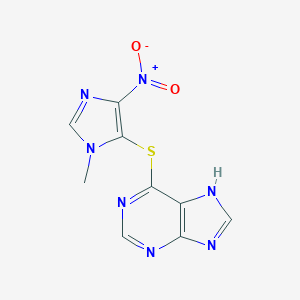
Azathioprine
Overview
Description
Azathioprine is an immunosuppressive medication used primarily to prevent organ transplant rejection and to treat autoimmune diseases such as rheumatoid arthritis, Crohn’s disease, and systemic lupus erythematosus . It was first synthesized in 1956 by Gertrude Elion, William Lange, and George Hitchings . This compound is a prodrug that is converted into 6-mercaptopurine in the body, which then interferes with purine synthesis, thereby inhibiting the proliferation of cells, particularly those of the immune system .
Preparation Methods
Azathioprine is synthesized through a multi-step process. The primary synthetic route involves the reaction of 6-mercaptopurine with 1-methyl-4-nitro-5-imidazole . The reaction conditions typically include the use of a solvent such as dimethylformamide and a base like sodium hydroxide. The reaction proceeds through the formation of an intermediate, which is then cyclized to form this compound .
Industrial production methods of this compound involve similar synthetic routes but are optimized for large-scale production. This includes the use of continuous flow reactors and automated systems to ensure consistent quality and yield .
Chemical Reactions Analysis
Conversion to 6-Mercaptopurine (6-MP)
- Azathioprine undergoes conversion to 6-mercaptopurine (6-MP) through a reductive cleavage of the thioether (-S-) . This process occurs non-enzymatically and is mediated by glutathione and similar compounds present in the intestinal wall, liver, and red blood cells .
- The enzyme thiopurine S-methyltransferase (TPMT) plays a role in the metabolism of this compound. TPMT is responsible for the methylation of 6-MP into the inactive metabolite 6-methylmercaptopurine, which prevents 6-MP from further conversion into active, cytotoxic thioguanine nucleotide metabolites .
Metabolism of 6-MP
6-MP is further metabolized through several pathways :
- Methylation: 6-MP is methylated to 6-methylmercaptopurine by thiopurine methyltransferase .
- Oxidation: 6-MP is oxidized to 6-thiouric acid by xanthine oxidase .
- Conversion to 6-Thioinosine-5'-monophosphate: 6-MP is converted to 6-thioinosine-5'-monophosphate by hypoxanthine phosphoribosyltransferase .
- Anabolic Pathway : requires intracellular activation by hypoxanthine phosphoribosyltransferase (HPRT) into 6-thioinosine-monophosphate (TIMP) which is converted, by the action of inosinemonophosphate dehydrogenase (IMPDH), to 6-thioxanthosine-monophosphate and finally, by the action of guanosine monophosphate synthase (GMPS) into thioguanine nucleotides (TGNs), the active cytotoxic metabolites .
Reaction with Cysteine
- This compound reacts with cysteine, a biogenic thiol. The reaction mechanism involves a nucleophilic attack of the COO- of cysteine on the C(5i) atom of the imidazole ring of this compound, followed by an intramolecular attack of the SH group of the cysteine residue . Biogenic thiols like glutathione or cysteine facilitate the first step of this compound metabolism because of the presence of COO(-), SH, and NH3(+) groups in their molecules .
Role of Gut Microbiota
- The gut microbiota can also metabolize this compound . The human GST enzyme is considered the most efficient enzyme in the bioactivation of prodrug this compound. The mechanism of action of human GSTs related to biotransformation of this compound includes a nucleophilic attack of the sulfur atom of deprotonated glutathione on the slightly electrophilic 5′ carbon atom in the imidazole moiety of this compound, forming 6-mercaptopurine and a glutathione-imidazole conjugate .
Electrochemical Behavior
- This compound's electrochemical behavior has been studied using voltammetric analysis . The reduction of this compound involves 2 electrons and 2 protons, leading to the conversion of the NO2 group to dihydroxylamine, which then dehydrates . The redox reactions of this compound and its interactions with DNA have been investigated .
Scientific Research Applications
Autoimmune Diseases
Azathioprine is commonly prescribed for several autoimmune diseases, including:
- Rheumatoid Arthritis : It helps reduce inflammation and slow disease progression.
- Lupus Nephritis : Used as part of a regimen to manage kidney inflammation associated with systemic lupus erythematosus.
- Crohn's Disease and Ulcerative Colitis : Functions as a maintenance therapy to induce and maintain remission in inflammatory bowel disease .
Organ Transplantation
In renal transplantation, this compound is utilized as an immunosuppressant to prevent organ rejection. It is often combined with other immunosuppressive agents to enhance efficacy while minimizing toxicity .
Dermatological Conditions
This compound is also indicated for various skin conditions such as:
- Severe Atopic Dermatitis : Helps control severe eczema that does not respond to conventional therapies.
- Lupus Erythematosus : Used in cases where skin manifestations are prominent and require systemic treatment .
Comparative Data Table
| Application | Indication | Dosage Form | Monitoring Requirements |
|---|---|---|---|
| Rheumatoid Arthritis | Chronic inflammation | Oral/Injection | Regular blood tests for liver function |
| Lupus Nephritis | Kidney inflammation | Oral/Injection | Regular blood tests for kidney function |
| Crohn's Disease | Inflammatory bowel disease | Oral | Regular blood tests for hematological status |
| Organ Transplantation | Prevention of rejection | Oral/Injection | Close monitoring for signs of rejection |
| Severe Atopic Dermatitis | Persistent eczema | Oral | Monitoring for skin reactions |
Case Study 1: this compound in Ulcerative Colitis
A comprehensive review highlighted the role of this compound in maintaining remission in patients with ulcerative colitis. The study noted that while this compound has been effective, there are concerns regarding its side effects and the need for regular monitoring to manage potential adverse reactions .
Case Study 2: this compound in Renal Transplantation
In a clinical trial involving renal transplant patients, this compound was shown to significantly reduce the incidence of acute rejection episodes when used alongside corticosteroids. The study underscored the importance of individualized dosing and monitoring strategies to optimize outcomes while minimizing risks .
Mechanism of Action
Azathioprine exerts its effects by being converted into 6-mercaptopurine in the body . 6-mercaptopurine is then further metabolized into active metabolites that inhibit purine synthesis. This inhibition leads to a reduction in the proliferation of rapidly dividing cells, particularly those of the immune system . The primary molecular targets of this compound include enzymes involved in purine synthesis, such as hypoxanthine-guanine phosphoribosyltransferase and thiopurine S-methyltransferase . These pathways ultimately lead to the suppression of the immune response .
Comparison with Similar Compounds
Azathioprine is part of the thiopurine class of drugs, which also includes 6-mercaptopurine and thioguanine . Compared to these compounds, this compound has a unique structure that includes an additional imidazole ring bonded to the sulfur atom . This structural difference gives this compound a distinct pharmacokinetic profile and makes it more suitable for certain clinical applications .
Similar Compounds
6-Mercaptopurine: Used primarily in the treatment of leukemia and autoimmune diseases.
Thioguanine: Used in the treatment of acute myeloid leukemia.
Methotrexate: Another immunosuppressive drug used in the treatment of cancer and autoimmune diseases.
This compound’s unique structure and mechanism of action make it a valuable drug in the treatment of various autoimmune conditions and in preventing organ transplant rejection .
Biological Activity
Azathioprine (AZA) is a purine analog widely used as an immunosuppressant in various medical conditions, particularly in the management of autoimmune diseases and organ transplantation. This article delves into its biological activity, including its mechanism of action, metabolism, clinical applications, and associated risks.
This compound is a prodrug that is metabolized into its active forms, primarily 6-mercaptopurine (6-MP) and 6-thioguanine nucleotides (6-TGN). The conversion occurs through enzymatic pathways involving hypoxanthine-guanine phosphoribosyltransferase (HPRT) and thiopurine methyltransferase (TPMT) . The active metabolites inhibit purine synthesis, which is critical for DNA replication and cell division. This mechanism is particularly effective against lymphocytes, which rely on the de novo pathway for purine synthesis due to their limited salvage pathway capabilities .
Metabolism
The metabolism of this compound involves several key enzymes:
- TPMT : Variability in TPMT activity among individuals can significantly affect the therapeutic response and toxicity of AZA. Low TPMT activity leads to increased levels of 6-TGN, enhancing efficacy but also raising the risk of myelosuppression .
- Xanthine oxidase : This enzyme competes with TPMT in the metabolism of 6-MP, influencing the levels of active metabolites and their effects .
Table 1 summarizes the metabolic pathways and their implications for therapy:
| Enzyme | Role in Metabolism | Clinical Implications |
|---|---|---|
| HPRT | Converts AZA to 6-MP | Essential for activation of AZA |
| TPMT | Converts 6-MP to 6-TGN | Low activity increases risk of toxicity |
| Xanthine oxidase | Competes with TPMT | Influences therapeutic levels of 6-TGN |
Clinical Applications
This compound is primarily used in:
- Rheumatoid Arthritis : It helps manage symptoms and prevent disease progression.
- Organ Transplantation : AZA is employed to prevent rejection by suppressing the immune response.
- Inflammatory Bowel Disease (IBD) : It maintains remission in conditions like Crohn's disease and ulcerative colitis .
Case Studies
- Kidney Transplant Patient with Diarrhea : A case study reported a renal transplant patient who developed severe diarrhea while on AZA. Despite normal renal function, high levels of 6-TGN were noted, leading to a change in therapy that resolved gastrointestinal symptoms .
- Undetectable 6-TGN Levels : Another patient experienced acute cellular rejection despite receiving adequate doses of AZA. Subsequent testing revealed undetectable levels of 6-TGN, indicating potential issues with drug metabolism or adherence .
Efficacy and Safety Profile
The efficacy of this compound can vary based on genetic factors affecting metabolism. Studies indicate that patients with low TPMT activity benefit from lower doses to avoid toxicity, while those with high activity may require higher doses for therapeutic effect .
Risks Associated with this compound
While AZA is effective, it carries risks:
- Myelosuppression : A significant risk associated with high levels of 6-TGN.
- Cancer Risk : There is an established association between AZA use and an increased risk of lymphoma among patients with IBD . However, some studies suggest a protective effect against colorectal cancer .
Table 2 outlines the potential adverse effects linked to this compound:
| Adverse Effect | Description | Risk Factors |
|---|---|---|
| Myelosuppression | Decreased blood cell production | Low TPMT activity |
| Lymphoma | Increased risk in IBD patients | Long-term use |
| Hepatotoxicity | Liver damage due to metabolite accumulation | High doses or genetic predisposition |
Q & A
Basic Research Questions
Q. What methodologies are recommended for determining the thermodynamic dissociation constants (pKa) of azathioprine under physiological conditions?
- Methodological Answer: Use multiwavelength spectrophotometric pH-titration with nonlinear regression (e.g., SPECFIT32, SQUAD84) and factor analysis (INDICES programme) to resolve spectral data. Validate results against computational predictions (e.g., PALLAS software) for structural accuracy. At 25°C, pKa = 8.07 ± 0.01; at 37°C, pKa = 7.84 ± 0.01 .
- Key Data: Ionic strength (0.01–0.2), temperature (25°C and 37°C), and rigorous goodness-of-fit tests ensure reliability.
Q. How should researchers design experiments to assess this compound’s immunosuppressive efficacy in autoimmune disorders?
- Methodological Answer: Use randomized controlled trials (RCTs) with placebo comparators, focusing on relapse frequency and disability progression. For example:
- Primary Endpoints: Relative risk reduction (RRR) of relapses at 1–3 years (e.g., RRR = 18–23% in multiple sclerosis trials).
- Secondary Endpoints: Adverse effects (e.g., gastrointestinal disturbances, hepatic toxicity) requiring dose adjustments .
Q. What experimental approaches are validated for monitoring this compound metabolite levels in inflammatory bowel disease (IBD) patients?
- Methodological Answer: Quantify thioguanine diphosphate (TGDP) and triphosphate (TGTP) metabolites via high-performance liquid chromatography (HPLC). TGTP levels correlate with therapeutic response; ratios < 20 predict poor outcomes .
- Statistical Consideration: Use receiver operating characteristic (ROC) curves to establish metabolite thresholds for clinical decision-making.
Advanced Research Questions
Q. How can researchers resolve contradictions in clinical trial outcomes, such as this compound’s divergent efficacy in idiopathic pulmonary fibrosis (IPF) versus multiple sclerosis (MS)?
- Methodological Answer: Conduct meta-analyses with subgroup stratification by disease pathophysiology. For IPF:
- Key Finding: this compound combined with prednisone and N-acetylcysteine increased mortality (8 vs. 1 deaths, p = 0.01) and hospitalization (23 vs. 7 cases, p < 0.001) compared to placebo .
- Mechanistic Hypothesis: Contrast IPF’s fibrotic microenvironment with MS’s inflammatory milieu to explain differential drug effects.
Q. What statistical frameworks are optimal for comparing this compound’s efficacy against biologics (e.g., dupilumab) in atopic dermatitis?
- Methodological Answer: Use surface-under-the-curve (SUCRA) analysis in network meta-analyses. Example:
- Result: High-dose cyclosporine (300 mg/day) outperformed dupilumab (low certainty), while this compound showed inferior efficacy (p < 0.05) .
- Sensitivity Analysis: Address heterogeneity via leave-one-out tests and GRADE certainty assessments.
Q. How can researchers improve the predictive power of this compound’s pharmacokinetic models using computational chemistry?
- Methodological Answer: Integrate density functional theory (DFT) with molecular dynamics simulations to predict this compound’s solubility and membrane permeability. Validate against experimental pKa and partition coefficient (logP) data .
- Open Science: Share code repositories (e.g., GitHub) for model reproducibility .
Q. What strategies mitigate bias in retrospective studies assessing this compound’s long-term cancer risk?
- Methodological Answer: Apply propensity score matching to balance cohorts by age, dosage, and comorbidities. Use competing-risk regression models to account for mortality unrelated to malignancy .
- Ethical Reporting: Disclose conflicts of interest and funding sources per CONSORT guidelines .
Q. Methodological Best Practices
- Data Reproducibility: Publish raw spectrophotometric data (e.g., absorbance vs. wavelength) and regression code in supplementary materials .
- Clinical Trial Design: Pre-register protocols on ClinicalTrials.gov and adhere to SPIRIT checklists for adverse event reporting .
- Literature Synthesis: Use PICO framework (Population, Intervention, Comparison, Outcome) to structure systematic reviews .
Properties
IUPAC Name |
6-(3-methyl-5-nitroimidazol-4-yl)sulfanyl-7H-purine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C9H7N7O2S/c1-15-4-14-7(16(17)18)9(15)19-8-5-6(11-2-10-5)12-3-13-8/h2-4H,1H3,(H,10,11,12,13) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
LMEKQMALGUDUQG-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CN1C=NC(=C1SC2=NC=NC3=C2NC=N3)[N+](=O)[O-] | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C9H7N7O2S | |
| Record name | AZATHIOPRINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19837 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
55774-33-9 (hydrochloride salt) | |
| Record name | Azathioprine [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000446866 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID4020119 | |
| Record name | Azathioprine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4020119 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
277.27 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Azathioprine appears as pale yellow crystals or yellowish powder. Decomposes at 243-244 °C. Used for the treatment of rheumatoid arthritis. A known carcinogen., Solid | |
| Record name | AZATHIOPRINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19837 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Azathioprine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015128 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
>41.6 [ug/mL] (The mean of the results at pH 7.4), less than 1 mg/mL at 73 °F (NTP, 1992), Insoluble, Very slightly soluble in ethanol and chloroform; sparingly soluble in dilute mineral acids; soluble in dilute alkali solutions, Insoluble in water, 1.07e+00 g/L | |
| Record name | SID26670994 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | AZATHIOPRINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19837 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Azathioprine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00993 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | AZATHIOPRINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7084 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Azathioprine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015128 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Azathioprine's mechanism of action is not entirely understood but it may be related to inhibition of purine synthesis, along with inhibition of B and T cells. 6-thioguanine triphosphate, a metabolite of azathioprine, modulates activation of rac1 when costimulated with CD28, inducing T cell apoptosis. This may be mediated through rac1's action on mitogen-activated protein kinase, NF-kappaB., Following exposure to nucleophiles ... azathioprine is cleaved to 6-mercaptopurine which, in turn, is converted to additional metabolites that inhibit de novo purine synthesis. 6-Thio-IMP, a fraudulent nucleotide, is converted to 6-thio-GMP and finally to 6-thio-GTP, which is incorporated into DNA and gene translation is inhibited. Cell proliferation is prevented, inhibiting a variety of lymphocyte functions., Azathioprine (AZA), one of the antimetabolite drugs, is a purine analog that is more potent than the prototype 6-mercaptopurine, as an inhibitor of cell replication. Immunosuppression likely occurs because of the ability of the drug to inhibit purine biosynthesis. ... Although T-cell functions are the primary targets for this drug, inhibition of /(natural killer cells)/ NK function and macrophage activities has also been reported., Azathioprine inhibits DNA synthesis and, as a purine antimetabolite, exerts its effect on activated lymphocytes, which requires purines during their proliferative phase. It inhibits both cellular and humoral responses, but does not interfere with phagocytosis or interferon production. It is a nonspecific cytotoxic agent. Its immunosuppressive effect is believed to be due to mercaptopurine, to which it is metabolized., The exact mechanism of immunosuppressive action is unknown since the exact mechanism of the immune response itself is complex and not completely understood. The immunosuppressive effects of azathioprine involve a greater suppression of delayed hypersensitivity and cellular cytotoxicity tests than of antibody responses. Azathioprine antagonizes purine metabolism and may inhibit synthesis of DNA, RNA, and proteins; it may also interfere with cellular metabolism and inhibit mitosis., For more Mechanism of Action (Complete) data for AZATHIOPRINE (6 total), please visit the HSDB record page. | |
| Record name | Azathioprine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00993 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | AZATHIOPRINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7084 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Pale yellow crystals from 50% aq acetone | |
CAS No. |
446-86-6 | |
| Record name | AZATHIOPRINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19837 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Azathioprine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=446-86-6 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Azathioprine [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000446866 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Azathioprine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00993 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | azathioprine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=755900 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | azathioprine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=39084 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Azathioprine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4020119 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Azathioprine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.006.525 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | AZATHIOPRINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/MRK240IY2L | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | AZATHIOPRINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7084 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Azathioprine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015128 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
469 to 471 °F (decomposes) (NTP, 1992), dec 243-244 °C, 243.5 °C | |
| Record name | AZATHIOPRINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19837 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Azathioprine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00993 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | AZATHIOPRINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7084 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Azathioprine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015128 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














