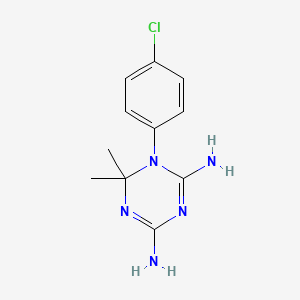
Cycloguanil
Overview
Description
Cycloguanil is a dihydrofolate reductase inhibitor and a metabolite of the antimalarial drug proguanil. It is primarily responsible for the antimalarial activity of proguanil.
Preparation Methods
Cycloguanil can be synthesized through a multi-step processThis intermediate is then condensed with acetone to produce this compound . Industrial production methods may involve optimizing reaction conditions to maximize yield and purity.
Chemical Reactions Analysis
Cycloguanil undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized under specific conditions, leading to the formation of different oxidation products.
Reduction: Reduction reactions can convert this compound into its reduced forms.
Substitution: this compound can undergo substitution reactions, where specific functional groups are replaced by others.
Common reagents and conditions used in these reactions include oxidizing agents, reducing agents, and catalysts. The major products formed from these reactions depend on the specific reagents and conditions employed .
Scientific Research Applications
Antimalarial Activity
Mechanism of Action
Cycloguanil functions primarily as an inhibitor of dihydrofolate reductase (DHFR), an enzyme critical for folate metabolism in Plasmodium species, the parasites responsible for malaria. By inhibiting DHFR, this compound disrupts nucleotide synthesis, ultimately leading to the death of the parasite .
Combination Therapies
The efficacy of this compound is often enhanced when used in combination with other antimalarial agents, such as atovaquone. The combination therapy known as Malarone® (atovaquone-proguanil) exploits this synergy, providing a robust treatment option against both Plasmodium falciparum and Plasmodium vivax infections .
Clinical Studies
Recent studies have demonstrated that proguanil, through its conversion to this compound, exhibits significant antimalarial activity even in populations with high levels of resistance to other treatments. For example, in Vanuatu, patients with poor metabolizer genotypes (CYP2C19) showed high efficacy from proguanil monotherapy .
Cancer Treatment
Inhibition of Human DHFR
This compound has been investigated for its potential as an anticancer agent due to its ability to inhibit human DHFR. This inhibition disrupts folate metabolism critical for rapidly dividing cancer cells . Research indicates that this compound and its analogues can exhibit potent anticancer activity, making them candidates for further development in cancer chemotherapy .
Case Studies and Research Findings
- In Vitro Studies : this compound has shown promising results in vitro against various cancer cell lines. Its analogues have been reported to have IC50 values below 1 µM, indicating strong inhibitory effects comparable to established chemotherapeutic agents like methotrexate .
- Animal Models : In rat models of leukemia, this compound was found to prolong survival when administered as part of a treatment regimen targeting DHFR .
Antiviral Applications
Recent research has expanded the scope of this compound beyond antimalarial and anticancer applications to include antiviral properties. This compound has been identified as a potential host-directed antiviral agent targeting DHFR in host cells, which could inhibit the replication of viruses such as influenza and respiratory syncytial virus (RSV) .
Comparative Data Table
Mechanism of Action
Cycloguanil exerts its effects by inhibiting dihydrofolate reductase, an enzyme crucial for the synthesis of nucleic acids in the Plasmodium parasite. By inhibiting this enzyme, this compound disrupts the parasite’s ability to synthesize DNA and RNA, ultimately leading to its death . The molecular targets and pathways involved include the folate pathway and the synthesis of nucleic acids .
Comparison with Similar Compounds
Cycloguanil is structurally similar to other dihydrofolate reductase inhibitors, such as pyrimethamine and methotrexate. this compound is unique in its specific activity against the Plasmodium parasite. Similar compounds include:
Pyrimethamine: Another dihydrofolate reductase inhibitor used in antimalarial treatments.
Methotrexate: A dihydrofolate reductase inhibitor used in cancer therapy.
Biological Activity
Cycloguanil, the active metabolite of proguanil, is primarily recognized for its role as a dihydrofolate reductase (DHFR) inhibitor, which has significant implications in both cancer therapy and malaria treatment. This article delves into the biological activity of this compound, highlighting its mechanisms, efficacy, and potential applications through a review of recent research findings.
This compound exerts its biological effects primarily by inhibiting DHFR, an enzyme crucial for folate metabolism. This inhibition disrupts nucleotide synthesis, leading to impaired cell proliferation in both cancerous and parasitic cells.
- Dihydrofolate Reductase Inhibition : this compound binds to DHFR with high affinity, effectively blocking the enzyme's activity. This mechanism is critical in the context of cancer therapies where rapid cell division is prevalent .
- Pteridine Reductase Inhibition : Recent studies have also identified this compound as an inhibitor of pteridine reductase (PTR) in Trypanosoma brucei, suggesting additional targets for its action beyond DHFR . The inhibition of PTR can circumvent resistance mechanisms that rely on DHFR mutations.
Anticancer Activity
Recent investigations have demonstrated that this compound and its analogues show promising anticancer properties:
- Target Engagement : this compound analogues were shown to engage DHFR at sub-nanomolar concentrations, indicating potent activity. However, growth impairments in cancer cells were observed only at higher concentrations .
- Metabolite Profiles : The metabolite profiles induced by this compound were found to be similar to those produced by established DHFR inhibitors like methotrexate, suggesting a comparable mechanism of action .
Antimalarial Activity
This compound's role in malaria treatment has been well-documented:
- Proguanil Activation : this compound is formed from proguanil via cytochrome P450 metabolism. This conversion is essential for proguanil's antimalarial efficacy . Studies indicate that proguanil has limited intrinsic activity without this metabolic conversion.
- Synergistic Effects : this compound has been shown to enhance the activity of other antimalarial drugs, such as atovaquone, providing a synergistic effect that can improve treatment outcomes against resistant strains of malaria .
Case Study 1: Cancer Cell Lines
A study involving various cancer cell lines demonstrated that this compound analogues could effectively inhibit cell growth at concentrations that engage DHFR without causing toxicity at lower doses. The use of folinic acid was able to rescue some viability impairments, indicating potential off-target effects or alternative pathways involved in this compound's action .
Case Study 2: Malaria Treatment Efficacy
Clinical trials assessing the efficacy of proguanil and this compound in malaria-endemic regions showed significant antimalarial activity. Notably, patients with genetic variations affecting CYP2C19 enzyme activity exhibited varying responses to proguanil treatment, further emphasizing the importance of metabolic activation to this compound for therapeutic effectiveness .
Data Tables
Q & A
Basic Research Questions
Q. What is the primary mechanism of action of cycloguanil in targeting malaria parasites and cancer cells?
this compound inhibits dihydrofolate reductase (DHFR), a critical enzyme in folate metabolism required for DNA synthesis. In Plasmodium falciparum, DHFR inhibition disrupts pyrimidine biosynthesis, leading to parasite death. In cancer cells, this compound analogs impair STAT3-dependent gene expression and folate metabolism, inducing anti-proliferative effects . Target engagement assays confirm sub-nanomolar DHFR binding, though growth inhibition occurs at higher concentrations, suggesting time-dependent cellular effects .
Q. How does this compound differ pharmacologically from its parent compound, proguanil?
this compound is the active metabolite of proguanil, generated via hepatic CYP2C19-mediated metabolism. While this compound directly inhibits DHFR, proguanil exhibits antimalarial activity through a distinct, non-DHFR target. This differentiation was demonstrated using P. falciparum transformed with human DHFR, where this compound retained activity but proguanil did not .
Q. What methodologies are used to quantify this compound and proguanil in pharmacokinetic studies?
High-performance liquid chromatography (HPLC) with UV detection is standard for plasma and urine quantification. Validated assays achieve lower limits of quantification (LLOQ) of 0.5 μg/L (this compound) and 1 μg/L (proguanil) in plasma, with precision CV ≤6.5%. Non-compartmental analysis in software like Phoenix® WinNonlin® calculates AUC, Cmax, and elimination half-life, while metabolic ratios (this compound/proguanil AUC) assess CYP2C19 activity .
Advanced Research Questions
Q. How do PfDHFR mutations (e.g., A16V + S108T) confer resistance to this compound, and what strategies overcome this?
The A16V mutation introduces steric hindrance, reducing this compound binding, while S108T alters hydrogen bonding. Structure-activity relationship (SAR) studies show that flexible 6-aryl substituents on the triazine ring bypass steric clashes, restoring potency against mutant PfDHFR. Molecular docking (GOLD, Molegro) and dynamics simulations optimize analogs by predicting binding poses and stability .
Q. Why do some this compound analogs retain anti-cancer activity despite folinic acid rescue?
Folinic acid rescues viability in DHFR-dependent models, but certain analogs (e.g., NSC127159) maintain efficacy, suggesting off-target effects. Metabolomic profiling and STAT3 transcriptional assays reveal additional pathways, such as thymidylate synthase inhibition or redox metabolism disruption. Target deconvolution via CRISPR screening or proteomics is recommended to identify secondary targets .
Q. How does CYP2C19 genetic polymorphism influence this compound efficacy, and how is this addressed in clinical research?
CYP2C19 poor metabolizers (*2/*2 genotype) exhibit 8–134-fold higher proguanil/cycloguanil metabolic ratios, reducing this compound exposure. Pharmacogenomic stratification in trials ensures dosing adjustments (e.g., higher proguanil doses for *1/*2 or *2/*2 patients). Co-administration with CYP2C19 inhibitors (e.g., esomeprazole) exacerbates variability, necessitating therapeutic drug monitoring .
Q. What computational approaches optimize this compound analogs for dual activity against cancer and resistant malaria?
Comparative molecular field analysis (CoMFA) identifies steric/electrostatic requirements for PfDHFR binding (q² = 0.77). Hansch 2D-QSAR models link hydrophobicity to wild-type affinity, while ensemble docking (GOLD, Glide) prioritizes compounds with balanced selectivity for human and Plasmodium DHFR. In vitro validation includes enzyme kinetics and NCI-60 cell line screening .
Q. How do drug-drug interactions (e.g., with acid-reducing agents) impact this compound pharmacokinetics?
Proton pump inhibitors (e.g., esomeprazole) increase proguanil AUC by 40–60% via CYP2C19 inhibition, reducing this compound formation. In contrast, tegoprazan (a potassium-competitive acid blocker) shows negligible interaction, making it preferable for co-administration. Clinical studies use crossover designs with geometric mean ratio (GMR) analysis of AUC and Cmax to quantify interactions .
Q. What experimental models validate this compound’s anti-cancer activity beyond DHFR inhibition?
Metabolomic profiling (LC-MS) compares this compound-treated cancer cells to methotrexate controls, identifying shared folate pathway disruptions. STAT3 luciferase reporter assays and ChIP-seq confirm transcriptional inhibition. Xenograft models with DHFR-knockout cells distinguish on-target vs. off-target effects .
Q. How do deuterated isotopes (e.g., this compound-D6) enhance mechanistic studies?
Deuterated analogs improve mass spectrometry sensitivity for tracking drug distribution and metabolism in vitro/in vivo. They also stabilize hydrogen-deuterium exchange in crystallography, resolving binding interactions with PfDHFR or human DHFR. However, isotopic effects on potency require validation via IC50 comparisons (Note: Use with caution due to source reliability concerns).
Methodological Notes
- Contradiction Management : When folinic acid fails to rescue this compound effects, combine siRNA knockdown and metabolomics to confirm secondary targets .
- Resistance Studies : Use isogenic P. falciparum lines with PfDHFR mutations (e.g., A16V + S108T) for IC50 comparisons and cross-resistance profiling .
- Clinical Translation : Incorporate CYP2C19 genotyping in Phase I trials to stratify patients and optimize dosing .
Properties
IUPAC Name |
1-(4-chlorophenyl)-6,6-dimethyl-1,3,5-triazine-2,4-diamine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C11H14ClN5/c1-11(2)16-9(13)15-10(14)17(11)8-5-3-7(12)4-6-8/h3-6H,1-2H3,(H4,13,14,15,16) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
QMNFFXRFOJIOKZ-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1(N=C(N=C(N1C2=CC=C(C=C2)Cl)N)N)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C11H14ClN5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
152-53-4 (hydrochloride) | |
| Record name | Cycloguanil | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000516212 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID9022867 | |
| Record name | Cycloguanil | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9022867 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
251.71 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
516-21-2 | |
| Record name | Cycloguanil | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=516-21-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Cycloguanil | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000516212 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Cycloguanil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB14763 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Cycloguanil | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9022867 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | CYCLOGUANIL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/26RM326WVN | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















