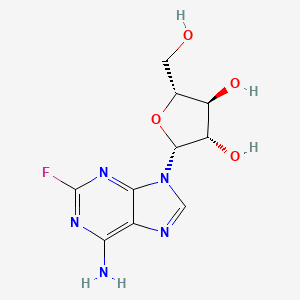
Fludarabine
Overview
Description
Fludarabine is a purine analogue and antineoplastic agentThis compound is primarily used in the treatment of hematological malignancies such as chronic lymphocytic leukemia, non-Hodgkin’s lymphoma, acute myeloid leukemia, and acute lymphocytic leukemia . This compound works by interfering with the duplication of DNA, making it a crucial chemotherapeutic agent .
Preparation Methods
Synthetic Routes and Reaction Conditions: Fludarabine can be synthesized using 2-fluoro-9-beta-D-(2’,3’,5’-tri-alkoxyarabinofuranosyl)adenine as a raw material. The reaction involves a sodium hydroxide and ammonia water mixed solution as a reagent and a water and 2-methyltetrahydrofuran mixed solution as a solvent. The reaction is carried out at 0-5°C for 1-3 hours, followed by neutralization with glacial acetic acid, vacuum filtration, recrystallization, and decolorization with activated carbon to obtain pure this compound .
Industrial Production Methods: Industrial production of this compound involves the esterification and hydrolysis of this compound and phosphorus oxychloride. The process includes coupling purine ring and sugar ring mother nucleus as initial materials, followed by multi-step operations and deprotection to obtain the target product .
Chemical Reactions Analysis
Types of Reactions: Fludarabine undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized to form different metabolites.
Reduction: Reduction reactions can modify the functional groups on the this compound molecule.
Substitution: Substitution reactions can occur at the purine ring or sugar moiety.
Common Reagents and Conditions:
Oxidation: Common oxidizing agents include hydrogen peroxide and potassium permanganate.
Reduction: Reducing agents such as sodium borohydride and lithium aluminum hydride are used.
Substitution: Substitution reactions often involve nucleophiles like halides or amines under basic or acidic conditions.
Major Products: The major products formed from these reactions include various metabolites and derivatives of this compound, which can have different pharmacological properties .
Scientific Research Applications
Chronic Lymphocytic Leukemia (CLL)
Fludarabine has become a standard treatment for CLL, particularly in patients with advanced disease or those who are refractory to previous therapies. The drug is often used alone or in combination with other agents like cyclophosphamide and rituximab.
Case Study: this compound-Cyclophosphamide-Rituximab Regimen
- Patient Cohort : 300 patients with CLL
- Overall Response Rate : 95%
- Complete Remission : 72%
- Median Time to Progression : 80 months
- Survival Rates : Six-year overall survival was reported at 77% .
Allogeneic Stem Cell Transplantation
This compound is frequently included in conditioning regimens prior to allogeneic stem cell transplantation due to its immunosuppressive effects, which facilitate engraftment and reduce the risk of graft-versus-host disease.
Research Findings:
- This compound enhances the engraftment of human hematopoietic cells in immunocompromised mouse models.
- Studies indicate that this compound-treated mice exhibit improved levels of human hematopoiesis compared to controls .
Multiple Myeloma
In multiple myeloma, this compound has been studied as part of combination therapies aimed at improving response rates and prolonging survival.
Clinical Trial Results:
- A study demonstrated that the addition of this compound to standard therapy significantly improved outcomes in relapsed multiple myeloma patients, with notable increases in overall response rates .
Comparative Efficacy
This compound's efficacy has been compared against other therapeutic agents, such as chlorambucil, particularly in elderly patients with CLL.
| Treatment | Overall Response Rate | Complete Remission Rate | Median Progression-Free Survival |
|---|---|---|---|
| Chlorambucil | 51% | 0% | 19 months |
| This compound | 72% | 7% | 18 months |
These results indicate that while this compound may not significantly extend progression-free survival compared to chlorambucil, it does provide a higher overall response rate .
Safety Profile
While this compound is effective, it is associated with adverse effects including cytopenias and increased susceptibility to infections due to its immunosuppressive nature. Long-term studies have highlighted the need for careful monitoring during treatment.
Mechanism of Action
Fludarabine phosphate is rapidly dephosphorylated to 2-fluoro-ara-A and then phosphorylated intracellularly by deoxycytidine kinase to the active triphosphate, 2-fluoro-ara-ATP. This metabolite inhibits DNA polymerase alpha, ribonucleotide reductase, and DNA primase, resulting in the inhibition of DNA synthesis and destruction of cancer cells .
Comparison with Similar Compounds
Clofarabine: Another purine nucleoside analog with similar mechanisms but higher toxicity in the upfront leukemia setting.
Bendamustine: Used as an alternative lymphodepleting regimen with potentially similar response rates and decreased toxicity.
Uniqueness: Fludarabine is unique due to its high selectivity for lymphocytes and its ability to inhibit DNA synthesis in both dividing and resting cells. Its immunosuppressive effects also make it valuable in conditioning regimens prior to stem cell transplantation .
Biological Activity
Fludarabine is a purine analog primarily used in the treatment of hematological malignancies, particularly chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma. Its biological activity is characterized by its ability to inhibit DNA synthesis, induce apoptosis, and exhibit antiviral properties. This article synthesizes findings from various studies to provide a comprehensive overview of the biological activity of this compound, supported by data tables and case studies.
This compound is metabolized in the body to its active triphosphate form, which incorporates into DNA and RNA, thereby inhibiting further synthesis. The compound exerts its effects through several mechanisms:
- Inhibition of DNA Synthesis : this compound inhibits ribonucleotide reductase, leading to a depletion of deoxynucleotide pools necessary for DNA repair and replication. This action enhances its incorporation into newly synthesized DNA, ultimately leading to cell death .
- Induction of Apoptosis : It triggers apoptosis by increasing pro-apoptotic proteins (e.g., Bax) while decreasing anti-apoptotic proteins (e.g., XIAP and survivin) .
- Antiviral Activity : Recent studies have shown that this compound exhibits broad-spectrum antiviral activity against RNA viruses such as Zika virus and SFTS phlebovirus. It inhibits viral RNA replication and protein expression in various cell types .
Efficacy in Clinical Trials
This compound has been evaluated in multiple clinical trials for its efficacy in treating CLL and other hematological cancers. Below is a summary of key findings from notable studies:
| Study Type | Comparison | Response Rate | Progression-Free Survival | Overall Survival |
|---|---|---|---|---|
| First-line therapy vs Chlorambucil | This compound (72%) vs Chlorambucil (51%) | Higher complete remission rate for this compound (7% vs 0%) | Similar (19 months for this compound vs 18 months for chlorambucil) | No significant difference (46 months vs 64 months) |
| Multicenter trial | Untreated patients | This compound (71%) vs Control (60%) | Not specified | Not specified |
| Retrospective analysis | Pre-treated patients | This compound (48%) vs Control (27%) | Not specified | Not specified |
These results indicate that while this compound may achieve higher response rates compared to chlorambucil, it does not significantly prolong overall survival or progression-free survival in certain patient populations .
Case Studies
- Chronic Lymphocytic Leukemia : In a cohort study involving elderly patients with CLL, this compound was administered as a first-line treatment. The study reported a complete remission rate of 7% and an overall response rate of 72%, suggesting that this compound may be particularly effective in early-stage disease but does not significantly improve outcomes in advanced stages .
- Combination Therapy : A study explored the combination of this compound with imatinib mesylate in patients with resistant forms of leukemia. The combination demonstrated synergistic effects, enhancing the overall response rates compared to monotherapy .
Antiviral Properties
This compound's antiviral activity has garnered attention due to its potential application beyond oncology:
Q & A
Basic Research Questions
Q. What experimental methodologies are recommended for assessing Fludarabine’s mechanism of action in hematologic malignancies?
Answer: Studies should integrate in vitro cytotoxicity assays (e.g., apoptosis via Annexin V/PI staining) with ex vivo analyses of patient-derived B-cell chronic lymphocytic leukemia (CLL) samples. Dose-response curves and time-course experiments are critical to differentiate between direct cytotoxicity and immune-modulatory effects. Clinical trials should adhere to standardized response criteria, such as the NCI-WG guidelines, which specify lymphocyte counts (<5 × 10⁹/L) and bone marrow involvement thresholds for remission .
Q. How should researchers design phase I trials to evaluate this compound’s maximum tolerated dose (MTD) in combination therapies?
Answer: Utilize a 3+3 dose-escalation design with toxicity monitoring (e.g., grade 4 leukopenia or pneumonitis). For example, in NSCLC trials, this compound was administered during weeks 5–6 of radiotherapy, starting at 10 mg/m²/day and escalating by 3 mg/m²/day. MTD determination requires rigorous hematologic profiling (lymphocyte counts) and dose-limiting toxicity (DLT) assessment over 2–4 weeks post-treatment .
Q. What are the key considerations for selecting patient cohorts in this compound studies for CLL?
Answer: Prioritize patients with Rai stage III/IV or those refractory to alkylating agents. Eligibility should align with NCI-WG criteria, including absolute lymphocyte counts and bone marrow biopsy results (<30% lymphocytes for complete remission). Exclude individuals with active infections due to this compound’s immunosuppressive effects .
Advanced Research Questions
Q. How can contradictory data on this compound’s efficacy in solid tumors be systematically addressed?
Answer: Conduct meta-analyses stratifying outcomes by tumor type, dosing schedules, and combination regimens (e.g., radiotherapy vs. chemoimmunotherapy). For NSCLC, conflicting results on pneumonitis rates may arise from variations in radiation field size or this compound timing; standardized toxicity reporting (CTCAE v5.0) and subgroup analyses are essential .
Q. What methodologies optimize this compound’s synergy with monoclonal antibodies (e.g., Rituximab) in CLL?
Answer: Use sequential dosing (this compound → Rituximab) to mitigate overlapping myelosuppression. Validate synergy via in vitro complement-dependent cytotoxicity (CDC) assays and correlate with clinical endpoints (e.g., progression-free survival). Pharmacodynamic studies should monitor CD20 expression pre/post-Fludarabine exposure .
Q. How should researchers resolve discrepancies between NCI-WG and IWCLL response criteria in this compound trials?
Answer: Perform sensitivity analyses using both frameworks. For example, NCI-WG mandates stricter bone marrow criteria (<30% lymphocytes) versus IWCLL’s allowance for focal infiltrates. Cross-tabulate outcomes to assess the clinical relevance of these differences, particularly in trials evaluating minimal residual disease (MRD) .
Q. What longitudinal study designs are suitable for evaluating this compound’s long-term immunosuppressive effects?
Answer: Implement 5–10-year follow-up cohorts with serial immune profiling (e.g., CD4+/CD8+ ratios, immunoglobulin levels). Compare with age-matched controls to distinguish treatment-related lymphocytopenia from natural aging. Adjust for confounders like prior therapies using multivariate regression .
Q. Methodological Guidance
Q. How to ensure reproducibility in this compound pharmacokinetic (PK) studies?
Answer: Standardize plasma sampling times (e.g., 0, 1, 4, 24 hrs post-dose) and use LC-MS/MS for metabolite quantification. Report inter-individual variability using coefficient of variation (CV%) and validate assays per FDA bioanalytical guidelines .
Q. What statistical approaches are recommended for analyzing this compound’s dose-response relationships?
Answer: Apply nonlinear mixed-effects modeling (NONMEM) to account for patient heterogeneity. Use Akaike Information Criterion (AIC) to compare log-linear vs. Emax models. For combination therapies, evaluate interaction terms via Chou-Talalay synergy plots .
Q. How to address missing data in this compound clinical trial datasets?
Answer: Employ multiple imputation (MI) for ≤20% missingness, validated against complete-case analyses. For toxicity data, use pattern-mixture models to assess bias. Pre-specify handling methods in the statistical analysis plan (SAP) to maintain rigor .
Q. Data Presentation Standards
- Tables: Include dose-escalation schematics (e.g., cohort size, DLTs) and comparative response rates between NCI-WG/IWCLL criteria.
- Figures: Use Kaplan-Meier curves for survival outcomes and heatmaps for immune cell subset changes post-therapy.
Properties
IUPAC Name |
(2R,3S,4S,5R)-2-(6-amino-2-fluoropurin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C10H12FN5O4/c11-10-14-7(12)4-8(15-10)16(2-13-4)9-6(19)5(18)3(1-17)20-9/h2-3,5-6,9,17-19H,1H2,(H2,12,14,15)/t3-,5-,6+,9-/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
HBUBKKRHXORPQB-FJFJXFQQSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1=NC2=C(N=C(N=C2N1C3C(C(C(O3)CO)O)O)F)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C1=NC2=C(N=C(N=C2N1[C@H]3[C@H]([C@@H]([C@H](O3)CO)O)O)F)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C10H12FN5O4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID4039657 | |
| Record name | Fludarabine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4039657 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
285.23 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Sparingly sol water, org solvents | |
| Record name | Fludarabine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01073 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | FLUDARABINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6964 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
Fludarabine phosphate is rapidly dephosphorylated to 2-fluoro-ara-A and then phosphorylated intracellularly by deoxycytidine kinase to the active triphosphate, 2-fluoro-ara-ATP. This metabolite appears to act by inhibiting DNA polymerase alpha, ribonucleotide reductase and DNA primase, thus inhibiting DNA synthesis. The mechanism of action of this antimetabolite is not completely characterized and may be multi-faceted., Fluorinated adenine analog causes inhibition of DNA synthesis by inhibiting ribonucleotide reductase & DNA polymerase., Fludarabine is a purine antimetabolite. Activity occurs as the result of activation to 2-fluoro-ara-ATP and includes inhibition of DNA synthesis (primarily in the S-phase of cell division by inhibition of ribonucleotide reductase and the DNA polymerases. It is also postulated that fludarabine interferes with RNA by decreased incorporation of uridine and leucine into RNA and protein, respectively. Fludarabine is also active against non-proliferating cells., This review establishes the pharmacokinetic characteristics of the major nucleoside analogs with cytotoxic activity. Cytarabine, pentostatin, fludarabine, cladribine & gemcitabine are all prodrugs whose plasma pharmacokinetics do not fully reflect their therapeutic activity; after cellular uptake, these compounds undergo phosphorylation by deoxycytidine kinase before their incorporation into DNA results in cell death. Cytarabine is principally active in the S phase of the cell cycle & is most toxic to replicating cells, whereas pentostatin, fludarabine & cladribine are incorporated into DNA during the process in which strand breaks are repaired & are therefore cytotoxic to slowly replicating cells (although the action of pentostatin results from its inhibition of adenosine deaminase). Gemcitabine is unusual in being highly metabolized in solid tumor cells. The cytotoxic activity of pentostatin, fludarabine and cladribine against the clonal cells of lymphoproliferative disorders is accompanied by damage to normal lymphoid cells, which results in significant & long-lasting immunosuppression. Useful interactions between nucleoside analogs have been defined. Cells that are primed by exposure to fludarabine or cladribine exhibit enhanced accumulation of cytarabine triphosphate (the cytotoxic nucleotide of cytarabine) & an improved therapeutic effect against acute myeloid leukemia & chronic lymphocytic leukemia can be achieved by clinical schedules that exploit this effect. Combinations of alkylating agents & fludarabine or cladribine are also synergistic in producing significantly enhanced activity against refractory lymphoid malignancies, but at the cost of increased hematological toxicity. Developments in the clinical admin of gemcitabine are concentrating on efforts to extend the duration of exposure to the drug as a means of counteracting its rapid catabolism in the circulation. Future developments with this group of agents will further explore the use of fludarabine-based combination therapies to produce a transient period of myelosuppression & immunosuppression that is sufficient to permit the engraftment of allogeneic hemopoietic stem cells & also exploit the immunological benefits of graft-versus-tumor reactions. In addition, the clinical spectrum of activity of gemcitabine is also being extended by combining the drug with other active chemotherapeutic agents, such as cisplatin, & by early studies of its role as a radiosensitiser. | |
| Record name | Fludarabine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01073 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | FLUDARABINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6964 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals | |
CAS No. |
21679-14-1 | |
| Record name | Fludarabine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=21679-14-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Fludarabine [INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0021679141 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Fludarabine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01073 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Fludarabine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4039657 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Fludarabine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.040.462 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | FLUDARABINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/P2K93U8740 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | FLUDARABINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6964 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Melting Point |
260 °C | |
| Record name | Fludarabine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01073 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | FLUDARABINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6964 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















