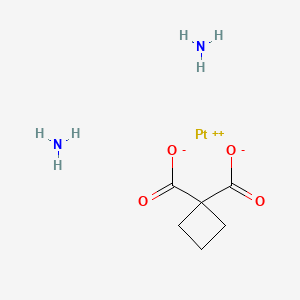
Carboplatin
Overview
Description
Carboplatin is a platinum-based chemotherapy medication used to treat various forms of cancer, including ovarian cancer, lung cancer, head and neck cancer, brain cancer, and neuroblastoma . It was developed as a less toxic analogue of cisplatin and was approved for medical use in 1989 . This compound works by interfering with the duplication of DNA, thereby inhibiting the growth of cancer cells .
Preparation Methods
Synthetic Routes and Reaction Conditions: Carboplatin is synthesized through the reaction of cisplatin with cyclobutane-1,1-dicarboxylic acid in the presence of ammonia . The reaction conditions typically involve heating the mixture to facilitate the formation of the this compound complex.
Industrial Production Methods: In industrial settings, this compound is produced by dissolving cisplatin in water and adding cyclobutane-1,1-dicarboxylic acid and ammonia. The mixture is then heated to promote the reaction, and the resulting this compound is purified through crystallization .
Chemical Reactions Analysis
Types of Reactions: Carboplatin undergoes various chemical reactions, including substitution reactions. One notable reaction is its activation by chloride ions, which leads to the removal of the malonate moiety and the formation of cisplatin .
Common Reagents and Conditions:
Chloride Ions: Used to activate this compound and convert it to cisplatin.
Acidic Conditions: Strongly acidic environments facilitate the conversion of this compound to cisplatin.
Major Products Formed:
Scientific Research Applications
Carboplatin has a wide range of scientific research applications:
Mechanism of Action
Carboplatin exerts its effects by forming reactive platinum complexes that cause intra- and inter-strand cross-linkage of DNA molecules within the cell . This modification of DNA structure inhibits DNA synthesis and affects the cell cycle, ultimately leading to cell death . The molecular targets of this compound include DNA, where it forms covalent bonds and disrupts the replication process .
Comparison with Similar Compounds
Cisplatin: The predecessor of carboplatin, known for its higher toxicity but similar mechanism of action.
Oxaliplatin: Another platinum-based chemotherapy drug with a different spectrum of activity and toxicity profile.
Comparison:
Cisplatin vs. This compound: this compound is less toxic than cisplatin, particularly in terms of nephrotoxicity and gastrointestinal side effects.
Oxaliplatin vs. This compound: Oxaliplatin has a different spectrum of activity and is often used to treat colorectal cancer.
This compound’s reduced toxicity and similar efficacy make it a valuable alternative to cisplatin in chemotherapy regimens, particularly for patients who cannot tolerate the side effects of cisplatin .
Biological Activity
Carboplatin, a platinum-based chemotherapeutic agent, is widely used in the treatment of various cancers, particularly ovarian and lung cancers. This article delves into its biological activity, mechanisms of action, efficacy in clinical settings, and associated toxicities, supported by data tables and case studies.
This compound exerts its anticancer effects primarily through the formation of DNA cross-links. Upon entering the cell, this compound undergoes hydrolysis to become positively charged, allowing it to bind covalently to nucleophilic sites on DNA, particularly the N7 position of guanine bases. This binding disrupts DNA replication and transcription, leading to apoptosis in cancer cells .
Key Mechanisms:
- DNA Cross-linking: Formation of DNA adducts that inhibit replication.
- Protein Interactions: this compound also interacts with RNA and proteins, contributing to its cytotoxic effects .
- Resistance Mechanisms: Cancer cells may develop resistance through various mechanisms, including increased drug efflux and enhanced DNA repair capabilities .
Efficacy in Clinical Trials
A significant body of research has evaluated the efficacy of this compound in clinical settings. A randomized clinical trial compared single-agent this compound with combination therapies involving paclitaxel. The results indicated that single-agent this compound had significantly worse survival outcomes compared to combination regimens in older patients with ovarian cancer .
Summary of Clinical Findings:
| Treatment Regimen | Completion Rate | Survival Outcomes |
|---|---|---|
| This compound + Paclitaxel (Q3W) | 65% | Better than single-agent |
| Single-Agent this compound | 48% | Significantly worse survival |
| Weekly this compound + Paclitaxel | 60% | Comparable to combination |
This trial underscores the importance of combination therapies in enhancing treatment efficacy while minimizing adverse effects.
Case Studies
In preclinical models, this compound has shown significant tumor growth inhibition. For instance, studies have demonstrated its effectiveness in subcutaneous lung cancer models, highlighting its potential as a first-line treatment option .
Notable Case Study:
- Patient Profile: An 80-year-old female with stage IV ovarian cancer.
- Treatment Regimen: this compound AUC 5 mg/mL·min + Paclitaxel.
- Outcome: The patient completed six cycles with manageable side effects but experienced disease progression post-treatment.
Toxicity and Side Effects
While this compound is effective against various tumors, it is associated with several toxicities. The most common adverse effects include:
- Hematological Toxicity: Neutropenia and thrombocytopenia are frequently observed.
- Neurotoxicity: this compound can cause peripheral neuropathy due to its effects on sensory neurons .
- Nausea and Vomiting: These are common side effects that can significantly impact patient quality of life.
Recent Research Findings
Recent studies have explored the enhancement of this compound's activity through various mechanisms. For example, research indicates that this compound enhances the activation of transient receptor potential ankyrin 1 (TRPA1) channels in sensory neurons, which may contribute to its neurotoxic effects but also suggests potential pathways for mitigating pain associated with chemotherapy-induced peripheral neuropathy (CIPN) .
Q & A
Basic Research Questions
Q. How is Carboplatin dosage calculated based on renal function in clinical research, and what methodological considerations ensure accuracy?
this compound dosing relies on the Calvert formula: Dose (mg) = Target AUC × (GFR + 25) . This formula was derived from pharmacokinetic studies linking this compound plasma clearance to glomerular filtration rate (GFR) and validated prospectively across diverse patient cohorts . Key considerations include:
- GFR measurement : Use standardized methods (e.g., iohexol clearance or Cockcroft-Gault equation).
- Target AUC selection : Tailor to patient history (e.g., AUC 5 for pretreated patients; AUC 7 for untreated patients).
- Nonrenal clearance validation : Ensure body surface area does not influence nonrenal clearance, as demonstrated in pharmacokinetic modeling .
Q. What are the standard efficacy metrics for evaluating this compound in solid tumors, and how are they applied in clinical trials?
Efficacy is assessed using RECIST (Response Evaluation Criteria in Solid Tumors) guidelines, which standardize tumor measurement and response classification (e.g., complete/partial response, stable/progressive disease) . Key metrics include:
- Overall Survival (OS) : Median survival and hazard ratios.
- Progression-Free Survival (PFS) : Time to disease progression or death.
- Toxicity grading : Use CTCAE (Common Terminology Criteria for Adverse Events) to quantify hematological/non-hematological adverse effects. Example: In NSCLC trials, this compound-paclitaxel showed a median survival of 7.9 months, comparable to cisplatin-based regimens .
Q. How should researchers design experiments to compare this compound monotherapy with combination regimens?
- Randomization : Assign patients to monotherapy vs. combination arms to minimize bias .
- Endpoint selection : Prioritize PFS for early efficacy signals and OS for long-term outcomes.
- Sample size calculation : Use power analysis to detect clinically meaningful differences (e.g., 80% power, α = 0.05).
- Toxicity monitoring : Predefine thresholds for dose modification (e.g., grade 3 thrombocytopenia). Example: A phase II trial comparing this compound-paclitaxel with/without cetuximab required 108 patients to detect a hazard ratio of 0.70 .
Advanced Research Questions
Q. How can pharmacokinetic modeling address interpatient variability in this compound response?
Pharmacokinetic models (e.g., two-compartment models) integrate GFR, nonrenal clearance, and AUC to predict drug exposure. Methodological steps include:
- Plasma sampling : Collect serial post-infusion samples to estimate clearance rates.
- Nonlinear mixed-effects modeling : Use software like NONMEM to account for covariates (e.g., age, albumin levels).
- Validation : Compare predicted vs. observed AUC values (e.g., ±15% error margin) . Example: A refined Calvert formula reduced interpatient variability in AUC prediction from 24% to 11% in prospective studies .
Q. What statistical approaches resolve contradictions in this compound efficacy data across studies?
- Meta-analysis : Pool data from trials with homogeneous designs (e.g., fixed cisplatin-carboplatin ratios) to identify trends.
- Subgroup analysis : Stratify by biomarkers (e.g., ERCC1 expression) or patient characteristics (e.g., smoking status).
- Bayesian hierarchical models : Adjust for confounding variables (e.g., prior therapies, supportive care differences). Example: A meta-analysis of 1,207 NSCLC patients found no survival difference between this compound-paclitaxel and cisplatin-gemcitabine, despite divergent toxicity profiles .
Q. How can researchers predict synergistic effects of this compound in multidrug cocktails using limited experimental data?
The "pairs model" predicts synergy by testing drug pairs and extrapolating to higher-order combinations. Steps include:
- Dose-response matrices : Measure viability curves for individual drugs and pairs.
- Bliss independence or Loewe additivity : Quantify synergy/antagonism.
- Validation in vivo : Use xenograft models to confirm efficacy. Example: A study combining this compound with etoposide and camptothecin achieved 80% tumor reduction in vitro using this approach .
Q. What strategies ensure reproducibility in preclinical studies evaluating this compound resistance mechanisms?
- NIH guidelines compliance : Document cell line authentication, passage numbers, and assay conditions (e.g., hypoxia vs. normoxia) .
- Open data practices : Share raw data (e.g., RNA-seq files) via repositories like GEO or Figshare.
- Replication cohorts : Validate findings in independent cell lines or patient-derived organoids. Example: A study on this compound resistance in ovarian cancer required three independent replicates for all qPCR and Western blot results .
Q. Methodological Tables
Table 1. Key Parameters in the Calvert Formula
| Parameter | Definition | Value Range |
|---|---|---|
| Target AUC | Area under the plasma concentration curve | 4–8 mg/mL·min |
| GFR | Glomerular filtration rate | 33–135 mL/min |
| Nonrenal clearance | Independent of body surface area | 20–30 mL/min |
Table 2. Efficacy of this compound in Phase III NSCLC Trials
| Regimen | Median OS (months) | 1-Year Survival (%) | Grade 3+ Toxicity (%) |
|---|---|---|---|
| This compound-Paclitaxel | 7.9 | 33 | 23 (neutropenia) |
| Cisplatin-Gemcitabine | 8.1 | 34 | 29 (nephrotoxicity) |
| Data from Schiller et al. (2002) . |
Properties
IUPAC Name |
azanide;cyclobutane-1,1-dicarboxylic acid;platinum(2+) | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C6H8O4.2H2N.Pt/c7-4(8)6(5(9)10)2-1-3-6;;;/h1-3H2,(H,7,8)(H,9,10);2*1H2;/q;2*-1;+2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
VSRXQHXAPYXROS-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CC(C1)(C(=O)O)C(=O)O.[NH2-].[NH2-].[Pt+2] | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C6H12N2O4Pt | |
| Record name | Carboplatin | |
| Source | Wikipedia | |
| URL | https://en.wikipedia.org/wiki/Carboplatin | |
| Description | Chemical information link to Wikipedia. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
371.25 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Sol in water, Water > 15 (mg/mL), pH 4 Acetate Buffer 5 - 10 (mg/mL), pH 9 Carbonate Buffer 5 - 10 (mg/mL), 10% Ethanol/H2O 5 - 10 (mg/mL), 95% Ethanol/H < 1 (mg/mL), 0.1NHC1 5 -10 (mg/mL), 0.1NNaOH 5 -10 (mg/mL), Methanol < 1 (mg/mL), Chloroform < 5 (mg/mL), Dimethylsulfoxide 5 (mg/mL), Acetic Acid < 1 (mg/mL), Trifluoroacetic Acid < 1 (mg/mL) | |
| Record name | CARBOPLATIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6957 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | CARBOPLATIN | |
| Source | NCI Investigational Drugs | |
| URL | http://dtp.nci.nih.gov/NCI-InvestigationalDrugsCI92/241240%20(1992).txt | |
| Description | An investigational drug is one that is under study but does not have permission from the U.S. Food and Drug Administration (FDA) to be legally marketed and sold in the United States. NCI provides the investigational drug to the physicians who are participating in clinical trials or TRC protocols. For more information please visit NCI investigational drug website: https://www.cancer.gov/about-cancer/treatment/drugs/investigational-drug-access-fact-sheet | |
Color/Form |
White crystals | |
CAS No. |
41575-94-4 | |
| Record name | carboplatin | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758182 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | carboplatin | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=241240 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | carboplatin | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=201345 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Diammine[cyclobutane-1,1-dicarboxylato(2-)-O,O']platinum | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.050.388 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CARBOPLATIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6957 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















