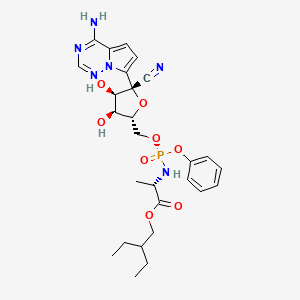
Remdesivir
Overview
Description
Preparation Methods
Synthetic Routes and Reaction Conditions
The synthesis of GS-5734 involves multiple steps, starting from commercially available starting materials. The key steps include the formation of the nucleoside analog and its subsequent phosphorylation to produce the active triphosphate form. The synthetic route typically involves protection and deprotection steps, nucleophilic substitution reactions, and phosphorylation reactions under controlled conditions .
Industrial Production Methods
Industrial production of GS-5734 follows a similar synthetic route but is optimized for large-scale manufacturing. This involves the use of high-yielding reactions, efficient purification methods, and stringent quality control measures to ensure the consistency and purity of the final product .
Chemical Reactions Analysis
Types of Reactions
GS-5734 undergoes several types of chemical reactions, including:
Oxidation: GS-5734 can be oxidized to form various metabolites.
Reduction: Reduction reactions can modify the nucleoside analog structure.
Substitution: Nucleophilic substitution reactions are involved in the synthesis of GS-5734
Common Reagents and Conditions
Common reagents used in the synthesis and reactions of GS-5734 include:
Phosphorylating agents: Used for the phosphorylation steps.
Protecting groups: Used to protect functional groups during synthesis.
Solvents: Such as dimethyl sulfoxide (DMSO) and acetonitrile, used as reaction media.
Major Products Formed
The major products formed from the reactions of GS-5734 include its active triphosphate form, remdesivir triphosphate, and various metabolites formed through oxidation and reduction reactions .
Scientific Research Applications
Efficacy in Clinical Trials
Numerous clinical trials have evaluated the efficacy of Remdesivir in treating COVID-19. Key findings from several studies are summarized below:
Safety Profile
The safety profile of this compound has been evaluated across multiple studies. Common adverse effects include hepatic impairment; however, the incidence of adverse events was similar between treated and control groups . Ongoing studies continue to monitor long-term safety and efficacy.
Case Study: Efficacy in Severe COVID-19
A systematic review involving over 40 clinical trials indicated that this compound might significantly shorten recovery time among hospitalized adults with severe COVID-19. Observational studies reported reduced mortality rates associated with this compound treatment compared to standard care .
Observational Study: Impact on Mortality Rates
A large-scale observational study involving over 121,000 patients found that this compound-treated individuals had a lower mortality rate within 14 and 28 days compared to those not receiving the drug. The adjusted hazard ratios indicated a statistically significant reduction in mortality associated with this compound use .
Regulatory Approvals and Recommendations
This compound received Emergency Use Authorization from the U.S. Food and Drug Administration for treating COVID-19 in hospitalized patients. The National Institute of Allergy and Infectious Diseases has also endorsed its use based on clinical trial data demonstrating its benefits .
Mechanism of Action
GS-5734 exerts its antiviral effects by inhibiting the RNA-dependent RNA polymerase (RdRp) enzyme, which is essential for viral replication. Once inside the host cell, GS-5734 is metabolized to its active triphosphate form, which competes with natural nucleotides for incorporation into the viral RNA. This incorporation leads to premature termination of RNA synthesis, effectively inhibiting viral replication .
Comparison with Similar Compounds
Similar Compounds
GS-441524: A parent nucleoside analog of GS-5734 with similar antiviral activity.
Favipiravir: Another nucleoside analog with broad-spectrum antiviral activity.
Ribavirin: A nucleoside analog used to treat various viral infections.
Uniqueness of GS-5734
GS-5734 is unique due to its broad-spectrum antiviral activity and its ability to inhibit a wide range of RNA viruses, including coronaviruses and filoviruses. Its efficacy against SARS-CoV-2 has made it a critical component in the fight against COVID-19 .
Biological Activity
Remdesivir, a nucleotide analog prodrug, has garnered significant attention as a therapeutic agent against viral infections, particularly COVID-19. This article delves into the biological activity of this compound, focusing on its mechanism of action, pharmacokinetics, clinical efficacy, and ongoing research.
This compound (GS-5734) is designed to inhibit viral RNA-dependent RNA polymerase (RdRp), crucial for the replication of RNA viruses. Upon entering host cells, this compound is metabolized into its active form, GS-443902, which mimics adenosine triphosphate (ATP). This active form competes with ATP for incorporation into the viral RNA strand during replication. The incorporation of GS-443902 leads to premature termination of RNA synthesis, thereby limiting viral replication.
Key Mechanisms:
- Nucleotide Analog: Resembles ATP and integrates into viral RNA.
- Chain Termination: Causes premature termination of RNA synthesis.
- Broad Spectrum Activity: Effective against various coronaviruses (SARS-CoV, MERS-CoV) and other RNA viruses like Ebola .
Pharmacokinetics
The pharmacokinetics of this compound have been studied primarily through intravenous administration. A randomized controlled trial indicated that this compound exhibits linear pharmacokinetics across various doses (3 to 225 mg). Notably, it is a substrate for organic anion transporting polypeptides (OATP1B1 and OATP1B3), influencing its hepatic uptake and elimination .
Table 1: Pharmacokinetic Profile of this compound
| Parameter | Value |
|---|---|
| Bioavailability | Not applicable (IV only) |
| Half-life | Approximately 1 hour |
| Volume of distribution | 1 L/kg |
| Clearance | 0.5 L/h/kg |
Clinical Efficacy
Several clinical trials have evaluated the efficacy of this compound in treating COVID-19. The most notable studies include:
- ACTT-1 Trial: A randomized trial demonstrating that patients receiving this compound had a shorter time to recovery compared to those receiving standard care.
- SIMPLE Trials: These trials assessed the efficacy of 5-day versus 10-day treatment regimens in hospitalized patients with severe COVID-19. Results indicated that both regimens provided similar clinical improvements .
Table 2: Summary of Key Clinical Trials
Case Studies and Observational Data
Observational studies have also provided insights into the efficacy and safety profile of this compound. For instance, a study involving hospitalized patients showed that those treated with a 5-day course had statistically significant improvements in clinical status at Day 11 compared to those receiving standard care .
Case Study Example:
A cohort study analyzed outcomes in critically ill patients treated with this compound. It found that early administration correlated with improved survival rates and reduced need for mechanical ventilation.
Ongoing Research and Future Directions
Research continues to explore the full potential of this compound beyond COVID-19. Current studies are investigating:
- Combination therapies with other antiviral agents.
- Efficacy in pediatric populations.
- Alternative formulations to enhance delivery methods.
Q & A
Basic Research Questions
Q. How should researchers design randomized controlled trials (RCTs) for Remdesivir to ensure robust evaluation of efficacy and safety in COVID-19 patients?
Methodological Answer:
- Trial Design: Use a double-blind, placebo-controlled design with allocation ratios (e.g., 2:1 for treatment vs. placebo) to maximize data collection while maintaining rigor. Primary endpoints should include time to clinical improvement (e.g., recovery time, oxygen-support class changes) and mortality rates .
- Inclusion Criteria: Define severity thresholds (e.g., oxygen saturation ≤94%, radiologically confirmed pneumonia) to standardize patient cohorts .
- Data Sources: Leverage international registries (e.g., ICTRP, ClinicalTrials.gov ) and collaborate with regulatory agencies (EMA, FDA) to ensure transparency and data accessibility .
Q. What pharmacokinetic properties of this compound are critical for determining dosing regimens in clinical studies?
Methodological Answer:
- Metabolite Analysis: Focus on the prodrug conversion to its active metabolite GS-441524. Use liquid chromatography-tandem mass spectrometry (LC-MS/MS) to quantify plasma concentrations and assess bioavailability .
- Dosing Optimization: Conduct phase 3 trials comparing 5-day vs. 10-day courses, adjusting for baseline severity. Pharmacokinetic-pharmacodynamic (PK/PD) modeling can guide individualized dosing .
- Stability Testing: Validate drug stability under varying storage conditions (e.g., temperature, pH) to ensure efficacy in clinical settings .
Q. How can researchers validate analytical methods for quantifying this compound in pharmaceutical formulations?
Methodological Answer:
- Method Development: Compare normal-phase (NP) and reversed-phase (RP) HPLC for separation efficiency. Validate parameters per ICH Q2(R1): linearity (1–50 µg/mL), precision (%RSD <2%), and accuracy (98–102%) .
- Degradation Studies: Perform forced degradation under acidic/alkaline conditions to identify stability-indicating markers .
- Cross-Validation: Transfer methods to quality control (QC) labs using inter-laboratory reproducibility tests .
Advanced Research Questions
Q. How can researchers resolve contradictions in this compound’s reported clinical efficacy across heterogeneous trials?
Methodological Answer:
- Individual Patient Data (IPD) Meta-Analysis: Pool data from RCTs (e.g., ACTT-1, SOLIDARITY) to stratify outcomes by covariates like ventilation status, age, and comorbidities. Use mixed-effects models to adjust for confounding variables .
- Bayesian Adaptive Trials: Implement Bayesian frameworks to dynamically re-estimate treatment effects as new data emerge, reducing heterogeneity in interim analyses .
- Sensitivity Analyses: Test robustness by excluding outlier studies (e.g., open-label trials) and applying GRADE criteria to assess evidence quality .
Q. What mechanisms underlie potential antiviral resistance to this compound in coronaviruses?
Methodological Answer:
- In Vitro Mutagenesis: Serial passage SARS-CoV-2 in Vero E6 cells under this compound pressure to identify mutations in RNA-dependent RNA polymerase (RdRp). Characterize viral fitness via plaque assays .
- Structural Modeling: Use cryo-EM to map RdRp-Remdesivir interactions. Identify residues (e.g., NSP12 Val557) associated with reduced binding affinity .
- Clinical Correlates: Compare viral load kinetics in responders vs. non-responders using RT-PCR and next-generation sequencing (NGS) .
Q. What advanced computational tools can predict this compound’s efficacy against emerging viral variants?
Methodological Answer:
- Deep Learning Models: Train neural networks on viral genomic data (e.g., GISAID) and biochemical profiles of this compound to predict inhibitory concentrations (IC50) for variants like Omicron .
- Molecular Dynamics (MD) Simulations: Simulate RdRp-Remdesivir binding dynamics to assess steric hindrance from variant-specific mutations (e.g., P323L) .
- Bibliometric Trend Analysis: Use Scopus/PubMed datasets to identify research gaps, such as understudied variants or combination therapies .
Q. How can researchers improve preclinical-to-clinical translation of this compound analogs?
Methodological Answer:
- Animal Models: Use transgenic mice expressing human ACE2 to evaluate lung viral load reduction and cytokine profiles. Compare with non-human primate data .
- Dose Bridging: Apply allometric scaling from animal PK data (e.g., cynomolgus monkeys) to estimate human-equivalent doses, adjusting for metabolic differences .
- Biomarker Integration: Measure interferon-stimulated genes (ISGs) in preclinical models to correlate antiviral response with clinical outcomes .
Q. Data Management and Reproducibility
Q. What strategies ensure data integrity in this compound trials during public health emergencies?
Methodological Answer:
- Centralized Data Platforms: Use platforms like WHO’s COVID-NMA to harmonize data collection across trials. Implement FAIR (Findable, Accessible, Interoperable, Reusable) principles .
- Independent Audits: Engage third-party statisticians to verify analyses, particularly for open-label studies with subjective endpoints .
- Pre-registration: Register protocols on PROSPERO or ClinicalTrials.gov to mitigate reporting bias .
Q. Tables: Key Findings from Select Studies
Properties
IUPAC Name |
2-ethylbutyl (2S)-2-[[[(2R,3S,4R,5R)-5-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxyoxolan-2-yl]methoxy-phenoxyphosphoryl]amino]propanoate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C27H35N6O8P/c1-4-18(5-2)13-38-26(36)17(3)32-42(37,41-19-9-7-6-8-10-19)39-14-21-23(34)24(35)27(15-28,40-21)22-12-11-20-25(29)30-16-31-33(20)22/h6-12,16-18,21,23-24,34-35H,4-5,13-14H2,1-3H3,(H,32,37)(H2,29,30,31)/t17-,21+,23+,24+,27-,42-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
RWWYLEGWBNMMLJ-YSOARWBDSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCC(CC)COC(=O)C(C)NP(=O)(OCC1C(C(C(O1)(C#N)C2=CC=C3N2N=CN=C3N)O)O)OC4=CC=CC=C4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCC(CC)COC(=O)[C@H](C)N[P@](=O)(OC[C@@H]1[C@H]([C@H]([C@](O1)(C#N)C2=CC=C3N2N=CN=C3N)O)O)OC4=CC=CC=C4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C27H35N6O8P | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID701022537 | |
| Record name | Remdesivir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID701022537 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
602.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
COVID-19 is caused by the positive-sense RNA virus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Replication of the viral genome is a key step in the infectious cycle of RNA viruses, including those of the _Filoviridae_, _Paramyxoviridae_, _Pneumoviridae_, and _Coronaviridae_ families, and is carried out by viral RNA-dependent RNA polymerase (RdRp) enzymes or enzyme complexes. For both SARS-CoV and SARS-CoV-2, the RdRp comprises nsp7, nsp8, and nsp12 subunits under physiological conditions, although functional RdRp complexes can be reassembled _in vitro_ that incorporate only the nsp8 and nsp12 subunits, similar to the Middle East respiratory syndrome coronavirus (MERS-CoV). Remdesivir is a phosphoramidite prodrug of a 1'-cyano-substituted adenosine nucleotide analogue that competes with ATP for incorporation into newly synthesized viral RNA by the corresponding RdRp complex. Remdesivir enters cells before being cleaved to its monophosphate form through the action of either carboxylesterase 1 or cathepsin A; it is subsequently phosphorylated by undescribed kinases to yield its active triphosphate form remdesivir triphosphate (RDV-TP or GS-443902). RDV-TP is efficiently incorporated by the SARS-CoV-2 RdRp complex, with a 3.65-fold selectivity for RDV-TP over endogenous ATP. Unlike some nucleoside analogues, remdesivir provides a free 3'-hydroxyl group that allows for continued chain elongation. However, modelling and _in vitro_ experiments suggest that at _i_ + 4 (corresponding to the position for the incorporation of the fourth nucleotide following RDV-TP incorporation), the 1'-cyano group of remdesivir sterically clashes with Ser-861 of the RdRp, preventing further enzyme translocation and terminating replication at position _i_ + 3. This mechanism was essentially identical between SARS-CoV, SARS-CoV-2, and MERS-CoV, and genomic comparisons reveal that Ser-861 is conserved across alpha-, beta-, and deltacoronaviruses, suggesting remdesivir may possess broad antiviral activity. Considerations for the use of nucleotide analogues like remdesivir include the possible accumulation of resistance mutations. Excision of analogues through the 3'-5' exonuclease (ExoN) activity of replication complexes, mediated in SARS-CoV by the nsp14 subunit, is of possible concern. Murine hepatitis viruses (MHVs) engineered to lack ExoN activity are approximately 4-fold more susceptible to remdesivir, supporting the proposed mechanism of action. However, the relatively mild benefit of ExoN activity to remdesivir resistance is proposed to involve its delayed chain termination mechanism, whereby additional endogenous nucleotides are incorporated following RDV-TP. In addition, serial passage of MHV in increasing concentrations of the remdesivir parent molecule [GS-441524] led to the development of resistance mutations F476L and V553L, which maintain activity when transferred to SARS-CoV. However, these mutant viruses are less fit than wild-type in both competition assays and _in vivo_ in the absence of selective pressure. To date, no clinical data on SARS-CoV-2 resistance to remdesivir have been described. | |
| Record name | Remdesivir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB14761 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
1809249-37-3 | |
| Record name | L-Alanine, N-[(S)-hydroxyphenoxyphosphinyl]-, 2-ethylbutyl ester, 6-ester with 2-C-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-2,5-anhydro-D-altrononitrile | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1809249-37-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Remdesivir [USAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1809249373 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Remdesivir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB14761 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Remdesivir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID701022537 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 2-ethylbutyl (2S)-2-[[[(2R,3S,4R,5R)-5-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxyoxolan-2-yl]methoxy-phenoxyphosphoryl]amino]propanoate | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | REMDESIVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/3QKI37EEHE | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















