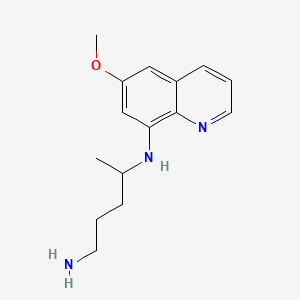
Primaquine
Overview
Description
Primaquine is a medication primarily used to treat and prevent malaria, specifically targeting the dormant liver forms of the parasite Plasmodium vivax and Plasmodium ovale . It is also used in combination with other medications to treat Pneumocystis pneumonia . This compound was first synthesized in 1946 and is included in the World Health Organization’s List of Essential Medicines .
Preparation Methods
Synthetic Routes and Reaction Conditions
Primaquine is synthesized through a multi-step process starting from 8-aminoquinoline. . The reaction conditions typically involve the use of strong bases and organic solvents to facilitate the substitution reactions.
Industrial Production Methods
Industrial production of this compound involves large-scale synthesis using similar reaction conditions as in the laboratory synthesis but optimized for higher yields and purity. The process includes rigorous purification steps to ensure the final product meets pharmaceutical standards .
Chemical Reactions Analysis
Types of Reactions
Primaquine undergoes various chemical reactions, including:
Reduction: The compound can also undergo reduction reactions, particularly in the presence of reducing agents.
Substitution: This compound can participate in substitution reactions, especially at the amino and methoxy groups.
Common Reagents and Conditions
Common reagents used in the reactions of this compound include oxidizing agents like hydrogen peroxide, reducing agents such as sodium borohydride, and various organic solvents . The reactions are typically carried out under controlled temperature and pH conditions to ensure the desired products are formed.
Major Products Formed
The major products formed from the reactions of this compound include its oxidized and reduced forms, as well as various substituted derivatives . These products are often studied for their potential pharmacological activities.
Scientific Research Applications
Clinical Applications
-
Radical Cure of P. vivax and P. ovale :
- Primaquine is essential for preventing relapses in patients with P. vivax and P. ovale malaria due to its ability to eliminate hypnozoites.
- A systematic review indicated that this compound significantly reduces the risk of relapse compared to placebo or no treatment over extended follow-up periods .
-
Mass Drug Administration (MDA) :
- Recent studies have evaluated this compound's role in mass drug administration strategies aimed at eliminating P. vivax malaria in low-endemicity areas.
- A proof-of-concept study demonstrated the effectiveness and safety of this compound MDA (pMDA), suggesting its potential as a public health intervention .
- Combination Therapy :
Safety and Pharmacogenomics
G6PD Deficiency :
- The most significant safety concern associated with this compound is hemolytic toxicity in individuals with G6PD deficiency, affecting approximately 8% of people in malaria-endemic regions.
- Recent meta-analyses have highlighted the importance of screening for G6PD deficiency prior to this compound treatment to mitigate risks .
Dosing Strategies :
- Research indicates that higher doses of this compound can be effective but must be carefully managed to avoid adverse effects. A multicenter trial found that high-dose short-course this compound was well tolerated in patients with adequate G6PD activity, reducing subsequent P. vivax episodes significantly .
Case Study 1: Efficacy in G6PD Normal Patients
A multicenter trial involving patients with normal G6PD levels demonstrated that a total dose of 7 mg/kg over seven days effectively reduced the risk of subsequent P. vivax parasitemia by fivefold compared to standard treatments . This study underscores the importance of individualized treatment strategies based on genetic screening.
Case Study 2: Implementation of pMDA
In a tropical country, a study assessing pMDA with this compound showed promising results in reducing malaria incidence in low-endemic areas. The acceptability and safety profile were favorable, suggesting that pMDA could be a viable strategy for malaria elimination efforts .
Mechanism of Action
The exact mechanism of action of primaquine is not entirely understood. it is believed to interfere with the energy production in the malaria parasite by targeting its mitochondria . This disruption leads to the generation of reactive oxygen species, which damage the parasite’s cellular components and ultimately kill it . This compound may also bind to and alter the properties of protozoal DNA .
Comparison with Similar Compounds
Primaquine belongs to the class of 8-aminoquinoline compounds. Similar compounds include:
Tafenoquine: Like this compound, tafenoquine is used to treat and prevent malaria.
Chloroquine: Although not an 8-aminoquinoline, chloroquine is another antimalarial drug that targets the blood stages of the parasite.
This compound’s uniqueness lies in its ability to eliminate the dormant liver forms of malaria parasites, making it essential for preventing relapses .
Biological Activity
Primaquine is an 8-aminoquinoline compound primarily used in the treatment of malaria, particularly effective against the liver stages of Plasmodium vivax and Plasmodium ovale. Despite its long history of use, the exact mechanisms underlying its biological activity remain partially elucidated. This article explores the biological activity of this compound, its pharmacokinetics, mechanisms of action, and relevant clinical studies.
The biological activity of this compound is attributed to several mechanisms:
-
Metabolism and Active Metabolites :
- This compound is metabolized in the liver to several active metabolites, including carboxythis compound (CPQ) and hydroxylated this compound metabolites (OH-PQm). The efficacy against liver stages is significantly influenced by the host's CYP2D6 enzyme status, which is polymorphic in humans. Inhibition of CYP2D6 can lead to accumulation of this compound and reduced levels of active metabolites .
-
Oxidative Stress Induction :
- One proposed mechanism is the generation of reactive oxygen species (ROS) that induce oxidative stress within the malaria parasites. This oxidative damage disrupts mitochondrial function, leading to parasite death . The quinoline core structure of this compound is believed to play a crucial role in this redox cycling process .
- Interference with Electron Transport :
- DNA Interaction :
Pharmacokinetics
This compound exhibits rapid absorption following oral administration, with peak plasma concentrations typically occurring around 1.5 hours post-dose. The drug is extensively metabolized in the liver, with only about 1% excreted unchanged in urine. The primary route of excretion is fecal .
Clinical Efficacy
Recent studies have highlighted the efficacy and safety profile of this compound in various clinical settings:
- A multicenter trial demonstrated that high-dose short-course this compound significantly reduced the risk of subsequent P. vivax parasitemia by five-fold in patients with adequate G6PD enzyme activity (70% or greater). This suggests that universal radical cure strategies using this compound could offer substantial public health benefits .
- Another study emphasized that while this compound has modest antimalarial efficacy against gametocytes, its potency can be enhanced significantly (up to 1000-fold) in the presence of cytochrome P450 NADPH:oxidoreductase from liver tissues .
Case Studies
Several case studies illustrate the practical application and effectiveness of this compound:
- Case Study 1 : In a cohort of patients treated for P. vivax malaria, those receiving this compound alongside chloroquine showed a marked decrease in relapse rates compared to those treated with chloroquine alone.
- Case Study 2 : A study involving patients with G6PD deficiency highlighted the importance of monitoring enzyme levels prior to this compound administration to prevent hemolytic anemia while still achieving effective malaria treatment outcomes.
Summary Table: Key Attributes of this compound
| Attribute | Details |
|---|---|
| Chemical Class | 8-aminoquinoline |
| Primary Use | Treatment of P. vivax and P. ovale malaria |
| Mechanisms | ROS generation, mitochondrial interference, DNA binding |
| Metabolism | Hepatic; active metabolites include CPQ and OH-PQm |
| Pharmacokinetics | Rapid absorption; peak concentration at ~1.5h; fecal excretion |
| Clinical Efficacy | Reduces relapse rates; effective against liver stages |
Q & A
Basic Research Questions
Q. What are the standard in vitro protocols for assessing Primaquine’s efficacy against Plasmodium strains, and how can researchers ensure reproducibility?
To evaluate this compound’s antimalarial activity, use standardized in vitro assays such as the SYBR Green I fluorescence-based method for quantifying parasite growth inhibition. Key parameters include:
- Cell lines : Utilize synchronized cultures of Plasmodium falciparum (e.g., 3D7 or Dd2 strains).
- Drug concentrations : Test a range (e.g., 0.1–100 µM) with triplicate wells for statistical robustness.
- Controls : Include chloroquine as a positive control and solvent-only wells as negative controls.
- Incubation time : 72 hours under low oxygen conditions (5% O₂) to mimic intra-erythrocytic environments.
For reproducibility, document all reagents (e.g., batch numbers), equipment calibration data, and environmental conditions (temperature, humidity) .
Q. How should researchers design preclinical studies to assess this compound’s hemolytic toxicity in G6PD-deficient models?
Adopt the PICOT framework (Population, Intervention, Comparison, Outcome, Time):
- Population : Use murine models with humanized G6PD mutations (e.g., G6PD A− variant).
- Intervention : Administer this compound at clinically relevant doses (e.g., 0.5–2 mg/kg/day).
- Comparison : Compare hematological parameters (e.g., hemoglobin decline, reticulocyte count) between G6PD-deficient and wild-type cohorts.
- Outcome : Quantify hemolysis via plasma free hemoglobin and haptoglobin levels.
- Time : Monitor for 7–14 days post-treatment.
Include detailed protocols for animal husbandry and ethical compliance in supplementary materials .
Q. What statistical methods are appropriate for analyzing this compound’s dose-response relationships in clinical trials?
Use non-linear regression models (e.g., log-logistic or Emax models) to calculate EC₅₀ values. For time-to-event data (e.g., parasite clearance), apply Kaplan-Meier survival analysis with Cox proportional hazards regression to adjust for covariates like baseline parasitemia and patient age. Report confidence intervals and effect sizes to enhance interpretability .
Advanced Research Questions
Q. How can researchers resolve contradictions in pharmacokinetic data for this compound across different patient populations?
Address variability by:
- Population stratification : Segment data by genetic polymorphisms (e.g., CYP2D6 metabolizer status) or comorbidities (e.g., hepatic impairment).
- Bioanalytical validation : Use LC-MS/MS to quantify this compound and its metabolites (e.g., carboxythis compound) with stringent sensitivity thresholds (LOQ ≤ 1 ng/mL).
- Modeling : Apply physiologically based pharmacokinetic (PBPK) models to simulate interindividual variability in drug absorption and clearance.
Publish raw datasets and modeling code to enable independent verification .
Q. What methodologies are optimal for investigating this compound’s hypnozoitocidal mechanisms in Plasmodium vivax liver stages?
Combine transcriptomic profiling (single-cell RNA-seq of infected hepatocytes) and high-content imaging to map this compound’s effects on hypnozoite dormancy. Key steps:
Primary hepatocyte culture : Use cryopreserved human hepatocytes infected with P. vivax sporozoites.
Drug exposure : Treat with this compound (1–10 µM) for 48 hours.
Endpoint analysis : Quantify hypnozoite viability via Plasmodium HSP70 immunofluorescence.
Validate findings with CRISPR-Cas9 knockouts of putative drug targets (e.g., mitochondrial enzymes) .
Q. How can researchers optimize this compound combination therapies to reduce relapse rates without exacerbating toxicity?
Implement a fractional factorial design to test drug combinations (e.g., this compound + tafenoquine or chloroquine). Variables include:
- Dose ratios : this compound (7.5–30 mg) paired with partner drugs at sub-therapeutic to therapeutic levels.
- Outcome metrics : Relapse rate (PCR-confirmed), hemoglobin drop, and adverse event frequency.
Use Bayesian adaptive trials to dynamically adjust dosing based on interim safety/efficacy data. Publish full trial protocols and adverse event logs to support meta-analyses .
Methodological Notes
- Data Integrity : Adhere to NIH guidelines for preclinical reporting, including raw data deposition in repositories like Dryad or Zenodo .
- Reproducibility : Provide step-by-step protocols for in vitro and in vivo assays, including reagent catalog numbers and software settings (e.g., ImageJ macros for parasite quantification) .
- Ethical Compliance : Disclose IRB/IACUC approval numbers and patient/mouse strain sourcing details .
Properties
IUPAC Name |
4-N-(6-methoxyquinolin-8-yl)pentane-1,4-diamine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C15H21N3O/c1-11(5-3-7-16)18-14-10-13(19-2)9-12-6-4-8-17-15(12)14/h4,6,8-11,18H,3,5,7,16H2,1-2H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
INDBQLZJXZLFIT-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(CCCN)NC1=C2C(=CC(=C1)OC)C=CC=N2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C15H21N3O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
63-45-6 (1:2 PO4) | |
| Record name | Primaquine [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000090346 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID8023509 | |
| Record name | Primaquine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8023509 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
259.35 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Primaquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015219 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Boiling Point |
175-179 °C | |
| Record name | Primaquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01087 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PRIMAQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6516 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Solubility |
5.64e-02 g/L | |
| Record name | Primaquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015219 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Primaquine's mechanism of action is not well understood. It may be acting by generating reactive oxygen species or by interfering with the electron transport in the parasite. Also, although its mechanism of action is unclear, primaquine may bind to and alter the properties of protozoal DNA., The precise mechanism of action has not been determined, but may be based on primaquine's ability to bind to and alter the properties of DNA. Primaquine is highly active against the exoeryhrocytic stages of plasmodium vivax and plasmodium ovale and against the primary exoerythrocytic stages of plasmodium falciparum. It is also highly active against the sexual forms of (gametocytes) plasmodia, especially P. falciparum, disrupting transmission of the disease by eliminating the reservoir from which the mosquito carrier is infected., /Primaquine/ disrupts the parasitic mitochondria, thereby interrupting metabolic processes requiring energy., ... /Primaquine is one/ of /aromatic amine-containing/ xenobiotics ... capable to inducing oxidative injury in erythrocytes. These agents appear to potentiate the normal redox reactions and are capable of overwhelming the usual protective mechanisms. The interaction between these xenobiotics and hemoglobin leads to the formation of free radicals that denature critical proteins, including hemoglobin, thiol-dependent enzymes, and components of the erythrocyte membrane ... Oxidative denaturation of the globin chain decreases its affinity for the heme group, which may dissociate from the globin chain during oxidative injury ... The generation of free radicals may also lead to peroxidation of membrane lipids. This may affect the deformability of the erythrocyte and the permeability of the membrane to potassium. The alteration of the Na(+)/K(+) gradient is ... potentially lethal to the affected erythrocyte. Oxidative injury also impairs the metabolic machinery of the erythrocyte, resulting in a decrease in the concentration of ATP. Damage to the membrane can also permit leakage of denatured hemoglobin from the cell. Such free denatured hemoglobin can be toxic on its own. Free hemoglobin may irreversibly bind nitric oxide, resulting in vasoconstriction. Released hemoglobin may form nephrotoxic hemoglobin dimers, leading to kidney damage. /Oxidative hemolysis/ | |
| Record name | Primaquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01087 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PRIMAQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6516 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Viscous liquid | |
CAS No. |
90-34-6 | |
| Record name | Primaquine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=90-34-6 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Primaquine [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000090346 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Primaquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01087 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PRIMAQUINE | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=27296 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Primaquine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8023509 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Primaquine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.001.807 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PRIMAQUINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/MVR3634GX1 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | PRIMAQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6516 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Primaquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015219 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
< 25 °C | |
| Record name | Primaquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01087 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Primaquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015219 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods
Procedure details




Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













