Gefitinib
Overview
Description
Gefitinib, sold under the brand name Iressa, is a medication primarily used for the treatment of certain types of cancer, including non-small cell lung cancer and breast cancer. It is a selective inhibitor of the epidermal growth factor receptor (EGFR) tyrosine kinase, which plays a crucial role in the regulation of cell growth and survival .
Preparation Methods
Synthetic Routes and Reaction Conditions: Gefitinib is synthesized through a multi-step process starting from 4,5-dimethoxy-2-nitrobenzoic acid. The synthesis involves several key steps, including demethylation, esterification, side-chain alkylation, reduction, cyclohexylamine formation, chlorination, and ammonia substitution .
Industrial Production Methods: In industrial settings, this compound is produced using optimized synthetic routes to ensure high yield and purity. The process involves strict control of reaction conditions, such as temperature, pH, and solvent selection, to achieve the desired product quality .
Chemical Reactions Analysis
Types of Reactions: Gefitinib undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized to form different metabolites.
Reduction: Reduction reactions can modify the quinazoline ring structure.
Substitution: Substitution reactions can occur at the aromatic ring, leading to the formation of derivatives.
Common Reagents and Conditions:
Oxidation: Common oxidizing agents include hydrogen peroxide and potassium permanganate.
Reduction: Reducing agents such as sodium borohydride and lithium aluminum hydride are used.
Substitution: Halogenating agents like chlorine and bromine are often employed.
Major Products: The major products formed from these reactions include various this compound derivatives with altered pharmacological properties .
Scientific Research Applications
Non-Small Cell Lung Cancer (NSCLC)
- First-Line Treatment : Gefitinib is approved for use as a first-line treatment in patients with advanced NSCLC harboring sensitive EGFR mutations. Clinical trials have shown that this compound significantly improves progression-free survival compared to standard chemotherapy in this patient population .
- Second-Line Treatment : In cases where patients have previously received chemotherapy, this compound remains an effective option. Studies indicate that it offers better tolerability and quality of life compared to traditional chemotherapy regimens .
- Brain Metastases : One of the notable advantages of this compound is its ability to penetrate the blood-brain barrier effectively. This property makes it a viable option for treating patients with brain metastases from NSCLC, where other therapies may fail to achieve therapeutic concentrations .
Other Cancers
While this compound's primary application is in NSCLC, research is ongoing into its efficacy against other malignancies:
- Head and Neck Cancers : Some studies suggest potential benefits in head and neck squamous cell carcinoma, particularly in tumors expressing high levels of EGFR .
- Combination Therapies : Recent investigations have explored the synergistic effects of this compound when combined with other agents like anlotinib, showing enhanced efficacy against resistant NSCLC cell lines .
Case Study 1: Efficacy in Asian Populations
A significant body of research indicates that this compound is particularly effective among Asian populations with adenocarcinoma histology and those who have never smoked. The IRESSA Pan-Asia Study (IPASS) revealed an overall response rate exceeding 80% in patients with EGFR mutation-positive tumors . This study emphasizes the importance of genetic profiling in optimizing treatment strategies.
Case Study 2: Long-Term Outcomes
A longitudinal study involving patients treated with this compound over several years demonstrated sustained efficacy and manageable side effects. Patients reported improved quality of life metrics compared to those receiving standard chemotherapy regimens .
Adverse Effects
While this compound is generally well-tolerated, it can cause side effects such as skin rash, diarrhea, and liver enzyme elevation. Monitoring and management strategies are essential to mitigate these effects during treatment .
Mechanism of Action
Gefitinib is often compared with other EGFR inhibitors, such as erlotinib and afatinib. While all three compounds target the EGFR tyrosine kinase, they differ in their pharmacokinetic properties and clinical efficacy:
Erlotinib: Similar mechanism of action but has a different side effect profile and dosing regimen.
Afatinib: Irreversible inhibitor of EGFR, offering a broader spectrum of activity against EGFR mutations
Uniqueness: this compound is unique in its selective inhibition of the EGFR tyrosine kinase and its ability to target specific mutations in cancer cells, making it a valuable tool in personalized cancer therapy .
Comparison with Similar Compounds
- Erlotinib
- Afatinib
- Lapatinib
- Osimertinib
Biological Activity
Gefitinib, an epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, is primarily used in the treatment of non-small cell lung cancer (NSCLC) with specific EGFR mutations. This article explores the biological activity of this compound, focusing on its mechanisms of action, efficacy in clinical trials, and emerging research findings.
This compound selectively binds to the ATP-binding site of the EGFR tyrosine kinase domain, inhibiting its phosphorylation and subsequent activation of downstream signaling pathways. This results in decreased cell proliferation and increased apoptosis in cancer cells expressing mutant forms of EGFR. The compound has shown significant effects on various cellular processes, including:
- Inhibition of Cell Proliferation : this compound suppresses the growth of cancer cells by blocking EGFR-mediated signaling pathways.
- Induction of Apoptosis : It promotes programmed cell death through alterations in mitochondrial function and expression levels of Bcl-2 family proteins, such as Bcl-xL and Bax .
- Impact on Mitochondrial Activity : Recent studies indicate that this compound enhances mitochondrial functions, such as succinate-tetrazolium reductase (STR) activity, particularly in high-density cell cultures .
Efficacy in Clinical Trials
This compound has been evaluated extensively in clinical trials for its efficacy and safety profile. Key findings from several studies are summarized below:
Case Studies
- Mitochondrial Activity Enhancement : In a study involving HCC827 cells (EGFR mutation positive), this compound was shown to enhance mitochondrial membrane potential and STR activity, indicating its role as a mitochondrial protector during combination therapy with doxorubicin .
- Resistance Development : Despite its initial effectiveness, resistance to this compound often develops within one to two years due to various mechanisms including secondary mutations in the EGFR gene or activation of alternative signaling pathways . A case study highlighted a patient who initially responded well but later exhibited resistance due to a T790M mutation.
- Urothelial Carcinoma : In vitro studies demonstrated this compound's inhibitory effects on growth and invasion in urothelial carcinoma cell lines, suggesting potential applications beyond NSCLC .
Q & A
Basic Research Questions
Q. What is the molecular mechanism of Gefitinib in targeting EGFR mutations, and how does this inform preclinical study design?
this compound inhibits epidermal growth factor receptor (EGFR) tyrosine kinase activity by competitively binding to the ATP-binding site, thereby blocking downstream signaling pathways critical for tumor survival and proliferation . Preclinical studies should prioritize cell lines or patient-derived xenograft (PDX) models harboring specific EGFR mutations (e.g., exon 19 deletions or exon 21 L858R point mutations) to replicate clinical responses. Western blotting for phosphorylated EGFR (p-EGFR) and downstream markers (e.g., MAPK/ERK) is essential to validate target engagement .
Q. How do EGFR exon 19 deletions versus exon 21 point mutations influence this compound sensitivity in non-small cell lung cancer (NSCLC)?
Exon 19 deletions are associated with higher this compound sensitivity compared to exon 21 L858R mutations due to structural differences in the EGFR kinase domain that affect drug-binding affinity. Researchers should stratify clinical or preclinical cohorts by mutation type and use dose-response assays (e.g., IC50 calculations) to quantify differential efficacy. Immunoblotting can further reveal variations in downstream signaling inhibition .
Q. What are the key biomarkers to monitor this compound response in vitro and in vivo?
Primary biomarkers include:
- EGFR mutation status : Confirmed via Sanger sequencing or next-generation sequencing (NGS).
- p-EGFR levels : Assessed via Western blot or immunofluorescence .
- Extracellular matrix (ECM) synthesis : Relevant in studies of degenerative diseases (e.g., intervertebral disc repair), measured via COL2A1 or aggrecan expression .
- Apoptosis markers : e.g., cleaved caspase-3 in tumor models.
Advanced Research Questions
Q. How can researchers design experiments to investigate acquired resistance to this compound in NSCLC?
Acquired resistance often involves secondary EGFR mutations (e.g., T790M) or activation of bypass pathways (e.g., MET amplification). Methodological approaches include:
- Longitudinal sampling : Collect tumor biopsies pre-treatment, during response, and at progression for genomic profiling .
- Combination therapies : Test this compound with MET inhibitors (e.g., crizotinib) in vitro using synergy assays (e.g., Chou-Talalay method).
- CRISPR screens : Identify genes conferring resistance by knocking out candidate pathways in this compound-sensitive cell lines.
- Liquid biopsy analysis : Detect emerging resistance mutations via circulating tumor DNA (ctDNA) .
Q. What strategies reconcile contradictory clinical data on this compound efficacy across demographic groups (e.g., higher response rates in Asian populations)?
Contradictions often arise from population-specific EGFR mutation prevalence. Researchers should:
- Control for mutation frequency : Use meta-analyses to adjust for EGFR mutation rates in different cohorts.
- Incorporate pharmacogenomic variables : e.g., CYP3A4/5 polymorphisms affecting drug metabolism.
- Leverage multi-center trials : Ensure diverse participant inclusion and standardized mutation screening protocols .
Q. How can this compound delivery systems (e.g., injectable hydrogels) be optimized for preclinical testing in non-cancer applications?
For conditions like intervertebral disc degeneration:
- Hydrogel characterization : Assess drug release kinetics via HPLC and biocompatibility via cytotoxicity assays (e.g., CCK-8).
- Functional outcomes : Measure ECM synthesis (COL2, aggrecan) and autophagy markers (LC3-II) in 3D cell cultures or rodent models .
- Comparative dosing : Test localized vs. systemic delivery to minimize off-target effects.
Q. What frameworks (e.g., FINER, PICO) are optimal for formulating this compound-related research questions?
- PICO Framework : Define P opulation (e.g., NSCLC patients with exon 19 deletions), I ntervention (this compound + adjuvant therapy), C omparator (standard chemotherapy), O utcome (progression-free survival).
- FINER Criteria : Ensure questions are F easible (adequate patient cohorts), I nteresting (addresses resistance mechanisms), N ovel (e.g., combinatorial regimens), E thical, and R elevant (clinical translatability) .
Q. Methodological Considerations
Q. How should researchers validate this compound’s off-target effects in complex disease models (e.g., degenerative disorders)?
- Transcriptomic profiling : Use RNA-seq to identify non-EGFR pathways modulated by this compound.
- High-content imaging : Quantify changes in cellular morphology or matrix deposition in 3D cultures .
- Pharmacological inhibition : Combine this compound with EGFR siRNA to isolate EGFR-dependent effects.
Q. What statistical approaches are critical for analyzing this compound trial data with heterogeneous responses?
- Subgroup analysis : Stratify patients by mutation type, smoking history, or ethnicity.
- Kaplan-Meier survival curves : Compare time-to-progression between subgroups.
- Multivariate regression : Adjust for confounders like CYP3A4 activity or drug-drug interactions .
Q. How can preclinical studies improve translational relevance for this compound-based therapies?
- Use PDX models : Preserve patient tumor heterogeneity and microenvironment interactions.
- Mimic clinical dosing : Align in vitro drug concentrations with human pharmacokinetic data.
- Co-clinical trials : Parallel preclinical and clinical testing to rapidly validate biomarkers .
Properties
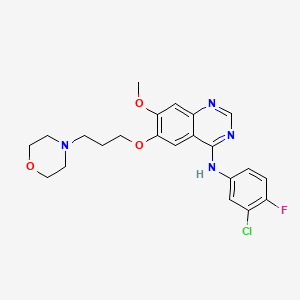
IUPAC Name |
N-(3-chloro-4-fluorophenyl)-7-methoxy-6-(3-morpholin-4-ylpropoxy)quinazolin-4-amine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H24ClFN4O3/c1-29-20-13-19-16(12-21(20)31-8-2-5-28-6-9-30-10-7-28)22(26-14-25-19)27-15-3-4-18(24)17(23)11-15/h3-4,11-14H,2,5-10H2,1H3,(H,25,26,27) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XGALLCVXEZPNRQ-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
COC1=C(C=C2C(=C1)N=CN=C2NC3=CC(=C(C=C3)F)Cl)OCCCN4CCOCC4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H24ClFN4O3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID8041034 | |
| Record name | Gefitinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8041034 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
446.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Gefitinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014462 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Sparingly soluble (|
|
|
| Record name | Gefitinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00317 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Gefitinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014462 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Gefitinib is an inhibitor of the epidermal growth factor receptor (EGFR) tyrosine kinase that binds to the adenosine triphosphate (ATP)-binding site of the enzyme. EGFR is often shown to be overexpressed in certain human carcinoma cells, such as lung and breast cancer cells. Overexpression leads to enhanced activation of the anti-apoptotic Ras signal transduction cascades, subsequently resulting in increased survival of cancer cells and uncontrolled cell proliferation. Gefitinib is the first selective inhibitor of the EGFR tyrosine kinase which is also referred to as Her1 or ErbB-1. By inhibiting EGFR tyrosine kinase, the downstream signaling cascades are also inhibited, resulting in inhibited malignant cell proliferation. | |
| Record name | Gefitinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00317 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
184475-35-2 | |
| Record name | Gefitinib | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=184475-35-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Gefitinib [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0184475352 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Gefitinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00317 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Gefitinib | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759856 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Gefitinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8041034 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Gefitinib | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | GEFITINIB | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/S65743JHBS | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Gefitinib | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014462 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details







Synthesis routes and methods II
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













