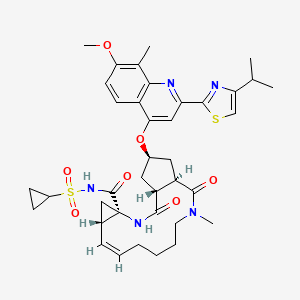
Simeprevir
Overview
Description
TMC435, also known as simeprevir, is a potent, once-daily, noncovalent, oral hepatitis C virus NS3/4A protease inhibitor. It is used in combination with pegylated interferon and ribavirin for the treatment of chronic hepatitis C virus genotype 1 infection. This compound has been approved for use in several countries, including Japan, Canada, the United States, Russia, the European Union, Mexico, and Australia .
Preparation Methods
The preparation of TMC435 involves several synthetic routes and reaction conditions. One of the key intermediates in the synthesis is a bicyclic lactone amide. The preparation method for this intermediate involves multiple steps, including the formation of a macrocyclic compound . The industrial production of TMC435 typically involves the crystallization of the compound to obtain a stable and pure form suitable for pharmaceutical use .
Chemical Reactions Analysis
TMC435 undergoes various chemical reactions, including oxidation, reduction, and substitution. Common reagents used in these reactions include organic solvents, acids, and bases. The major products formed from these reactions are typically derivatives of the original compound, which may have different pharmacological properties .
Scientific Research Applications
Clinical Efficacy in Hepatitis C Treatment
Simeprevir has demonstrated significant efficacy in treating chronic hepatitis C, particularly in genotype 1 infections. The following key studies highlight its clinical applications:
- PROMISE Study : In this pivotal Phase 3 study, this compound was administered to treatment-experienced patients, resulting in a sustained virologic response (SVR12) rate of 79%, compared to only 37% in the placebo group. This study established this compound's effectiveness when combined with pegylated interferon and ribavirin .
- QUEST Studies : A pooled analysis from the QUEST-1 and QUEST-2 trials showed that 80% of treatment-naïve patients achieved SVR12 when treated with this compound plus pegylated interferon and ribavirin. This was a significant improvement over historical controls .
- This compound Plus Sofosbuvir : A study evaluating this compound combined with sofosbuvir for 12 weeks reported superior SVR12 rates compared to historical controls for both treatment-naïve and treatment-experienced patients .
Patient Subpopulations
This compound's effectiveness varies among different patient demographics and conditions:
- Cirrhosis Patients : In patients with cirrhosis, SVR12 rates were reported at 83% for those treated with this compound plus sofosbuvir. This indicates that even patients with advanced liver disease can benefit significantly from this combination therapy .
- IL28B Genotype Impact : The efficacy of this compound also correlates with the IL28B genotype. In the PROMISE study, SVR12 rates varied significantly based on genotype, highlighting the importance of genetic factors in treatment outcomes .
Safety Profile
This compound is generally well-tolerated, with manageable side effects. The PROMISE study reported on-treatment failure rates of 3% and relapse rates of 19% among patients treated with this compound, which were significantly lower than those observed in the placebo group . Adverse reactions are relatively infrequent and do not exacerbate anemia, a common side effect associated with other treatments like pegylated interferon .
Future Research Directions
Ongoing studies continue to explore the potential applications of this compound beyond its current indications:
- Combination Therapies : Research is being conducted on the use of this compound in combination with newer antiviral agents to enhance efficacy and reduce treatment duration for various HCV genotypes .
- Drug Repositioning : Preliminary investigations suggest that this compound may have applications beyond hepatitis C, including potential effects on other viral infections or conditions due to its mechanism of action targeting viral proteases .
Summary Table of Clinical Trials Involving this compound
| Study Name | Patient Population | Treatment Duration | SVR12 Rate (%) | Key Findings |
|---|---|---|---|---|
| PROMISE | Treatment-experienced | 12 weeks | 79 | Significant improvement over placebo |
| QUEST-1 & QUEST-2 | Treatment-naïve | 12 weeks | 80 | High efficacy in naïve patients |
| OPTIMIST | Patients with cirrhosis | 8-12 weeks | 83 | Effective even in advanced liver disease |
| This compound + Sofosbuvir | Mixed population | 12 weeks | Variable | Superior SVR12 rates compared to historical control |
Mechanism of Action
TMC435 exerts its effects by inhibiting the NS3/4A serine protease of the hepatitis C virus. This protease is essential for the replication of the virus. By inhibiting this enzyme, TMC435 prevents the virus from replicating and spreading within the host. The molecular targets of TMC435 include specific residues within the NS3/4A protease, which are critical for its enzymatic activity .
Comparison with Similar Compounds
TMC435 is similar to other hepatitis C virus NS3/4A protease inhibitors, such as telaprevir and boceprevir. TMC435 has several unique features that distinguish it from these compounds. For example, TMC435 has a more favorable resistance profile and is effective against a broader range of hepatitis C virus genotypes. Additionally, TMC435 has been shown to have a better safety and tolerability profile compared to other protease inhibitors .
Similar Compounds
- Telaprevir
- Boceprevir
- Grazoprevir
- Paritaprevir
Q & A
Basic Research Questions
Q. How should in vitro studies be designed to evaluate Simeprevir’s antiviral activity against HCV genotypes?
- Methodological Guidance : Use Huh7-Luc cells to measure dose-dependent inhibition of HCV replication. Calculate EC₅₀ and EC₉₀ values via dose-response curves. For genotype 1a, include Q80K polymorphism testing due to its impact on this compound resistance . Standardize assays using chimeric replicons with patient-derived NS3 sequences to model clinical resistance .
Q. What are the key parameters for optimizing this compound quantification in stability-indicating HPLC methods?
- Methodological Guidance : Employ a Quality by Design (QbD) approach for reverse-phase HPLC. Validate parameters such as mobile phase composition (e.g., acetonitrile-phosphate buffer), column type (C18), and detection wavelength (210 nm). Ensure linearity (5–50 µg/mL), precision (RSD <2%), and robustness against degradation products (e.g., acidic/oxidative conditions) .
Q. How do response-guided therapy (RGT) criteria influence this compound trial design?
- Methodological Guidance : In phase III trials (e.g., PROMISE study), apply RGT to shorten treatment duration. Patients achieving rapid virologic response (RVR; HCV RNA undetectable at week 4) may reduce therapy from 48 to 24 weeks. Include SVR12/SVR24 as primary endpoints and stratify by baseline viral load and fibrosis stage .
Advanced Research Questions
Q. How can resistance-associated substitutions (RAS) in NS3/4A be analyzed after this compound treatment failure?
- Methodological Guidance : Perform direct sequencing of NS3 protease domains from clinical isolates. Focus on mutations like D168V/E (conferring high-level resistance) and Q80K (genotype 1a). Use phenotypic assays with chimeric replicons to quantify fold changes in EC₅₀. Correlate RAS with virologic relapse rates in non-SVR cohorts .
Q. What pharmacokinetic interactions occur between this compound and immunosuppressants in transplant patients?
- Methodological Guidance : Conduct phase IIa studies in HCV-infected transplant recipients co-administered cyclosporine or tacrolimus. Monitor this compound plasma concentrations via LC-MS/MS. Cyclosporine inhibits OATP1B1/P-gp/CYP3A, increasing this compound AUC by 5.8-fold; avoid coadministration. Tacrolimus interactions are clinically insignificant .
Q. How do IL28B polymorphisms and fibrosis stages affect this compound’s SVR rates in genotype 1 patients?
- Methodological Guidance : Stratify patients by IL28B alleles (CC vs. non-CC) and METAVIR fibrosis scores (F0–F2 vs. F3–F4). In treatment-naïve CC patients, SVR rates reach 95% with this compound + PR vs. 82% with Sofosbuvir + PR. For F3–F4 fibrosis, this compound achieves 69% SVR vs. 62% with Telaprevir .
Q. Data Analysis & Contradiction Resolution
Q. How should conflicting SVR outcomes in this compound trials be reconciled (e.g., GALAXY vs. retrospective studies)?
- Methodological Guidance : Perform network meta-analyses comparing trial designs. For example, GALAXY (prospective) reported 100% SVR12 with this compound + Sofosbuvir, while retrospective studies (n=151) noted 88% SVR due to allograft cirrhosis. Adjust for confounders like ribavirin use, treatment duration, and patient heterogeneity .
Q. What statistical models predict this compound’s impact on healthcare resource utilization?
- Methodological Guidance : Use two-part multivariable models with phase III trial data. Logistic regression can link adverse events (e.g., hyperbilirubinemia) to hospital admissions. In UK cost analyses, shorter this compound regimens reduce outpatient consultations by 30% compared to 48-week PR .
Q. Experimental Design Considerations
Q. What strategies minimize bias in this compound vs. first-generation protease inhibitor trials?
- Methodological Guidance : Use randomized, double-blind designs with active comparators (e.g., Telaprevir). Control for baseline variables like HCV subtype (1a vs. 1b) and prior treatment history. Include blinded endpoint adjudication committees to reduce SVR assessment bias .
Q. How can in vitro resistance profiles inform clinical trial inclusion criteria?
- Methodological Guidance : Exclude genotype 1a patients with Q80K polymorphisms (prevalence ~30% in US/EU) due to 11-fold reduced this compound susceptibility. For genotype 1b, screen for L31 RAS, which reduces SVR by 50% in non-responders .
Q. Tables for Key Findings
| Parameter | This compound + PR | Sofosbuvir + PR | Telaprevir + PR |
|---|---|---|---|
| SVR in IL28B CC (naïve) | 95% | 98% | 71% |
| SVR in F3–F4 fibrosis | 69% | 85% | 62% |
| Treatment Duration (weeks) | 24 (RGT) | 12 | 48 |
| Resistance Mutation | Fold Change (EC₅₀) | Clinical Impact |
|---|---|---|
| Q80K (1a) | 11x | Avoid this compound use |
| D168V/E (1a/1b) | ≥50x | High-level resistance |
| L31M (1b) | 5x | Moderate relapse risk |
Properties
CAS No. |
923604-59-5 |
|---|---|
Molecular Formula |
C38H47N5O7S2 |
Molecular Weight |
749.9 g/mol |
IUPAC Name |
(4S)-N-cyclopropylsulfonyl-17-[7-methoxy-8-methyl-2-(4-propan-2-yl-1,3-thiazol-2-yl)quinolin-4-yl]oxy-13-methyl-2,14-dioxo-3,13-diazatricyclo[13.3.0.04,6]octadec-7-ene-4-carboxamide |
InChI |
InChI=1S/C38H47N5O7S2/c1-21(2)30-20-51-35(40-30)29-18-32(26-13-14-31(49-5)22(3)33(26)39-29)50-24-16-27-28(17-24)36(45)43(4)15-9-7-6-8-10-23-19-38(23,41-34(27)44)37(46)42-52(47,48)25-11-12-25/h8,10,13-14,18,20-21,23-25,27-28H,6-7,9,11-12,15-17,19H2,1-5H3,(H,41,44)(H,42,46)/t23?,24?,27?,28?,38-/m0/s1 |
InChI Key |
JTZZSQYMACOLNN-ZMKZURCUSA-N |
SMILES |
CC1=C(C=CC2=C1N=C(C=C2OC3CC4C(C3)C(=O)N(CCCCC=CC5CC5(NC4=O)C(=O)NS(=O)(=O)C6CC6)C)C7=NC(=CS7)C(C)C)OC |
Isomeric SMILES |
CC1=C(C=CC2=C1N=C(C=C2OC3CC4C(C3)C(=O)N(CCCCC=CC5C[C@@]5(NC4=O)C(=O)NS(=O)(=O)C6CC6)C)C7=NC(=CS7)C(C)C)OC |
Canonical SMILES |
CC1=C(C=CC2=C1N=C(C=C2OC3CC4C(C3)C(=O)N(CCCCC=CC5CC5(NC4=O)C(=O)NS(=O)(=O)C6CC6)C)C7=NC(=CS7)C(C)C)OC |
Color/Form |
White to almost white powder |
Pictograms |
Irritant; Environmental Hazard |
solubility |
Insoluble Practically insoluble in water over a wide pH range Practically insoluble in propylene glycol; very slightly soluble in ethanol; slightly soluble in acetone. Soluble in dichloromethane; freely soluble in some organic solvents |
Synonyms |
435, TMC 435350, TMC N-(17-(2-(4-isopropylthiazole-2-yl)-7-methoxy-8-methylquinolin-4-yloxy)-13-methyl-2,14-dioxo-3,13-diazatricyclo(13.3.0.04,6)octadec-7-ene-4-carbonyl)(cyclopropyl)sulfonamide Olysio simeprevir TMC 435 TMC 435350 TMC-435 TMC-435350 TMC435 TMC435350 |
vapor_pressure |
5.9X10-27 mm Hg at 25 °C (est) |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















