Cladribine
Overview
Description
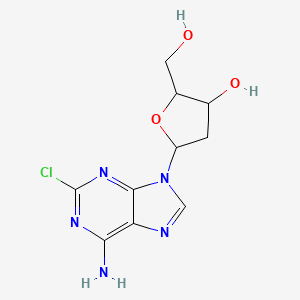
Cladribine, also known as 2-chlorodeoxyadenosine, is a synthetic purine nucleoside analog. It is primarily used in the treatment of hairy cell leukemia and multiple sclerosis. This compound mimics the nucleoside deoxyadenosine but is resistant to breakdown by the enzyme adenosine deaminase, allowing it to accumulate in targeted cells and interfere with DNA synthesis .
Preparation Methods
Synthetic Routes and Reaction Conditions: Cladribine can be synthesized through the direct coupling of O-protected 2-deoxy-ribofuranose with silylated 2-chloroadenine. This is followed by deprotection of the resultant protected nucleoside in a separate step and then a purification step . The process involves dissolving crude this compound in a protic solvent in the presence of a base to form a solution. This solution is maintained at an elevated temperature until the amount of protected or partially protected nucleoside impurities is reduced. The solution is then cooled to form and isolate this compound crystals .
Industrial Production Methods: The industrial production of this compound involves similar synthetic routes but on a larger scale. The process ensures high purity and yield, making it suitable for pharmaceutical applications .
Chemical Reactions Analysis
Types of Reactions: Cladribine undergoes various chemical reactions, including phosphorylation, dephosphorylation, and incorporation into DNA. It is relatively resistant to oxidation and reduction due to its stable structure .
Common Reagents and Conditions:
Phosphorylation: This reaction is catalyzed by the enzyme deoxycytidine kinase, converting this compound into its active triphosphorylated form.
Dephosphorylation: This reaction is mediated by 5’-nucleotidase, which can deactivate this compound in certain cell types.
Major Products: The major product of this compound’s phosphorylation is 2-chlorodeoxyadenosine triphosphate, which is incorporated into DNA and triggers apoptosis .
Scientific Research Applications
Cladribine is a synthetic deoxyadenosine analog prodrug with properties of selective lymphocyte suppression . It is used to treat multiple sclerosis (MS) and hairy cell leukemia . this compound selectively targets both resting and dividing lymphocytes and may be able to destroy the activated lymphocytes that induce CNS demyelination, potentially stabilizing or improving chronic MS .
Scientific Research Applications
This compound has been studied and applied in the treatment of multiple sclerosis (MS), particularly in relapsing forms, and in hairy cell leukemia. Research has explored its efficacy, safety, and mechanisms of action in these contexts [1, 6, 10].
Multiple Sclerosis (MS)
This compound tablets are approved for the treatment of relapsing forms of MS, including relapsing-remitting disease and active secondary progressive disease, in adults . The drug's use is generally recommended for patients who have had an inadequate response to, or are unable to tolerate, an alternate drug indicated for MS .
Efficacy:
- This compound has demonstrated clinically and statistically significant benefits in patients across the relapsing MS (RMS) spectrum, whether in early or late stages, and in treatment-naive or experienced patients .
- A study showed that this compound, when added to interferon-beta (IFN-β) therapy, reduced relapses and MRI activity in patients with active relapsing MS despite IFN-β treatment .
- Compared to placebo, this compound treatment was associated with reduced brain atrophy and a reduced risk of disability progression over 2 years .
- Data from the CLARITY Extension study indicated that long-term dosing did not increase efficacy, but later treatment was associated with a shorter time to first relapse. Patients who received this compound in the CLARITY trial but placebo in the CLARITY Extension maintained a durable clinical response, with the proportion free from relapses and disease progression remaining unchanged .
- This compound is more effective when placed early in the treatment algorithm .
Safety and Tolerability:
- Studies have evaluated the safety and tolerability of this compound tablets added to IFN-β therapy, focusing on adverse events (AEs) and laboratory toxicities, including infections, neoplasms, and hematologic and liver function parameters .
- Real-world data confirms the effectiveness and safety of this compound, with the majority of patients achieving no evidence of disease activity (NEDA-3) at 24 months .
Mechanism of Action:
- This compound is a synthetic deoxyadenosine analog prodrug that is activated through phosphorylation by deoxycytidine kinase, accumulating to cytotoxic levels in lymphocytes. This results in targeted and sustained reductions of T and B lymphocytes implicated in MS pathogenesis .
Hairy Cell Leukemia
Intravenous this compound is indicated for the treatment of active hairy cell leukemia, as defined by clinically significant anemia, neutropenia, thrombocytopenia, or disease-related symptoms . this compound is a purine antimetabolite .
Additional Considerations
- The World Health Organization (WHO) includes this compound on its list of essential medicines for the treatment of multiple sclerosis, recognizing its clinical benefit, safety, and suitability for use in different patient populations .
- This compound's mechanism of action involves selectively targeting lymphocytes, which play a key role in the pathogenesis of MS . By reducing the number of these cells, this compound can reduce the inflammation and demyelination that characterize MS .
- The approved treatment regimen involves two short courses of this compound tablets across two consecutive years (cumulative dose 3.5 mg/kg), followed by two years without treatment .
Mechanism of Action
Cladribine exerts its effects by mimicking deoxyadenosine and being incorporated into DNA. Once inside the cell, it is phosphorylated by deoxycytidine kinase to produce 2-chlorodeoxyadenosine triphosphate. This active form disrupts DNA synthesis and repair, leading to apoptosis. This compound selectively targets lymphocytes due to the high kinase:phosphatase ratio in these cells, making it effective in treating diseases involving abnormal lymphocyte activity .
Comparison with Similar Compounds
Pentostatin: Another purine analog used to treat hairy cell leukemia.
Fludarabine: A purine analog used in the treatment of chronic lymphocytic leukemia.
Uniqueness of Cladribine: this compound’s resistance to adenosine deaminase and its selective targeting of lymphocytes make it unique among purine analogs. Its ability to be administered orally for multiple sclerosis treatment also sets it apart from other similar compounds .
Biological Activity
Cladribine, a synthetic purine nucleoside analog, is primarily known for its application in treating multiple sclerosis (MS) and hairy cell leukemia (HCL). This article delves into the biological activity of this compound, focusing on its mechanisms of action, efficacy, safety profiles, and its impact on various immune cells.
This compound exerts its biological effects primarily through the induction of lymphocyte apoptosis. It is incorporated into DNA during the S-phase of the cell cycle, leading to single-strand breaks and ultimately triggering apoptotic pathways. The compound also influences mitochondrial function, which can lead to caspase-dependent and -independent apoptosis.
Key Mechanisms:
- Induction of Apoptosis : this compound causes DNA breaks that activate poly(ADP-ribose) polymerase (PARP), depleting intracellular NAD and ATP levels, resulting in cell death .
- Immunomodulatory Effects : Beyond inducing apoptosis in lymphocytes, this compound modulates the activity of dendritic cells and monocytes, which may enhance its therapeutic efficacy in autoimmune conditions like MS .
Clinical Efficacy in Multiple Sclerosis
This compound has shown significant efficacy in clinical trials for patients with highly active relapsing MS. Notably, the CLARITY trial demonstrated that this compound effectively reduces relapse rates and MRI activity compared to placebo.
Clinical Trial Findings:
- CLARITY Trial : Patients treated with this compound exhibited a 63% reduction in relapse rates compared to placebo .
- Long-term Efficacy : A follow-up study over eight years indicated that a substantial percentage of patients remained free from clinical or MRI activity post-treatment .
Safety Profile
The safety profile of this compound has been evaluated across multiple studies. While it is generally well-tolerated, there are concerns regarding potential adverse effects related to immunosuppression.
Adverse Events:
- Common treatment-emergent adverse events include infections and hematological toxicities. Monitoring lymphocyte counts is crucial due to the drug's immunosuppressive effects .
- In a systematic review encompassing 7244 MS patients, this compound was deemed safe with manageable side effects .
Case Studies and Observational Data
Several case studies highlight the real-world effectiveness and safety of this compound in managing MS. For instance:
- Real-World Data : A cohort study involving 187 patients showed that 88% were free from clinical activity after three years .
- Retreatment Efficacy : In a subset of patients receiving retreatment with this compound tablets, 83% had no clinical or MRI activity after four years .
Comparative Efficacy in Hematologic Malignancies
This compound's application extends beyond MS; it has shown remarkable efficacy in treating HCL. The overall response rate (ORR) in HCL is reported at 98%, with a complete response rate (CRR) of 91% .
| Condition | Overall Response Rate | Complete Response Rate |
|---|---|---|
| Hairy Cell Leukemia | 98% | 91% |
| Multiple Sclerosis | 63% (reduction in relapses) | N/A |
Q & A
Basic Research Question: What is the mechanism of action of cladribine in modulating immune responses in multiple sclerosis (MS), and how does this inform experimental design?
Answer: this compound, a purine nucleoside analog, induces selective apoptosis of lymphocytes (primarily B and T cells) by accumulating in cells with high deoxycytidine kinase activity, leading to DNA strand breaks and mitochondrial dysfunction . This lymphotoxicity reduces inflammatory activity in MS. Methodologically, researchers should design in vitro studies to quantify this compound's effect on lymphocyte subpopulations (e.g., flow cytometry for CD4+/CD8+ T cells) and correlate these findings with clinical outcomes (e.g., relapse rates, MRI lesion counts) . Preclinical models, such as experimental autoimmune encephalomyelitis (EAE), can elucidate its neuroprotective effects via intracerebroventricular administration .
Basic Research Question: What standardized protocols exist for this compound administration and outcome measurement in clinical trials?
Answer: The CLARITY trial established a benchmark protocol: two annual short-course oral doses (3.5 or 5.25 mg/kg body weight) over 96 weeks, with MRI monitoring at baseline, 24, 48, and 96 weeks . Key outcomes include annualized relapse rate (ARR), time to sustained disability progression (EDSS score), and "no evidence of disease activity" (NEDA), which integrates relapse frequency, disability progression, and MRI lesions . Researchers must adhere to CONSORT guidelines for reporting adverse events (e.g., lymphopenia, herpes zoster) and ensure rigorous blinding to mitigate bias .
Advanced Research Question: How can researchers reconcile contradictory findings on this compound’s efficacy across studies (e.g., EDSS progression vs. relapse reduction)?
Answer: Discrepancies in outcomes like EDSS progression may arise from heterogeneous patient cohorts, differences in baseline disability, or variable follow-up durations. To address this:
- Conduct stratified analyses by disease activity levels (e.g., highly active vs. moderate MS) .
- Use meta-analytic approaches to pool data from trials (CLARITY, ORACLE MS) and observational registries, adjusting for confounders like prior DMT use .
- Apply causal inference methods (e.g., propensity score matching) to real-world data to isolate this compound’s effect .
Advanced Research Question: What methodologies are recommended for assessing this compound’s long-term safety beyond year 4 of treatment?
Answer: Post-approval data gaps beyond year 4 necessitate:
- Longitudinal cohort studies : Track malignancy rates, lymphocyte recovery, and opportunistic infections using registries (e.g., CLASSIC-MS) .
- Pooled safety analyses : Combine data from extension studies (CLARITY Extension, PREMIERE) to evaluate rare adverse events .
- Comparative effectiveness research : Compare this compound’s safety profile with other DMTs using standardized metrics (e.g., incidence per 1,000 patient-years) .
Basic Research Question: What are the critical safety parameters to monitor in this compound trials, and how should they be operationalized?
Answer: Essential parameters include:
Advanced Research Question: How can real-world evidence (RWE) be integrated with clinical trial data to optimize this compound’s use in diverse populations?
Answer: RWE integration strategies include:
- Hybrid study designs : Embed pragmatic trials within registries to evaluate this compound in underrepresented groups (e.g., elderly patients) .
- Covariate adjustment : Use multivariate regression to account for differences in baseline characteristics (e.g., EDSS, prior DMT failures) .
- Dynamic treatment regimens : Apply machine learning to RWE to identify optimal retreatment timing based on lymphocyte recovery and disease activity .
Advanced Research Question: What statistical approaches address variability in individual responses to this compound therapy?
Answer: Heterogeneity in treatment effects can be analyzed via:
- Mixed-effects models : Account for individual variability in longitudinal outcomes (e.g., EDSS trajectories) .
- Biomarker discovery : Use omics data (e.g., proteomics of CSF) to identify predictors of response .
- Bayesian adaptive trials : Dynamically adjust dosing based on interim analyses of efficacy/safety .
Basic Research Question: How should researchers design preclinical studies to evaluate this compound’s neuroprotective effects?
Answer: Key steps include:
- Animal models : Use EAE models with this compound administered via intracerebroventricular (icv) routes to assess direct CNS effects .
- Outcome measures : Quantify synaptic glutamate alterations (microdialysis) and axonal preservation (histopathology) .
- Dose translation : Apply allometric scaling from rodent doses to human equivalents, ensuring pharmacokinetic comparability .
Advanced Research Question: What strategies optimize this compound’s dosing regimen to balance efficacy and safety?
Answer:
- Pharmacokinetic/pharmacodynamic (PK/PD) modeling : Use population PK models from CLARITY substudies to simulate lymphocyte depletion dynamics .
- Dose de-escalation trials : Test lower cumulative doses (e.g., 2.5 mg/kg) in patients with high infection risk .
- Therapeutic drug monitoring : Correlate plasma this compound levels with ALC and relapse rates to personalize dosing .
Advanced Research Question: How can researchers resolve conflicting evidence on this compound’s malignancy risk?
Answer: Contradictory findings (e.g., CLARITY vs. pooled analyses) require:
- Standardized reporting : Align malignancy definitions across studies (e.g., WHO ICD codes) .
- Comparative risk assessment : Calculate standardized incidence ratios (SIRs) vs. age-matched general populations .
- Mechanistic studies : Investigate this compound’s impact on DNA repair pathways in in vitro lymphocyte models .
Properties
IUPAC Name |
(2R,3S,5R)-5-(6-amino-2-chloropurin-9-yl)-2-(hydroxymethyl)oxolan-3-ol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C10H12ClN5O3/c11-10-14-8(12)7-9(15-10)16(3-13-7)6-1-4(18)5(2-17)19-6/h3-6,17-18H,1-2H2,(H2,12,14,15)/t4-,5+,6+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
PTOAARAWEBMLNO-KVQBGUIXSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1C(C(OC1N2C=NC3=C(N=C(N=C32)Cl)N)CO)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C1[C@@H]([C@H](O[C@H]1N2C=NC3=C(N=C(N=C32)Cl)N)CO)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C10H12ClN5O3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID8022828 | |
| Record name | Cladribine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8022828 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
285.69 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Cladribine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014387 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
6.35e+00 g/L | |
| Record name | Cladribine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014387 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Cladribine is structurally related to fludarabine and pentostatin but has a different mechanism of action. Although the exact mechanism of action has not been fully determined, evidence shows that cladribine is phosphorylated by deoxycytidine kinase to the nucleotidecladribine triphosphate (CdATP; 2-chloro-2′-deoxyadenosine 5′-triphosphate), which accumulates and is incorporated into DNA in cells such as lymphocytes that contain high levels of deoxycytidine kinase and low levels of deoxynucleotidase, resulting in DNA strand breakage and inhibition of DNA synthesis and repair. High levels of CdATP also appear to inhibit ribonucleotide reductase, which leads to an imbalance in triphosphorylated deoxynucleotide (dNTP) pools and subsequent DNA strand breaks, inhibition of DNA synthesis and repair, nicotinamide adenine dinucleotide (NAD) and ATP depletion, and cell death. Unlike other antimetabolite drugs, cladribine has cytotoxic effects on resting as well as proliferating lymphocytes. However, it does cause cells to accumulate at the G1/S phase junction, suggesting that cytotoxicity is associated with events critical to cell entry into S phase. It also binds purine nucleoside phosphorylase (PNP), however no relationship between this binding and a mechanism of action has been established., Cladribine is an antimetabolite. The exact mechanism of action in hairy cell leukemia is unknown. Cladribine is resistant to the action of adenosine deaminase (ADA), which deaminates deoxyadenosine to deoxyinosine. The phosphorylated metabolites of cladribine accumulate in cells with a high ratio of deoxycytidine kinase activity to 5' nucleotidase activity (lymphocytes, monocytes ) and are converted to the active triphosphate deoxynucleotide. Intracellular accumulation of toxic deoxynucleotides selectively kills these cells, which become unable to properly repair single-strand DNA breaks, leading to disruption of cell metabolism. In addition, there is some evidence that deoxynucleotides are incorporated into the DNA of dividing cells and impair DNA synthesis. Cladribine also induces apoptosis (a form of programmed cell death in sensitive cells). Cladribine's action is cell cycle-phase nonspecific; cladribine equally affects dividing and resting lymphocytes., Cladribine has immunosuppressant activity ; restoration of lymphocyte subsets after treatment takes at least 6 to 12 months, although clinical immunocompetence is usually restored after about a month. Significant reductions in T and B lymphocytes occur during treatment (both CD4 and CD8 are affected) and CD4 counts recover more slowly after treatment., /Investigators/ have studied the role of caspases and mitochondria in apoptosis induced by 2-chloro-2'-deoxyadenosine (cladribine) in several human leukemic cell lines. Cladribine treatment induced mitochondrial transmembrane potential (DeltaPsi(m)) loss, phosphatidylserine exposure, caspase activation and development of typical apoptotic morphology in JM1 (pre-B), Jurkat (T) and U937 (promonocytic) cells. Western-blot analysis of cell extracts revealed the activation of at least caspases 3, 6, 8 and 9. Co-treatment with Z-VAD-fmk (benzyloxy-carbonyl-Val-Ala-Asp-fluoromethylketone), a general caspase inhibitor, significantly prevented cladribine-induced death in JM1 and Jurkat cells for the first approximately 40 h, but not for longer times. Z-VAD-fmk also partly prevented some morphological and biochemical features of apoptosis in U937 cells, but not cell death. Co-incubation with selective caspase inhibitors Ac-DEVD-CHO (N-acetyl-Asp-Glu-Val-Asp-aldehyde), Ac-LEHD-CHO (N-acetyl-Leu-Glu-His-Asp-aldehyde) or Z-IETD-fmk (benzyloxycarbonyl-Ile-Glu-Thr-Asp-fluoromethylketone), inhibition of protein synthesis with cycloheximide or cell-cycle arrest with aphidicolin did not prevent cell death. Overexpression of Bcl-2, but not CrmA, efficiently prevented death in Jurkat cells. In all cell lines, death was always preceded by Delta Psi(m) loss and accompanied by the translocation of the protein apoptosis-inducing factor (AIF) from mitochondria to the nucleus. These results suggest that caspases are differentially involved in induction and execution of apoptosis depending on the leukemic cell lineage. In any case, Delta Psi(m) loss marked the point of no return in apoptosis and may be caused by two different pathways, one caspase-dependent and the other caspase-independent. Execution of apoptosis was always performed after Delta Psi(m) loss by a caspase-9-triggered caspase cascade and the action of AIF., Cladribine (chlorodeoxyadenosine, 2-CdA), a synthetic purine nucleoside, is an antineoplastic agent. ... The precise mechanism(s) of antileukemic action of cladribine has not been fully elucidated. Cladribine is phosphorylated by deoxycytidine kinase to the nucleotide cladribine triphosphate (CdATP; 2-chloro-2'-deoxyadenosine 5'-triphosphate), which accumulates and is incorporated into DNA in cells such as lymphocytes that have high levels of deoxycytidine kinase and low levels of deoxynucleotidase. High intracellular concentrations of cladribine triphosphate appear to inhibit ribonucleotide reductase, causing an imbalance in triphosphorylated deoxynucleotide (dNTP) pools and subsequent DNA strand breaks, inhibition of DNA synthesis and repair, nicotinamide adenine dinucleotide (NAD) and ATP depletion, and cell death. Incorporation of accumulated cladribine triphosphate into DNA also may contribute to DNA strand breakage and inhibition of DNA synthesis and repair. Unlike other commonly used antineoplastic drugs that affect purine and pyrimidine metabolism, cladribine has cytotoxic effects on resting as well as proliferating lymphocytes and monocytes. | |
| Record name | Cladribine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00242 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CLADRIBINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7564 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Impurities |
9-(2-deoxy-beta-D-erythro-pentofurnaosyl)-9H-purin-2,6-diamine; 9-(2-deoxy-beta-D-erythro-pentofurnaosyl)-2-methoxy-9H-purin-6-amine; 2-chloro-7H-purin-6-amine; 2-chloro-9-(2-deoxy-alpha-D-erythro-pentofurnaosyl)-9H-purin-6-amine; 2-deoxy-D-erythro-pentofurnaose; 4-methylbenzamide; methyl 4-methylbenzoate | |
| Record name | CLADRIBINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7564 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from water, softens at 210-215 °C, solidifies and turns brown ... Also reported as crystals from ethanol | |
CAS No. |
4291-63-8 | |
| Record name | Cladribine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=4291-63-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Cladribine [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0004291638 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Cladribine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00242 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Cladribine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8022828 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (2R,3S,5R)-5-(6-Amino-2-chloropurin-9-yl)-2-(hydroxymethyl)oxalan-3-ol | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CLADRIBINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/47M74X9YT5 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | CLADRIBINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7564 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Cladribine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014387 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
220 °C (softens), resolidifies, turns brown and does not melt below 300 °C, 215 °C | |
| Record name | Cladribine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00242 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CLADRIBINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7564 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Cladribine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014387 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















