Misoprostol
Overview
Description
Misoprostol is a synthetic prostaglandin E1 analogue used for various medical purposes, including the prevention and treatment of stomach and duodenal ulcers, induction of labor, abortion, and treatment of postpartum bleeding due to poor contraction of the uterus . It is a versatile compound with significant applications in medicine.
Preparation Methods
Synthetic Routes and Reaction Conditions
Misoprostol is synthesized through a multi-step process starting from commercially available precursors. The synthesis involves the following key steps:
Formation of the cyclopentane ring: This step involves the cyclization of a suitable precursor to form the cyclopentane ring structure.
Introduction of the hydroxyl groups: Hydroxyl groups are introduced at specific positions on the cyclopentane ring through selective hydroxylation reactions.
Esterification: The hydroxylated cyclopentane intermediate is esterified to form the final this compound structure.
Industrial Production Methods
Industrial production of this compound involves large-scale synthesis using optimized reaction conditions to ensure high yield and purity. The process typically includes:
Batch or continuous flow reactors: These reactors are used to carry out the cyclization and hydroxylation reactions under controlled conditions.
Purification steps: The crude product is purified using techniques such as crystallization, distillation, and chromatography to obtain high-purity this compound.
Chemical Reactions Analysis
Types of Reactions
Misoprostol undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized to form corresponding ketones and aldehydes.
Reduction: Reduction reactions can convert this compound to its corresponding alcohols.
Substitution: this compound can undergo substitution reactions where functional groups are replaced with other groups.
Common Reagents and Conditions
Oxidizing agents: Common oxidizing agents include potassium permanganate and chromium trioxide.
Reducing agents: Sodium borohydride and lithium aluminum hydride are commonly used reducing agents.
Substitution reagents: Halogenating agents such as thionyl chloride and phosphorus tribromide are used for substitution reactions.
Major Products
Oxidation products: Ketones and aldehydes.
Reduction products: Alcohols.
Substitution products: Halogenated derivatives.
Scientific Research Applications
Gastroenterology
Ulcer Prevention
Misoprostol is primarily indicated for the prevention of nonsteroidal anti-inflammatory drug (NSAID)-induced gastric ulcers. It acts by inhibiting gastric acid secretion through direct stimulation of prostaglandin E1 receptors on parietal cells, promoting mucosal protection .
Efficacy and Limitations
While effective, this compound is often less tolerated than alternative treatments like omeprazole, leading to its limited use as a first-line therapy due to side effects such as diarrhea and the need for multiple daily doses .
| Indication | Effectiveness | Alternative Treatments |
|---|---|---|
| NSAID-induced gastric ulcers | Effective | Omeprazole |
| Postoperative ulcer prevention | Moderate | Proton pump inhibitors |
Obstetrics
Labor Induction
this compound is widely used for cervical ripening and labor induction due to its ability to stimulate uterine contractions. It is often favored for its cost-effectiveness compared to dinoprostone .
Case Study: Labor Induction Efficacy
A study involving 500 women showed that this compound resulted in successful labor induction in 85% of cases, with fewer side effects compared to traditional methods .
| Study Parameter | Outcome |
|---|---|
| Total Participants | 500 |
| Successful Induction Rate | 85% |
| Adverse Effects Reported | Minimal |
Reproductive Health
Medical Abortion
this compound is commonly used in combination with mifepristone for medical abortion, achieving effectiveness rates between 95% and 98% . The regimen allows for non-invasive management of early pregnancy loss.
Factors Influencing Success Rates
Research indicates that factors such as the timing of administration and the presence of symptoms like abdominal pain can significantly affect the success rates of this compound in abortion procedures .
| Factor | Success Rate Impact |
|---|---|
| Timing of Administration | Higher success when within 24 hours of symptoms |
| Presence of Abdominal Pain | Increases likelihood of successful expulsion |
Postpartum Hemorrhage
This compound is also utilized in managing postpartum hemorrhage due to its uterotonic properties. It effectively reduces blood loss by promoting uterine contractions .
Safety and Adverse Effects
Despite its benefits, this compound is associated with several adverse effects, including uterine tachysystole and potential lacerations during labor induction . A comprehensive analysis from the FDA Adverse Event Reporting System highlighted various adverse events linked to this compound use, emphasizing the need for careful monitoring during its administration .
| Adverse Event | Frequency |
|---|---|
| Uterine Tachysystole | 95 occurrences |
| Uterine Rupture | 95 occurrences |
| Fetal Heart Rate Decrease | 93 occurrences |
Mechanism of Action
Misoprostol exerts its effects by mimicking the action of natural prostaglandins. It binds to prostaglandin receptors on target cells, leading to various physiological responses:
Gastric mucosa: Inhibits gastric acid secretion and stimulates mucus and bicarbonate secretion, providing a protective effect on the stomach lining.
Uterus: Induces uterine contractions and cervical ripening, facilitating labor induction and abortion.
Vascular smooth muscle: Causes vasodilation, improving blood flow and reducing blood pressure.
Comparison with Similar Compounds
Misoprostol is compared with other prostaglandin analogues such as gemeprost and isosorbide mononitrate:
Gemeprost: Similar to this compound, gemeprost is used for labor induction and abortion.
Isosorbide mononitrate: Used in combination with this compound for enhanced efficacy in medical abortion.
Similar Compounds
- Gemeprost
- Isosorbide mononitrate
- Dinoprostone
- Carboprost
This compound’s unique properties and wide range of applications make it a valuable compound in both research and clinical settings.
Biological Activity
Misoprostol, a synthetic analog of prostaglandin E1, is widely recognized for its diverse biological activities, particularly in gastroprotection and reproductive health. This article delves into the compound's mechanisms of action, clinical applications, and research findings, supported by data tables and case studies.
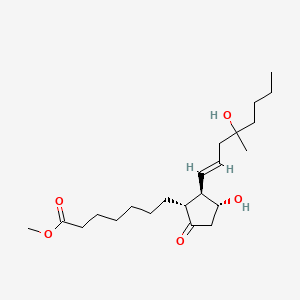
Chemical Structure : this compound is chemically defined as (±)-(11a,13 E)-11,16-dihydroxy-16-methyl-9-oxo-prost-13-en-1-oic acid methyl ester. Its molecular formula is C22H38O5.
Mechanism : this compound exerts its biological effects primarily through agonistic activity at the EP receptors (EP1, EP2, EP3, and EP4). It inhibits gastric acid secretion by stimulating prostaglandin E1 receptors on parietal cells in the stomach. This action reduces acid secretion triggered by various stimuli such as food intake and NSAIDs. Moreover, it promotes the secretion of mucus and bicarbonate, enhancing mucosal defense mechanisms .
Biological Activities
- Gastroprotective Effects : this compound is effective in preventing NSAID-induced gastric ulcers. It facilitates the thickening of the mucosal layer and enhances blood flow to the gastric mucosa, which aids in mucosal regeneration .
- Reproductive Health : this compound is utilized for medical abortion and management of early pregnancy loss. It has been shown to be highly effective when used alone or in combination with mifepristone. Studies indicate that the efficacy of this compound for pregnancy termination ranges from 84% to 96% depending on dosage and administration route .
Table 1: Efficacy of this compound in Medical Abortion
| Study Reference | Gestational Age | Efficacy Rate (%) | Complications (%) | Need for Surgical Intervention (%) |
|---|---|---|---|---|
| ≤63 days | 84 - 96 | 0.7 | 22 | |
| ≤63 days | 92 | 1.0 | 18 | |
| Early Pregnancy Loss | 90 | 3.5 | 10.5 |
Case Studies
Case Study 1: Medical Abortion with this compound Alone
A systematic review involving over 12,000 women demonstrated that this compound alone resulted in a complete abortion rate of approximately 90%. The study highlighted that complications were minimal, with only a small percentage requiring surgical intervention due to incomplete abortion .
Case Study 2: Mifepristone Plus this compound
In a cohort study assessing the combination therapy of mifepristone followed by this compound, it was found that this regimen significantly reduced the need for subsequent surgical procedures compared to this compound alone (10.5% vs. 14%) . This suggests that the combination may enhance efficacy while minimizing complications.
Research Findings
Recent studies have expanded our understanding of this compound's receptor interactions:
- Receptor Binding Studies : Research has shown that this compound binds effectively to EP receptors with Ki values indicating high affinity (EP1: 120 nM; EP3: 67 nM) . This binding is critical for its agonistic effects.
- Structural Analysis : A crystal structure analysis revealed detailed interactions between this compound and the EP3 receptor, providing insights into how structural modifications could lead to more effective analogs for therapeutic use .
Q & A
Basic Research Questions
Q. What experimental designs are optimal for evaluating Misoprostol’s pharmacokinetics in human serum?
- Methodological Answer : Utilize ultra-performance liquid chromatography coupled with tandem mass spectrometry (UPLC-MS/MS) for high sensitivity and specificity. Optimize chromatographic conditions (e.g., retention time of 2.2 minutes for this compound acid) and validate the method using spiked serum samples across a dynamic range (e.g., 3.1–18.4 ppb). Include longitudinal study designs to track metabolite clearance rates .
Q. How can clinical trials standardize outcome measures for this compound’s efficacy in postpartum hemorrhage (PPH) prevention?
- Methodological Answer : Adopt primary endpoints such as blood loss volume (mL), hemoglobin levels post-delivery, and incidence of adverse effects (e.g., pyrexia). Use randomized controlled trials (RCTs) with control groups receiving placebo or alternative uterotonics (e.g., oxytocin). Statistical significance should be assessed via χ² tests or t-tests for categorical and continuous variables, respectively, with P<0.05 .
Q. What are best practices for formulating research questions on this compound’s role in maternal health?
- Methodological Answer : Align questions with gaps identified in systematic reviews (e.g., dose optimization, mortality reduction). Ensure specificity (e.g., “Does 400 µg oral this compound reduce PPH incidence compared to 600 µg?”) and relevance to clinical guidelines. Use frameworks like PICOT (Population, Intervention, Comparison, Outcome, Time) for rigor .
Advanced Research Questions
Q. How can researchers resolve contradictions in maternal mortality data from this compound trials?
- Methodological Answer : Conduct meta-analyses with sensitivity analyses to address heterogeneity. For example, exclude hyperpyrexia from composite outcomes (e.g., “maternal death or severe morbidity”) to isolate blood loss-related effects. Use random-effects models to account for variability across studies (I² >50% indicates high heterogeneity) .
Q. What statistical methods are robust for analyzing adverse effects in this compound trials?
- Methodological Answer : Apply multivariate logistic regression to adjust for confounders (e.g., gestational age, parity). For rare outcomes like maternal death, use Monte Carlo simulations to assess bias-corrected effectiveness. Report risk ratios (RR) with 95% confidence intervals (CI) for outcomes such as meconium-stained liquor or pyrexia .
Q. How can mixed-methods approaches enhance understanding of this compound implementation barriers?
- Methodological Answer : Combine quantitative surveys (e.g., 21-item questionnaires on prescribing practices) with qualitative interviews to explore contextual factors (e.g., healthcare provider attitudes). Use thematic analysis for qualitative data and χ² tests for quantitative trends. Ensure data saturation in interviews for reliability .
Q. What methodologies optimize dose-response studies for this compound in labor induction?
- Methodological Answer : Compare lower doses (e.g., 25–50 µg) to standard regimens using RCTs stratified by gestational age. Measure outcomes such as time to vaginal delivery, cesarean rates, and uterine hyperstimulation. Employ survival analysis (e.g., Kaplan-Meier curves) to evaluate induction timelines .
Properties
IUPAC Name |
methyl 7-[(1R,2R,3R)-3-hydroxy-2-[(E)-4-hydroxy-4-methyloct-1-enyl]-5-oxocyclopentyl]heptanoate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H38O5/c1-4-5-14-22(2,26)15-10-12-18-17(19(23)16-20(18)24)11-8-6-7-9-13-21(25)27-3/h10,12,17-18,20,24,26H,4-9,11,13-16H2,1-3H3/b12-10+/t17-,18-,20-,22?/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
OJLOPKGSLYJEMD-URPKTTJQSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCCCC(C)(CC=CC1C(CC(=O)C1CCCCCCC(=O)OC)O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCCCC(C)(C/C=C/[C@H]1[C@@H](CC(=O)[C@@H]1CCCCCCC(=O)OC)O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H38O5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID7020897 | |
| Record name | Misoprostol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7020897 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
382.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Liquid | |
| Record name | Misoprostol | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015064 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
1.6mg/mL, Water-soluble, 1.64e-02 g/L | |
| Record name | Misoprostol | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00929 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | MISOPROSTOL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3573 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Misoprostol | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015064 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Misoprostol is a synthetic prostaglandin E1 analog that stimulates prostaglandin E1 receptors on parietal cells in the stomach to reduce gastric acid secretion. Mucus and bicarbonate secretion are also increased along with thickening of the mucosal bilayer so the mucosa can generate new cells. Misoprostol binds to smooth muscle cells in the uterine lining to increase the strength and frequency of contractions as well as degrade collagen and reduce cervical tone., Misoprostol enhances natural gastromucosal defense mechanisms and healing in acid-related disorders, probably by increasing production of gastric mucus and mucosal secretion of bicarbonate., Misoprostol inhibits basal and nocturnal gastric acid secretion by direct action on the parietal cells; also inhibits gastric acid secretion stimulated by food, histamine, and pentagastrin. It decreases pepsin secretion under basal, but not histamine stimulation. Misoprostol has no significant effect on fasting or postprandial gastrin or intrinsic factor output. | |
| Record name | Misoprostol | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00929 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | MISOPROSTOL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3573 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Light yellow oil, Viscous liquid | |
CAS No. |
59122-46-2 | |
| Record name | Misoprostol | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=59122-46-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Misoprostol [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0059122462 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Misoprostol | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00929 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Misoprostol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7020897 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Misoprostol | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | MISOPROSTOL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/0E43V0BB57 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | MISOPROSTOL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3573 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Misoprostol | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015064 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
261-263 | |
| Record name | Misoprostol | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00929 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















