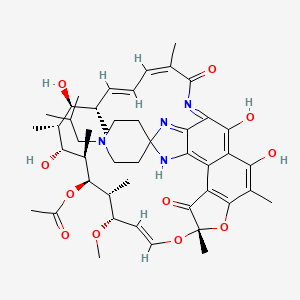
Rifabutin
Overview
Description
Preparation Methods
Synthetic Routes and Reaction Conditions
Rifabutin is synthesized through a semisynthetic process starting from rifamycin S. The process involves several steps, including the modification of the rifamycin S structure to introduce the spiropiperidyl group . The reaction conditions typically involve the use of organic solvents and catalysts to facilitate the chemical transformations.
Industrial Production Methods
Industrial production of this compound involves large-scale synthesis using optimized reaction conditions to ensure high yield and purity. The process is carefully controlled to maintain the quality and consistency of the final product. The production involves multiple purification steps, including crystallization and chromatography, to achieve the desired purity levels .
Chemical Reactions Analysis
Types of Reactions
Rifabutin undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized to form different derivatives.
Reduction: Reduction reactions can modify the functional groups on the this compound molecule.
Substitution: Substitution reactions can introduce new functional groups into the this compound structure.
Common Reagents and Conditions
Common reagents used in these reactions include oxidizing agents like potassium permanganate, reducing agents like sodium borohydride, and various organic solvents. The reactions are typically carried out under controlled temperature and pressure conditions to ensure the desired outcomes .
Major Products Formed
The major products formed from these reactions include various derivatives of this compound with modified antimicrobial properties. These derivatives are often studied for their potential use in treating different bacterial infections .
Scientific Research Applications
Treatment of Mycobacterial Infections
Tuberculosis and Non-Tuberculous Mycobacterial Infections
Rifabutin is most notably used in the treatment of tuberculosis (TB), especially for patients with rifampicin-resistant strains. Studies have shown that this compound can be effective in treating patients diagnosed with rifampicin-resistant tuberculosis (RR-TB), particularly when used in combination with other antitubercular agents. The Xpert MTB/RIF assay has significantly increased the identification of RR-TB cases, leading to a growing interest in this compound-containing regimens as viable therapeutic options for these patients .
Table 1: Treatment Success Rates for this compound vs. Rifampicin
| Study Type | This compound Treatment Success Rate (%) | Rifampicin Treatment Success Rate (%) |
|---|---|---|
| Randomized Trials | 54.7 | 67.5 |
| Observational Studies | Varies | Varies |
This table summarizes the findings from a meta-analysis comparing treatment success rates between this compound and rifampicin, indicating that while this compound is effective, rifampicin may have a higher overall success rate .
Repurposing for Other Bacterial Infections
Recent studies have explored the potential of repurposing this compound for treating infections caused by Staphylococcus aureus, particularly in cases of hospital-acquired infections (HAIs). The structural similarities between this compound and rifampicin allow for comparable antibacterial activity against various bacterial strains, making it a candidate for addressing antibiotic resistance challenges .
Case Study: Efficacy Against Staphylococcus aureus
In a recent investigation, researchers found that this compound demonstrated significant antibacterial effects against Staphylococcus aureus, suggesting its potential as an alternative treatment option in scenarios where traditional antibiotics fail due to resistance .
Use in HIV-Infected Patients
This compound is frequently employed in HIV-infected patients to prevent opportunistic infections such as Mycobacterium avium complex (MAC). The drug's ability to reduce the systemic exposure of other antiretroviral drugs has been documented, which is crucial for managing drug interactions in this population .
Table 2: Drug Interaction Impact of this compound
| Drug | Effect on Systemic Exposure |
|---|---|
| Maraviroc | Decreased systemic exposure |
| Clarithromycin | Potential protection against resistance |
This table illustrates the effects of this compound on other medications commonly used in HIV treatment, highlighting its role in mitigating resistance development .
Pharmacokinetics and Dosage Considerations
This compound exhibits distinct pharmacokinetic properties that influence its therapeutic applications. It has a longer half-life than rifampicin, allowing for less frequent dosing while maintaining effective drug levels in the body. Typical dosages range from 300 mg to 1200 mg per day, depending on the infection being treated and the patient's overall health status .
Table 3: Recommended Dosages for Various Indications
| Indication | Recommended Dosage (mg/day) |
|---|---|
| Tuberculosis | 300 - 600 |
| Mycobacterium avium complex (MAC) | 300 - 1200 |
| Staphylococcus infections | 600 - 900 |
This table provides an overview of recommended dosages for different clinical indications, emphasizing the flexibility of dosing based on specific patient needs .
Adverse Effects and Safety Profile
While generally well-tolerated, this compound can lead to adverse effects such as uveitis and gastrointestinal disturbances. Monitoring is essential when prescribing this medication, especially in populations at risk for drug interactions or those receiving multiple therapies .
Case Report: Uveitis Induced by this compound
A documented case highlighted the occurrence of uveitis in a patient undergoing treatment with this compound for MAC infection. This emphasizes the need for vigilance and patient education regarding potential side effects during therapy .
Mechanism of Action
Rifabutin exerts its effects by inhibiting the DNA-dependent RNA polymerase in bacteria. This inhibition prevents the transcription of bacterial DNA into RNA, thereby stopping bacterial growth and replication . The molecular targets of this compound include the RNA polymerase enzyme and various components of the bacterial transcription machinery .
Comparison with Similar Compounds
Similar Compounds
Rifampin: Another ansamycin antibiotic with a similar mechanism of action but different spectrum of activity.
Rifapentine: A derivative of rifamycin with a longer half-life and different pharmacokinetic properties.
Rifamycin S: The parent compound from which Rifabutin is synthesized.
Uniqueness
This compound is unique in its enhanced activity against Mycobacterium avium complex and its ability to induce cytochrome P-450 3A at lower levels compared to Rifampin . This makes this compound a valuable option for treating infections caused by resistant mycobacteria and for use in combination therapy with other antimicrobial agents .
Biological Activity
Rifabutin is a semi-synthetic derivative of rifampicin, primarily used as an antibiotic for treating mycobacterial infections, including tuberculosis and Mycobacterium avium complex (MAC). This article explores the biological activity of this compound, focusing on its mechanisms of action, efficacy against various pathogens, and clinical implications supported by research findings and case studies.
This compound exerts its antibacterial effects primarily through the inhibition of DNA-dependent RNA polymerase in susceptible bacteria. This action leads to the suppression of RNA synthesis, ultimately resulting in bacterial cell death. The drug has shown effectiveness against a range of bacteria, including:
- Mycobacterium tuberculosis
- Mycobacterium avium complex
- Acinetobacter baumannii
The selective uptake mechanism involves the FhuE receptor , which is upregulated in nutrient-limited conditions, enhancing this compound's activity in specific media such as RPMI supplemented with fetal calf serum (FCS) .
In Vitro Studies
This compound has demonstrated significant in vitro activity against carbapenem-resistant Acinetobacter baumannii (CRAB) isolates. In a study assessing the minimum inhibitory concentration (MIC), this compound exhibited potent activity, particularly when cellular uptake was facilitated by FhuE overexpression .
Clinical Efficacy
A meta-analysis comparing this compound-based regimens to rifampin-based regimens indicated that the pooled treatment success rate for this compound was 54.7% (95% CI: 41.0-67.0%) compared to 67.5% (95% CI: 55.7-77.4%) for rifampin . This suggests that while this compound is effective, it may be less successful than rifampin in certain contexts.
Case Studies
- Treatment of MAC : In a cohort study involving patients with disseminated MAC disease, those treated with this compound showed varying success rates based on the number of drugs used in their regimen:
- Staphylococcal Prosthetic Infections : A case series reported on the use of this compound in patients with staphylococcal infections associated with prosthetic devices. Some patients experienced adverse effects, but overall results supported further investigation into its use as an alternative to rifampin .
Pharmacokinetics and Drug Interactions
This compound is well absorbed orally, with an average bioavailability of approximately 20% and a high protein binding rate of about 85% . It undergoes hepatic metabolism, producing several active metabolites that contribute to its antimicrobial activity . Notably, this compound can interact with other medications; for example, it decreases the systemic exposure of maraviroc in HIV-negative adults .
Q & A
How can researchers formulate a focused, hypothesis-driven research question for Rifabutin studies?
Methodological Answer :
Use the PICOT framework (Population, Intervention, Comparison, Outcome, Time) to structure questions. For example:
- Population: Immunocompromised patients with Mycobacterium avium complex (MAC).
- Intervention: this compound dosage optimization.
- Comparison: Standard this compound regimens vs. adjusted doses in drug-drug interaction scenarios.
- Outcome: Efficacy (e.g., microbial clearance) and safety (e.g., adverse events).
- Time: Short-term (6 weeks) vs. long-term (12 months) outcomes.
Apply the FINER criteria (Feasible, Interesting, Novel, Ethical, Relevant) to refine the question. For instance, prioritize studies addressing this compound’s unique pharmacokinetic (PK) properties over older rifamycins .
Q. What experimental design considerations are critical for this compound PK/PD studies?
Methodological Answer :
- Population PK modeling : Account for covariates like hepatic impairment or concomitant CYP3A4 inhibitors (e.g., clarithromycin), which alter this compound metabolism. Use nonlinear mixed-effects modeling (NONMEM) to analyze sparse data .
- Dose fractionation studies : Isolate pharmacodynamic (PD) effects (e.g., AUC/MIC ratios) to distinguish bactericidal vs. post-antibiotic effects.
- Control groups : Include comparator arms with rifampicin to highlight this compound’s reduced CYP induction .
Q. How can researchers resolve contradictions in this compound’s clinical trial data?
Methodological Answer :
- Meta-analysis : Pool data from trials using standardized inclusion criteria (e.g., MAC prophylaxis in HIV patients) and adjust for heterogeneity via random-effects models.
- Sensitivity analysis : Test robustness by excluding outlier studies (e.g., those with non-standard dosing).
- Mechanistic modeling : Link PK variability (e.g., CYP3A5 polymorphisms) to divergent efficacy outcomes .
Q. What strategies improve reproducibility in this compound in vitro susceptibility testing?
Methodological Answer :
- Standardize inoculum preparation : Use McFarland 0.5 suspensions in Middlebrook 7H9 broth to minimize variability in minimum inhibitory concentration (MIC) measurements.
- Control for drug stability : this compound degrades in light; use amber vials and validate storage conditions.
- Reference strains : Include M. tuberculosis H37Rv as a control to calibrate assay conditions .
Q. How should researchers handle this compound’s photodegradation in preclinical studies?
Methodological Answer :
- Light-protected workflows : Conduct experiments under red light or in opaque containers.
- High-performance liquid chromatography (HPLC) : Quantify this compound and its active metabolite (25-O-desacetylthis compound) to confirm stability.
- Accelerated stability studies : Use Arrhenius modeling to predict degradation kinetics under varying temperatures .
Q. What advanced statistical methods are suitable for analyzing this compound’s resistance mechanisms?
Methodological Answer :
- Whole-genome sequencing (WGS) : Identify mutations in rpoB (rifampicin resistance-determining region) and correlate with phenotypic resistance via logistic regression.
- Machine learning : Train models on genomic and PK/PD data to predict resistance emergence.
- Time-kill assays : Use parametric survival models to quantify bacterial regrowth under sub-MIC this compound exposure .
Q. How can cross-disciplinary approaches enhance this compound research?
Methodological Answer :
- Immunopharmacology : Integrate cytokine profiling (e.g., IL-6, TNF-α) with PK data to assess immunomodulatory effects.
- Nanotechnology : Evaluate liposomal encapsulation to improve this compound’s bioavailability and target tissue penetration.
- Computational chemistry : Perform molecular docking studies to predict this compound’s binding affinity to mutant RNA polymerases .
Q. What are best practices for curating and reusing this compound datasets?
Methodological Answer :
- Metadata annotation : Include experimental conditions (e.g., pH, temperature), assay type (e.g., broth microdilution), and strain details.
- FAIR principles : Ensure datasets are Findable, Accessible, Interoperable, and Reusable. Use platforms like Zenodo or Figshare for deposition.
- Critical appraisal : Verify data provenance via peer-reviewed publications or trusted repositories (e.g., ChEMBL) .
Q. How to design translational studies bridging this compound’s in vitro and in vivo efficacy?
Methodological Answer :
- Hollow-fiber infection models : Simulate human PK profiles in vitro to identify optimal dosing regimens.
- Interspecies scaling : Apply allometric principles to extrapolate murine data to human equivalents.
- Biomarker validation : Corrogate in vitro MICs with clinical outcomes (e.g., sputum conversion rates) using receiver operating characteristic (ROC) curves .
Q. What ethical considerations apply to this compound trials in vulnerable populations?
Methodological Answer :
- Informed consent : Address literacy barriers in TB-endemic regions using visual aids or community liaisons.
- Safety monitoring : Implement DSMBs (Data Safety Monitoring Boards) for trials involving hepatotoxic regimens.
- Equitable access : Partner with local health systems to ensure post-trial access for control groups .
Properties
IUPAC Name |
[(7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17,32-tetrahydroxy-11-methoxy-3,7,12,14,16,18,22-heptamethyl-1'-(2-methylpropyl)-6,23-dioxospiro[8,33-dioxa-24,27,29-triazapentacyclo[23.6.1.14,7.05,31.026,30]tritriaconta-1(32),2,4,9,19,21,24,26,30-nonaene-28,4'-piperidine]-13-yl] acetate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C46H62N4O11/c1-22(2)21-50-18-16-46(17-19-50)48-34-31-32-39(54)28(8)42-33(31)43(56)45(10,61-42)59-20-15-30(58-11)25(5)41(60-29(9)51)27(7)38(53)26(6)37(52)23(3)13-12-14-24(4)44(57)47-36(40(32)55)35(34)49-46/h12-15,20,22-23,25-27,30,37-38,41,48,52-55H,16-19,21H2,1-11H3/b13-12+,20-15+,24-14-,47-36?/t23-,25+,26+,27+,30-,37-,38+,41+,45-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
ZWBTYMGEBZUQTK-PVLSIAFMSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1C=CC=C(C(=O)N=C2C(=C3C(=C4C2=NC5(N4)CCN(CC5)CC(C)C)C6=C(C(=C3O)C)OC(C6=O)(OC=CC(C(C(C(C(C(C1O)C)O)C)OC(=O)C)C)OC)C)O)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@H]1/C=C/C=C(\C(=O)N=C2C(=C3C(=C4C2=NC5(N4)CCN(CC5)CC(C)C)C6=C(C(=C3O)C)O[C@@](C6=O)(O/C=C/[C@@H]([C@H]([C@H]([C@@H]([C@@H]([C@@H]([C@H]1O)C)O)C)OC(=O)C)C)OC)C)O)/C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C46H62N4O11 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
847.0 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Minimally soluble (0.19 mg/mL) | |
| Record name | Rifabutin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00615 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Rifabutin acts via the inhibition of DNA-dependent RNA polymerase in gram-positive and some gram-negative bacteria, leading to a suppression of RNA synthesis and cell death. | |
| Record name | Rifabutin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00615 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
72559-06-9 | |
| Record name | Rifabutin [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0072559069 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Rifabutin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00615 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















