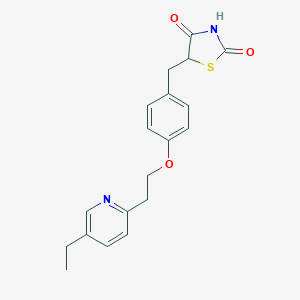
Pioglitazone
Overview
Description
Pioglitazone is a pharmaceutical compound belonging to the thiazolidinedione class, primarily used as an anti-diabetic medication to treat type 2 diabetes mellitus. It works by improving the sensitivity of tissues to insulin, thereby helping to control blood sugar levels. This compound is often used in combination with other anti-diabetic medications such as metformin, sulfonylureas, or insulin .
Preparation Methods
Synthetic Routes and Reaction Conditions
The synthesis of pioglitazone involves several steps, starting with the preparation of the thiazolidinedione ring. One common method involves the reaction of 2,4-thiazolidinedione with benzyl chloride in the presence of a base to form the intermediate compound. This intermediate is then reacted with 2-(5-ethylpyridin-2-yl)ethanol under basic conditions to yield this compound .
Industrial Production Methods
Industrial production of this compound typically involves the hydrogenation of an acid addition salt of a benzylidene compound under low hydrogen gas pressure. This process uses a reducing agent to achieve high yields of the thiazolidinedione derivative .
Chemical Reactions Analysis
Synthetic Reactions
Pioglitazone is synthesized via multi-step processes involving condensation, hydrogenation, and salt formation. Two optimized pathways are highlighted:
Pathway 2: Catalytic Hydrogenation (WO2005058827A1)
- A novel intermediate, 5-[[4-[2-(5-ethyl-2-pyridinyl)ethoxy]phenyl]methylene]-2,4-thiazolidinedione hydrochloride, undergoes catalytic hydrogenation in aqueous methanol with Pd/C (5–6 bar H₂, 50–60°C).
- Yield : 84% with >99.7% purity .
Table 1: Comparison of Synthetic Methods
| Parameter | Pathway 1 | Pathway 2 |
|---|---|---|
| Key Catalyst | Pd/C | Pd/C |
| Solvent | Methanol/DMF | Aqueous methanol |
| Hydrogen Pressure | ≤30 kg/cm² | 5–6 bar |
| Final Purity | ~97.7% | >99.7% |
| Yield | 63–73% (base) → 80–85% (HCl) | 84% (direct HCl salt) |
Metabolic Reactions
This compound is metabolized primarily in the liver via CYP2C8 and CYP3A4 enzymes, producing active metabolites:
Phase I Metabolism
- Hydroxylation : Formation of M-III (hydroxy-pioglitazone) and M-IV (keto-pioglitazone).
- Oxidation : Generation of sulfoxide and N-oxide derivatives.
Phase II Metabolism
Table 2: Key Metabolites and Enzymatic Pathways
| Metabolite | Enzyme Involved | Bioactivity |
|---|---|---|
| M-III | CYP2C8 | 60–80% activity of parent |
| M-IV | CYP3A4/CYP2C8 | Full PPARγ agonism |
| M-V | CYP1A1 (minor) | Inactive |
Degradation and Impurity Formation
This compound degrades under oxidative and hydrolytic conditions, leading to impurities identified via UHPLC-HRMS:
Common Degradation Pathways
- Oxidation : Formation of N-oxide and sulfone derivatives.
- Hydrolysis : Cleavage of the thiazolidinedione ring under acidic/alkaline conditions.
- Dimerization : Formation of dimeric impurities via radical intermediates .
Table 3: Identified Impurities in this compound Preparations
| Impurity | Structure | Origin |
|---|---|---|
| Impurity-1 | 5-Ethylpyridine sulfoxide | Oxidative degradation |
| Impurity-2 | Benzylidene dimer | Photolytic dimerization |
| Impurity-3 | Des-ethyl this compound | Incomplete synthesis |
Reaction with Excipients and Stabilizers
- PLGA Nanoparticles : this compound encapsulated in poly(lactic-co-glycolic acid) nanoparticles via nanoprecipitation shows controlled release kinetics (85% drug release in 72 hours) .
- HCl Salt Formation : Enhances solubility and stability; crystallized from ethanol/acetic acid mixtures .
Toxicological Byproducts
Scientific Research Applications
Type 2 Diabetes Management
Pioglitazone is indicated for patients with T2DM, particularly those whose blood glucose levels are inadequately controlled with other medications. It is often used in combination with metformin or sulfonylureas.
- Efficacy : In the PROactive study involving 5,238 patients, this compound demonstrated a significant reduction in secondary endpoints such as all-cause mortality and myocardial infarction compared to placebo . However, caution is advised as the primary endpoint did not reach significance.
Cardiovascular Benefits
Numerous studies have highlighted the cardiovascular protective effects of this compound.
- Mortality Reduction : An observational study indicated a 42% reduction in cardiovascular mortality among this compound users compared to those on alternative treatments .
- Atherosclerosis Progression : The CHICAGO trial showed that this compound slowed the progression of carotid intima-media thickness over 18 months compared to glimepiride .
Non-Alcoholic Fatty Liver Disease (NAFLD)
This compound has been studied for its effects on NAFLD and non-alcoholic steatohepatitis (NASH).
- Pleiotropic Effects : A randomized controlled trial found that this compound significantly improved liver histology in patients with NASH, suggesting its potential role in managing liver-related metabolic disorders .
Case Study 1: Cardiovascular Risk Reduction
In a cohort study analyzing over 10,000 patients with T2DM, those treated with this compound exhibited lower rates of heart failure and cardiovascular events compared to those treated with other antidiabetic agents. This study supports the hypothesis that this compound may confer additional cardiovascular benefits beyond glycemic control.
Case Study 2: Impact on Liver Function
A clinical trial involving patients diagnosed with NASH showed that treatment with this compound resulted in significant improvements in liver enzyme levels and histological features after 24 weeks of therapy. These findings underscore the drug's potential utility in treating liver complications associated with metabolic syndrome.
Table 1: Summary of Clinical Trials Involving this compound
| Study Name | Population | Duration | Primary Endpoint | Key Findings |
|---|---|---|---|---|
| PROactive | 5,238 T2DM patients | 3 years | All-cause mortality | Significant reduction in myocardial infarction and stroke |
| CHICAGO | 462 T2DM patients | 18 months | Carotid intima-media thickness | Slowed progression compared to glimepiride |
| NASH Trial | NASH patients | 24 weeks | Liver histology improvement | Significant improvement in liver enzymes |
Mechanism of Action
Pioglitazone acts as a selective agonist at the peroxisome proliferator-activated receptor-gamma (PPARγ) in target tissues such as adipose tissue, skeletal muscle, and liver. Activation of PPARγ increases the transcription of insulin-responsive genes involved in glucose and lipid metabolism. This leads to improved insulin sensitivity, enhanced glucose uptake, and better glycemic control .
Comparison with Similar Compounds
Similar Compounds
Rosiglitazone: Another thiazolidinedione with similar insulin-sensitizing effects but associated with higher cardiovascular risks.
Metformin: A non-thiazolidinedione anti-diabetic medication that improves insulin sensitivity but through a different mechanism.
Semaglutide: A glucagon-like peptide-1 receptor agonist that lowers blood sugar levels by stimulating insulin secretion.
Uniqueness of Pioglitazone
This compound is unique in its ability to improve lipid profiles by increasing high-density lipoprotein cholesterol and reducing triglycerides. It also has a favorable effect on urinary albumin/creatinine ratio, indicating potential renal benefits . Despite its benefits, this compound is associated with risks such as weight gain and heart failure, which need to be carefully managed .
Biological Activity
Pioglitazone is a thiazolidinedione class medication primarily used for the management of type 2 diabetes mellitus. Its biological activity encompasses various mechanisms that contribute to its therapeutic effects, including insulin sensitization, anti-inflammatory properties, and potential neuroprotective effects. This article delves into the biological activity of this compound, supported by relevant research findings, case studies, and data tables.
1. Insulin Sensitization
This compound acts primarily through the activation of peroxisome proliferator-activated receptor gamma (PPAR-γ), a nuclear receptor that regulates gene expression involved in glucose and lipid metabolism. This activation enhances insulin sensitivity in peripheral tissues, particularly in adipose tissue and skeletal muscle.
- Study Findings : A randomized trial demonstrated that this compound significantly increased insulin-stimulated glucose disposal by 30% and elevated plasma adiponectin levels by 79% after six months of treatment in patients with type 2 diabetes .
2. Anti-inflammatory Effects
This compound exhibits anti-inflammatory properties that may contribute to its cardiovascular benefits. It reduces levels of inflammatory markers such as C-reactive protein (CRP) and interleukin-6 (IL-6).
- Clinical Trial Data : In the PROactive study, patients treated with this compound showed a reduction in macrovascular events, suggesting that its anti-inflammatory effects may play a role in cardiovascular protection .
3. Antibacterial Activity
Recent studies have indicated that this compound possesses antibacterial activity against certain Gram-positive bacteria. This effect appears to be dose-dependent and may involve mechanisms unrelated to PPAR-γ activation, such as the generation of reactive oxygen species (ROS) which enhance the efficacy of common antibiotics .
Case Studies
1. PROactive Study
The PROactive study was a landmark clinical trial assessing the impact of this compound on macrovascular events in patients with type 2 diabetes. It involved over 5,000 participants and demonstrated that this compound significantly reduced the risk of major cardiovascular events compared to placebo.
| Parameter | This compound Group | Placebo Group | p-value |
|---|---|---|---|
| Major Cardiovascular Events | 14% | 17% | <0.05 |
| All-Cause Mortality | 6% | 8% | <0.05 |
This study highlights this compound's potential beyond glycemic control, emphasizing its role in cardiovascular health.
2. Neuroprotective Effects
A pilot clinical trial explored the safety and efficacy of this compound in patients with mild Alzheimer's disease (AD). The results indicated that this compound could penetrate the blood-brain barrier and exert beneficial effects on AD-related pathology.
| Outcome Measure | This compound Group | Control Group | p-value |
|---|---|---|---|
| ADAS-COG Score | -0.746 | -0.354 | <0.05 |
| CDR-SB Score | -0.354 | -0.150 | <0.05 |
These findings suggest that this compound may offer neuroprotective benefits, warranting further investigation into its use in neurodegenerative diseases .
Research Findings
1. Lipid Profile Improvement
This compound has been shown to positively influence lipid profiles in patients with type 2 diabetes, leading to significant reductions in triglycerides and increases in HDL cholesterol levels.
- Clinical Trial Results : In a six-month randomized controlled trial, patients receiving this compound exhibited mean decreases in triglycerides ranging from 39.1 to 65.3 mg/dL compared to placebo .
2. Activation of AMPK Pathway
Research indicates that this compound activates the AMP-activated protein kinase (AMPK) pathway, which plays a crucial role in cellular energy homeostasis and metabolism.
Q & A
Basic Research Questions
Q. What is the primary mechanism of action of pioglitazone, and how can it be experimentally validated?
this compound acts as a peroxisome proliferator-activated receptor gamma (PPAR-γ) agonist, enhancing insulin sensitivity by modulating adipocyte differentiation and lipid metabolism. To validate this, researchers can:
- Perform in vitro ligand-binding assays to confirm PPAR-γ activation .
- Use gene expression profiling in adipose tissue to track downstream targets like adiponectin .
- Conduct glucose clamp studies in animal models to quantify insulin sensitivity improvements .
Q. How should a clinical trial investigating this compound’s efficacy in type 2 diabetes mellitus (T2DM) be designed?
A robust trial design should incorporate:
- PICOT framework : Define Population (e.g., T2DM patients with insulin resistance), Intervention (this compound dosage), Comparison (placebo or standard care), Outcome (HbA1c reduction), and Time (6–12 months) .
- Stratified randomization to control for covariates like baseline HbA1c and comorbidities .
- Blinding and placebo-controlled protocols to minimize bias .
Q. What biomarkers are critical for assessing this compound’s metabolic effects?
Key biomarkers include:
- Glycemic control : HbA1c, fasting plasma glucose .
- Lipid metabolism : Triglycerides, HDL cholesterol .
- Inflammation : C-reactive protein (CRP), adiponectin levels .
- Hepatic insulin sensitivity : Measured via hyperinsulinemic-euglycemic clamp .
Advanced Research Questions
Q. How can conflicting data on this compound’s association with cancer risks be resolved?
Contradictory findings (e.g., prostate vs. bladder cancer risks) require:
- Network pharmacology : Use STRING and WebGestalt to map this compound’s target pathways (e.g., PPAR-γ, NF-κB) and correlate with oncogenic pathways .
- Genomic analysis : Query cBioPortal for cancer genomics data (e.g., mutations in PPAR-γ-associated genes) .
- Propensity score matching : Adjust for confounders like age, smoking, and diabetes duration in observational studies .
Q. What methodological approaches are optimal for studying this compound’s non-glycemic effects (e.g., on nonalcoholic steatohepatitis, NASH)?
- Histological validation : Liver biopsies to assess steatosis, ballooning necrosis, and fibrosis .
- Imaging : Magnetic resonance spectroscopy (MRS) for hepatic fat quantification .
- Multi-omics integration : Combine transcriptomic data (PPAR-γ targets) with metabolomic profiling of lipid species .
Q. How can researchers address this compound’s cardiovascular risk paradox (benefits vs. side effects like edema)?
- Subgroup analysis : Stratify patients by baseline cardiovascular risk (e.g., Framingham score) .
- Mechanistic studies : Investigate PPAR-γ’s role in fluid retention via renal sodium reabsorption assays .
- Real-world data : Use EHR-linked cohorts to track long-term outcomes like heart failure hospitalization .
Q. Methodological and Analytical Considerations
Q. What statistical methods are recommended for analyzing this compound’s dose-response relationships?
- Non-linear mixed-effects modeling : For pharmacokinetic/pharmacodynamic (PK/PD) data .
- Cox proportional hazards models : For time-to-event outcomes (e.g., cancer incidence) .
- Meta-regression : To explore heterogeneity in systematic reviews (e.g., variable follow-up durations) .
Q. How should confounding factors be controlled in observational studies on this compound?
- Propensity score matching : Balance covariates like age, BMI, and diabetes severity .
- Sensitivity analyses : Exclude high-risk subgroups (e.g., patients with prior cancer) .
- Instrumental variable methods : Address unmeasured confounders using genetic or geographic proxies .
Q. What experimental models are suitable for studying this compound’s neurovascular effects?
- Animal models : Fructose-fed rats to mimic insulin resistance and assess this compound’s impact on angiotensin II-induced vasoconstriction .
- Ex vivo assays : Isolated vessel preparations to measure endothelial function .
Q. Data Management and Reproducibility
Q. How can researchers ensure reproducibility in this compound-related pharmacokinetic studies?
- HPLC validation : Follow ICH guidelines for quantifying this compound and its metabolites (e.g., column temperature: 30°C, mobile phase: acetonitrile-phosphate buffer) .
- Open-data practices : Share raw chromatograms and calibration curves via repositories like Zenodo .
Q. What are best practices for curating this compound-related research data?
- FAIR principles : Ensure data are Findable, Accessible, Interoperable, and Reusable .
- Structured metadata : Include experimental conditions (e.g., dosage, cohort demographics) and assay protocols .
Properties
IUPAC Name |
5-[[4-[2-(5-ethylpyridin-2-yl)ethoxy]phenyl]methyl]-1,3-thiazolidine-2,4-dione | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C19H20N2O3S/c1-2-13-3-6-15(20-12-13)9-10-24-16-7-4-14(5-8-16)11-17-18(22)21-19(23)25-17/h3-8,12,17H,2,9-11H2,1H3,(H,21,22,23) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
HYAFETHFCAUJAY-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCC1=CN=C(C=C1)CCOC2=CC=C(C=C2)CC3C(=O)NC(=O)S3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C19H20N2O3S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID3037129 | |
| Record name | Pioglitazone | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3037129 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
356.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Pioglitazone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015264 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Practically insoluble, 4.42e-03 g/L | |
| Record name | Pioglitazone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01132 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Pioglitazone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015264 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Pioglitazone is a selective agonist at peroxisome proliferator-activated receptor-gamma (PPARγ) in target tissues for insulin action such as adipose tissue, skeletal muscle, and liver. Activation of PPARγ increases the transcription of insulin-responsive genes involved in the control of glucose and lipid production, transport, and utilization. Through this mechanism, pioglitazone both enhances tissue sensitivity to insulin and reduces the hepatic production of glucose (i.e. gluconeogenesis) - insulin resistance associated with type 2 diabetes mellitus is therefore improved without an increase in insulin secretion by pancreatic beta cells., Repeated administration of peroxisome proliferator-activated receptor gamma (PPARgamma) agonists reduces neuropathic pain-like behavior and associated changes in glial activation in the spinal cord dorsal horn. As PPARgamma is a nuclear receptor, sustained changes in gene expression are widely believed to be the mechanism of pain reduction. However, we recently reported that a single intrathecal (i.t.) injection of pioglitazone, a PPARgamma agonist, reduced hyperalgesia within 30 minutes, a time frame that is typically less than that required for genomic mechanisms. To determine the very rapid antihyperalgesic actions of PPARgamma activation, we administered pioglitazone to rats with spared nerve injury and evaluated hyperalgesia. Pioglitazone inhibited hyperalgesia within 5 minutes of injection, consistent with a nongenomic mechanism. Systemic or i.t. administration of GW9662, a PPARgamma antagonist, inhibited the antihyperalgesic actions of intraperitoneal or i.t. pioglitazone, suggesting a spinal PPAR?-dependent mechanism. To further address the contribution of nongenomic mechanisms, we blocked new protein synthesis in the spinal cord with anisomycin. When coadministered intrathecally, anisomycin did not change pioglitazone antihyperalgesia at an early 7.5-minute time point, further supporting a rapid nongenomic mechanism. At later time points, anisomycin reduced pioglitazone antihyperalgesia, suggesting delayed recruitment of genomic mechanisms. Pioglitazone reduction of spared nerve injury-induced increases in GFAP expression occurred more rapidly than expected, within 60 minutes. We are the first to show that activation of spinal PPARgamma rapidly reduces neuropathic pain independent of canonical genomic activity. We conclude that acute pioglitazone inhibits neuropathic pain in part by reducing astrocyte activation and through both genomic and nongenomic PPARgamma mechanisms., Pioglitazone hydrochloride is a thiazolidinedione that depends on the presence of insulin for its mechanism of action. Pioglitazone hydrochloride decreases insulin resistance in the periphery and in the liver resulting in increased insulin-dependent glucose disposal and decreased hepatic glucose output. Pioglitazone is not an insulin secretagogue. Pioglitazone is an agonist for peroxisome proliferator-activated receptor-gamma (PPARgamma). PPAR receptors are found in tissues important for insulin action such as adipose tissue, skeletal muscle, and liver. Activation of PPARgamma nuclear receptors modulates the transcription of a number of insulin responsive genes involved in the control of glucose and lipid metabolism., ... Thiazolidinediones reduce insulin resistance not only in type 2 diabetes but also in non-diabetic conditions associated with insulin resistance such as obesity. The mechanism of action involves binding to the peroxisome proliferator-activated receptor (PPAR)gamma, a transcription factor that regulates the expression of specific genes especially in fat cells but also in other tissues. It is likely that thiazolidinediones primarily act in adipose tissue where PPARgamma is predominantly expressed. Thiazolidinediones have been shown to interfere with expression and release of mediators of insulin resistance originating in adipose tissue (e.g. free fatty acids, adipocytokines such as tumor necrosis factor alpha, resistin, adiponectin) in a way that results in net improvement of insulin sensitivity (i.e. in muscle and liver). Nevertheless, a direct molecular effect in skeletal muscle cannot be excluded. ..., Pioglitazone, a full peroxisome proliferator-activated receptor (PPAR)-gamma agonist, improves insulin sensitivity by increasing circulating adiponectin levels. However, the molecular mechanisms by which pioglitazone induces insulin sensitization are not fully understood. In this study, we investigated whether pioglitazone improves insulin resistance via upregulation of either 2 distinct receptors for adiponectin (AdipoR1 or AdipoR2) expression in 3T3-L1 adipocytes. Glucose uptake was evaluated by 2-[(3)H] deoxy-glucose uptake assay in 3T3-L1 adipocytes with pioglitazone treatment. AdipoR1 and AdipoR2 mRNA expressions were analyzed by qRT-PCR. /The investigators/ first confirmed that pioglitazone significantly increased insulin-induced 2-deoxyglucose (2-DOG) uptake in 3T3-L1 adipocytes. Next, we investigated the mRNA expression and regulation of AdipoR1 and AdipoR2 after treatment with pioglitazone. Interestingly, pioglitazone significantly induced AdipoR2 expression but it did not affect AdipoR1 expression. In addition, adenovirus-mediated PPARgamma expression significantly enhanced the effects of pioglitazone on insulin-stimulated 2-DOG uptake and AdipoR2 expression in 3T3-L1 adipocytes. These data suggest that pioglitazone enhances adiponectin's autocrine and paracrine actions in 3T3-L1 adipocytes via upregulation of PPARgamma-mediated AdipoR2 expression. Furthermore, we found that pioglitazone significantly increased AMP-activated protein kinase (AMPK) phosphorylation in insulin-stimulated 3T3-L1 adipocytes, but it did not lead to the phosphorylation of IRS-1, Akt, or protein kinase ... Pioglitazone increases insulin sensitivity, at least partly, by PPARgamma-AdipoR2-mediated AMPK phosphorylation in 3T3-L1 adipocytes. In conclusion, the upregulation of AdipoR2 expression may be one of the mechanisms by which pioglitazone improves insulin resistance in 3T3-L1 adipocytes., For more Mechanism of Action (Complete) data for Pioglitazone (6 total), please visit the HSDB record page. | |
| Record name | Pioglitazone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01132 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Pioglitazone | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7322 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Colorless needles from dimethylformamide and water | |
CAS No. |
111025-46-8, 112529-15-4 | |
| Record name | Pioglitazone | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=111025-46-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Pioglitazone [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0111025468 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Pioglitazone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01132 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | pioglitazone hydrochloride | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758876 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Pioglitazone | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3037129 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 5-({4-[2-(5-ethylpyridin-2-yl)ethoxy]phenyl}methyl)-1,3-thiazolidine-2,4-dione | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.114.441 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PIOGLITAZONE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/X4OV71U42S | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Pioglitazone | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7322 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Pioglitazone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015264 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
193-194C, 183-184 °C, Colorless prisms from ethanol, MP: 193-194 °C. Soluble in dimethyl formamide; slightly soluble in ethanol; very slightly soluble in acetone, acetonitrile. Practically insoluble in water; insoluble in ether. /Pioglitazone hydrochloride/, 183 - 184 °C | |
| Record name | Pioglitazone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01132 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Pioglitazone | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7322 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Pioglitazone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015264 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details






Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














