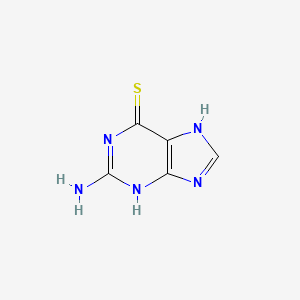
6-Thioguanine
Overview
Description
Preparation Methods
Synthetic Routes and Reaction Conditions
Tioguanine can be synthesized using 2,9-diacetyl guanine or 2-acetyl guanine as starting materials. The reaction involves sulfidizing these compounds with phosphorus pentasulfide in a pyridine solution, with a slight catalyst . This method offers mild reaction conditions, simple operation, and a high yield of up to 75% .
Industrial Production Methods
The industrial production of tioguanine follows similar synthetic routes but on a larger scale. The process involves the use of thiophosphoric anhydride and pyridine solution, with careful control of reaction conditions to ensure high yield and purity .
Chemical Reactions Analysis
Types of Reactions
Tioguanine undergoes various chemical reactions, including:
Deamination: The deamination reaction of tioguanine with water, hydroxide ions, and protonated forms has been studied extensively.
Oxidation: Tioguanine can be oxidized to form toxic metabolites, contributing to its cytotoxic effects.
Common Reagents and Conditions
Major Products Formed
Scientific Research Applications
Pharmacological Profile
Mechanism of Action
6-Thioguanine functions as an antimetabolite, primarily inhibiting DNA synthesis by incorporating into nucleic acids, particularly DNA. This incorporation disrupts the replication process, leading to cell death in rapidly dividing tumor cells. Evidence indicates that 6-TG is more effective in younger, rapidly growing tumor populations compared to older ones, highlighting the importance of timing in treatment regimens .
Pharmacokinetics
The pharmacokinetics of 6-TG have been extensively studied. It is metabolized by guanase and xanthine oxidase, with higher blood levels observed post-intravenous administration compared to oral dosing. Studies show that repeated doses lead to significant incorporation of 6-TG into DNA, particularly in bone marrow cells .
Clinical Applications
Cancer Treatment
this compound has been utilized in various types of cancer, particularly hematological malignancies such as acute leukemia and lymphomas. Its efficacy has been noted in patients with specific genetic profiles, particularly those with homozygous deletions of the methylthioadenosine phosphorylase (MTAP) gene. These tumors are often resistant to other therapies but may respond favorably to 6-TG when combined with methylthioadenosine (MTA), which protects normal tissues from toxicity while allowing higher doses of 6-TG .
Case Study: Leukemia Treatment
A retrospective study involving 30 patients with Crohn's Disease who failed conventional immunosuppression demonstrated the efficacy of 6-TG as a third-line treatment. The median dose was 40 mg daily over 21.5 months, with an initial clinical response in 60% of patients . This study underscores the drug's potential beyond oncology into inflammatory conditions.
Combination Therapies
Research suggests that combining this compound with other agents can enhance its therapeutic effects. For instance, combinations with methotrexate or pralatrexate have shown promising results against MTAP-deficient tumors. The synergistic effects observed in xenograft models indicate a potential for developing more effective treatment protocols .
Safety and Toxicity
While 6-TG is effective, it is not without risks. Hepatotoxicity remains a concern, particularly in long-term use. In the previously mentioned study on Crohn's Disease patients, some experienced abnormal liver function tests during treatment; however, these were mostly mild and transient . Monitoring liver function during therapy is crucial for patient safety.
Summary Table of Applications
| Application Area | Details |
|---|---|
| Cancer Types | Acute leukemia, lymphomas, solid tumors (e.g., mesothelioma, melanoma) |
| Mechanism | Incorporation into DNA/RNA leading to growth arrest and apoptosis |
| Combination Potential | Enhanced effects when used with MTA or other chemotherapeutics |
| Safety Concerns | Hepatotoxicity; requires monitoring during treatment |
Mechanism of Action
Tioguanine exerts its effects by competing with hypoxanthine and guanine for the enzyme hypoxanthine-guanine phosphoribosyltransferase (HGPRTase). It is converted to 6-thioguanilyic acid, which reaches high intracellular concentrations at therapeutic doses . The compound is incorporated into DNA during the synthesis phase, leading to cytotoxic effects . Additionally, tioguanine inhibits the GTP-binding protein Rac1, which regulates the Rac/Vav pathway .
Comparison with Similar Compounds
Tioguanine is part of the thiopurine family, which includes mercaptopurine and azathioprine . Compared to these compounds, tioguanine has a higher efficacy and faster action . its use is limited by concerns about hepatotoxicity and other side effects .
Similar Compounds
Mercaptopurine: Another purine analogue used in the treatment of leukemia and other conditions.
Azathioprine: A prodrug of mercaptopurine, used as an immunosuppressant in organ transplantation and autoimmune diseases.
Tioguanine’s unique properties, such as its incorporation into DNA and inhibition of specific molecular pathways, make it a valuable compound in both research and clinical settings .
Biological Activity
6-Thioguanine (6-TG) is a purine analog that has been utilized in the treatment of various hematological malignancies, particularly acute myeloid leukemia (AML). Since its approval in the 1960s, research has expanded to explore its biological activity beyond oncology, revealing significant effects in conditions like diabetic retinopathy and psoriasis. This article delves into the biological activity of 6-TG, supported by case studies, data tables, and comprehensive research findings.
6-TG exerts its effects primarily through the inhibition of purine metabolism and DNA synthesis. It gets incorporated into DNA, replacing guanine, which leads to the disruption of DNA replication and ultimately induces apoptosis in rapidly dividing cells, such as cancer cells and activated lymphocytes. This mechanism is particularly relevant in the treatment of leukemias and autoimmune diseases.
Key Mechanisms:
- Inhibition of DNA synthesis : 6-TG is metabolized to thioguanine nucleotides (TGNs), which interfere with DNA replication.
- Induction of apoptosis : Primarily observed in activated T lymphocytes, where it triggers programmed cell death.
- Immunosuppressive effects : Reduction in peripheral blood lymphocytes and total leukocyte counts.
1. Treatment of Psoriasis
A notable study involving 20 patients with moderate to severe plaque-type psoriasis demonstrated that 6-TG significantly improved disease severity. After six months of treatment:
- 12 patients were completely cleared of disease.
- 6 showed marked improvement .
- 2 did not respond .
Biopsy results indicated a reduction in keratinocyte proliferation and inflammation, correlating with clinical improvement. The therapy also led to a significant decrease in circulating lymphocytes by an average of 42% after two months .
2. Anti-Angiogenic Activity in Diabetic Retinopathy
Recent research highlighted the anti-angiogenic properties of 6-TG in diabetic retinopathy models. In vitro studies with human umbilical vein endothelial cells (HUVECs) exposed to high glucose levels showed:
- Marked reduction in tube formation at concentrations as low as 5 µM .
- In vivo studies demonstrated that diabetic mice treated with 6-TG exhibited reduced retinal vascular alterations, as confirmed by fluorescein angiography .
3. Efficacy in Leukemia Treatment
Comparative studies have shown that 6-TG is more effective than its counterpart, 6-mercaptopurine, particularly in reducing the risk of isolated central nervous system relapse. However, it carries a higher risk of severe adverse effects, including veno-occlusive disease .
Toxicity and Side Effects
While 6-TG has demonstrated efficacy across various conditions, its use is associated with several toxicities:
- Risk of infections due to immunosuppression.
- Development of liver complications such as veno-occlusive disease.
- Mutagenic potential leading to G→A mutations .
Summary Table: Clinical Findings
| Condition | Response Rate | Key Findings |
|---|---|---|
| Psoriasis | 90% | Significant reduction in skin lesions |
| Diabetic Retinopathy | N/A | Reduced retinal vascular changes |
| Childhood Leukemia | N/A | Lower CNS relapse risk but higher mortality |
Research Findings
A variety of studies have investigated the pharmacokinetics and biological effects of 6-TG:
- Pharmacokinetics : Blood levels were higher post-intravenous administration compared to oral doses, indicating better bioavailability and incorporation into DNA .
- Mutagenicity Studies : Research indicated that both 6-TG and its metabolites are mutagenic in human cell lines, raising concerns about long-term use .
- Immunological Effects : A decrease in circulating leukocytes was noted alongside clinical improvements in autoimmune conditions .
Q & A
Basic Research Questions
Q. How are 6-thioguanine nucleotide (6-TGN) levels quantified in erythrocytes, and what methodological variations affect clinical correlations?
- Methodology : 6-TGN levels are typically measured via high-performance liquid chromatography (HPLC) after acid hydrolysis of erythrocyte lysates. Key variations include hydrolysis conditions (e.g., 1 M perchloric acid vs. 0.5 M sulfuric acid) and reducing agents (e.g., dithiothreitol concentration), which impact nucleotide-to-base conversion efficiency . Standardization is critical, as differences in protocols lead to 2.6-fold discrepancies in reported 6-TGN levels, complicating cross-study comparisons .
Q. What experimental approaches are used to identify tautomeric forms of 6-TG in photochemical studies?
- Methodology : Resonance-enhanced multiphoton ionization (REMPI) and pump-probe spectroscopy are employed to study 6-TG's excited-state dynamics. Jet-cooled 6-TG samples allow differentiation of thiol tautomers, revealing distinct photophysical behaviors (e.g., lifetimes, energy transfer pathways) . IR-UV double resonance spectroscopy further resolves tautomer-specific vibrational modes .
Q. How is 6-TG incorporated into preclinical models to assess cytotoxicity and mutagenicity?
- Methodology : In vitro assays use cell lines (e.g., HCT116 colorectal cancer cells) to quantify apoptosis (via Annexin V/PI staining) and autophagy (LC3-II Western blotting). Mutagenicity is evaluated in V79 Chinese hamster lung fibroblasts using clonogenic survival and HPRT gene mutation assays . Dosing protocols often involve 24–72 hr exposures to 0.1–10 µM 6-TG .
Advanced Research Questions
Q. How do discrepancies in 6-TGN assay methodologies impact meta-analyses of inflammatory bowel disease (IBD) outcomes?
- Methodology : Heterogeneity in meta-analyses (e.g., pooled odds ratio [OR] = 3.3 in vs. OR = 3.15 in ) arises from inconsistent hydrolysis protocols and thresholds (230–260 pmol/8×10⁸ RBC). Studies using the Lennard reference method (acid hydrolysis + DTT) show reduced variability, emphasizing the need for standardized metabolite quantification .
Q. What CRISPR-based screening strategies identify genes conferring 6-TG resistance?
- Methodology : Genome-wide sgRNA libraries (e.g., pooled lentiviral constructs) are transduced into 4T1-luc or similar cell lines. Post-6-TG treatment, next-generation sequencing (NGS) of surviving cells reveals enriched sgRNAs targeting pathways like purine metabolism (IMPDH1/2) or DNA repair (MSH2) . Data analysis tools (e.g., MAGeCK) rank candidate resistance genes .
Q. How does 6-TG induce DNA demethylation, and what structural mechanisms underlie its interaction with methyltransferases?
- Methodology : 6-TG incorporates into DNA, forming S6-methylthioguanine adducts via SAM-dependent methylation. These adducts disrupt DNMT1 binding, as shown in crystallography studies. Demethylation is quantified via bisulfite sequencing in HCT116 cells treated with 1–5 µM 6-TG for 48 hr .
Q. What role do disulfide crosslinks play in 6-TG’s DNA interaction, and how are they characterized structurally?
- Methodology : X-ray crystallography of DNA duplexes with consecutive 6-TG residues reveals kinked helices at disulfide-bonded sites (6-thioG-6-thioG). Redox conditions (e.g., Cu²⁺/glutathione) reversibly modulate crosslinking, assessed via gel electrophoresis and circular dichroism .
Q. Data Contradiction and Resolution
Q. How can researchers resolve conflicting reports on 6-TG’s association with clinical remission in IBD?
- Methodology : Stratify studies by assay method (e.g., Lennard vs. Dervieux-Boulieu protocols) to isolate technical variability. Sensitivity analyses excluding outliers (e.g., studies with non-standard thresholds) reduce heterogeneity (I² from 75% to 30%) . Pooled ORs from homogenized subgroups provide more reliable effect estimates .
Q. Safety and Handling
Q. What precautions are critical for handling 6-TG in laboratory settings?
- Methodology : Use local exhaust ventilation and PPE (nitrile gloves, N95 masks) to avoid inhalation/contact. Store at 4°C in airtight, light-resistant containers. Decontaminate waste with 10% sodium hypochlorite before disposal .
Properties
IUPAC Name |
2-amino-3,7-dihydropurine-6-thione | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C5H5N5S/c6-5-9-3-2(4(11)10-5)7-1-8-3/h1H,(H4,6,7,8,9,10,11) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
WYWHKKSPHMUBEB-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1=NC2=C(N1)C(=S)N=C(N2)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C5H5N5S | |
| Record name | 6-THIOGUANINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21107 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
5580-03-0 (hemihydrate), 76078-67-6 (mono-Na salt) | |
| Record name | Thioguanine [USAN:USP] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000154427 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID6023652 | |
| Record name | 6-Thioguanine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6023652 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
167.19 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
6-thioguanine appears as odorless or almost odorless pale yellow crystalline powder. (NTP, 1992), Solid | |
| Record name | 6-THIOGUANINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21107 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Thioguanine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014496 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
less than 1 mg/mL at 73 °F (NTP, 1992), INSOL IN WATER, ALCOHOL, CHLOROFORM; FREELY SOL IN DIL SOLUTIONS OF ALKALI HYDROXIDES, 8.34e-01 g/L, Readily soluble in dilute aqueous alkali. Insoluble in water, alcohol, or chloroform. | |
| Record name | 6-THIOGUANINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21107 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Tioguanine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00352 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | THIOGUANINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/2504 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Thioguanine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014496 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
| Record name | THIOGUANINE | |
| Source | NCI Investigational Drugs | |
| URL | http://www.cancer.gov/about-cancer/treatment/drugs/investigational-drug-access-fact-sheet | |
| Description | An investigational drug is one that is under study but does not have permission from the U.S. Food and Drug Administration (FDA) to be legally marketed and sold in the United States. NCI provides the investigational drug to the physicians who are participating in clinical trials or TRC protocols. For more information please visit NCI investigational drug website: https://www.cancer.gov/about-cancer/treatment/drugs/investigational-drug-access-fact-sheet | |
Mechanism of Action |
Thioguanine competes with hypoxanthine and guanine for the enzyme hypoxanthine-guanine phosphoribosyltransferase (HGPRTase) and is itself converted to 6-thioguanilyic acid (TGMP), which reaches high intracellular concentrations at therapeutic doses. TGMP interferes with the synthesis of guanine nucleotides by its inhibition of purine biosynthesis by pseudofeedback inhibition of glutamine-5-phosphoribosylpyrophosphate amidotransferase, the first enzyme unique to the de novo pathway of purine ribonucleotide synthesis. TGMP also inhibits the conversion of inosinic acid (IMP) to xanthylic acid (XMP) by competition for the enzyme IMP dehydrogenase. Thioguanine nucleotides are incorporated into both the DNA and the RNA by phosphodiester linkages, and some studies have shown that incorporation of such false bases contributes to the cytotoxicity of thioguanine. Its tumor inhibitory properties may be due to one or more of its effects on feedback inhibition of de novo purine synthesis; inhibition of purine nucleotide interconversions; or incorporation into the DNA and RNA. The overall result of its action is a sequential blockade of the utilization and synthesis of the purine nucleotides., THIOGUANINE, THE 6-THIO ANALOGUE OF GUANINE, IS A PRODRUG THAT IS CONVERTED TO 6-THIOGUANINE-RIBOSE-PHOSPHATE, AN ACTIVE METABOLITE. ... 6-THIOGUANINE-RIBOSE-PHOSPHATE IS A FEEDBACK INHIBITOR OF THE INITIAL (AMIDOTRANSFERASE) STEP IN PURINE BIOSYNTHESIS. THIS METABOLITE ALSO BLOCKS CONVERSIONS OF INOSINIC ACID TO GUANYLIC ACID & OF GUANYLIC ACID TO GDP. THIOGUANINE ALSO IS CONVERTED TO THE DEOXYNUCLEOTIDE TRIPHOSPHATE, WHICH CAN BE INCORPORATED INTO TUMOR CELL DNA. ALTHOUGH SOME INVESTIGATORS BELIEVE THAT THIS IS THE MAJOR MECHANISM OF CYTOTOXICITY, THE RELATIVE IMPORTANCE OF THE VARIOUS SITES OF ACTION HAS NOT BEEN DETERMINED. ... THIOGUANINE IS CELL-CYCLE SPECIFIC FOR THE S PHASE., APPARENTLY, THE METABOLISM OF 6-THIOGUANINE TO 6-THIOGUANOSINE IN SARCOMA 180 & 180/TG CELLS IS MEDIATED BY PURINE NUCLEOSIDE PHOSPHORYLASE & NEWLY SYNTHESIZED 6-THIOGUANOSINE IS READILY EFFLUXED INTO THE CELLULAR ENVIRONMENT. THE FORMATION OF 6-THIOGUANINE, BY PURINE NUCLEOSIDE PHOSPHORYLASE, MAY BE IMPORTANT TO THE EXPRESSION OF CELLULAR SENSITIVITY TO 6-THIOGUANINE, IN THAT IT DECR THE AVAIL OF 6-THIOGUANINE FOR DIRECT CONVERSION BY HYPOXANTHINE-GUANINE PHOSPHORIBOSYLTRANSFERASE TO THE NUCLEOTIDE LEVEL, A PHENOMENON CRITICAL TO THE EXPRESSION OF THE ANTINEOPLASTIC ACTIVITY OF THE 6-THIOPURINES., PHENYL SUBSTITUTION AT C-8 OF 2-AMINO-6-MERCAPTOPURINE ABOLISHED THE IMMUNOSUPPRESSIVE ACTIVITY. THEREFORE, A NONSUBSTITUTED 8 POSITION OF 2-AMINO-6-MERCAPTOPURINE IS ESSENTIAL FOR IMMUNOSUPPRESSIVE ACTION., SPONTANEOUSLY CYCLING LYMPHOCYTES (IN CELL DIVISION IN CULTURES WITHOUT ADDITION OF PHYTOHEMAGGLUTININ, PHA) GO THROUGH VARIOUS PHASES OF 1ST DIVISION WITH THE SAME KINETICS AS PHA-STIMULATED CELLS. IN SAMPLES FROM 10 REFERENTS, THE FREQUENCY OF SPONTANEOUSLY CYCLING LYMPHOCYTES VARIED FROM 8.9X10-5 TO 9.5X10-3 AS INDICATED WITH AUTORADIOGRAPHY ON CELLS IN (S + G2) PHASE DETERMINED BY FLOW SORTING. IN PHA-STIMULATED SAMPLES FROM THE SAME PERSONS THE FREQUENCY OF 6-THIOGUANINE (TG)-RESISTANT VARIANTS WAS BETWEEN 4X10-7 & 2.6X10-6, WHICH INDICATES THAT MOST OF THE SPONTANEOUSLY CYCLING CELLS WERE TG-SENSITIVE., For more Mechanism of Action (Complete) data for THIOGUANINE (7 total), please visit the HSDB record page. | |
| Record name | Tioguanine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00352 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | THIOGUANINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/2504 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
NEEDLES FROM WATER, YELLOW, CRYSTALLINE POWDER | |
CAS No. |
154-42-7 | |
| Record name | 6-THIOGUANINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21107 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Thioguanine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=154-42-7 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Thioguanine [USAN:USP] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000154427 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Tioguanine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00352 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | thioguanine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757348 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 6-Thioguanine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=752 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 6-Thioguanine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6023652 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Tioguanine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.005.299 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | THIOGUANINE ANHYDROUS | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/WIX31ZPX66 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | THIOGUANINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/2504 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Thioguanine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014496 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
greater than 680 °F (NTP, 1992), >360 °C, GREATER THAN 360 °C, > 360 °C | |
| Record name | 6-THIOGUANINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/21107 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Tioguanine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00352 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | THIOGUANINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/2504 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Thioguanine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014496 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















