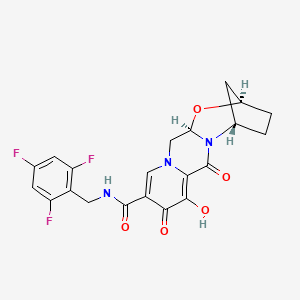
Bictegravir
Overview
Description
Bictegravir is a second-generation integrase strand transfer inhibitor developed by Gilead Sciences for the treatment of human immunodeficiency virus (HIV) infection. It is structurally derived from an earlier compound, dolutegravir. This compound is used in combination with other antiretroviral agents, such as tenofovir alafenamide and emtricitabine, to form a single-tablet regimen for the treatment of HIV-1 infection .
Preparation Methods
Bictegravir can be synthesized through a multi-step process involving several intermediates. One method involves the preparation of (2R,5S,13aR)-8-hydroxy-7,9-dioxo-N-(2,4,6-trifluorobenzyl)-2,3,4,5,7,9,13,13a-octahydro-2,5-methanopyrido[1’,2’:4,5]pyrazino[2,1-b][1,3]oxazepine-10-carboxamide or its pharmaceutically acceptable salts . Another method involves a continuous three-step synthetic strategy that optimizes solvents, temperature, and times to achieve better yields and lower costs .
Chemical Reactions Analysis
Table 1: Key Synthetic Steps and Conditions
- Critical Steps :
- Enzymatic Reduction : The stereoselective reduction of 3-carbonylcyclopentanecarboxylic acid to (3R)-3-hydroxycyclopentanecarboxylic acid ensures correct chirality for subsequent steps .
- Azide Cyclization : Forms the bicyclic oxazepine core under mild conditions, avoiding racemization .
- Final Coupling : HATU-mediated amide bond formation between the oxazepine intermediate and 2,4,6-trifluorobenzylamine achieves the target structure .
Metabolic Reactions
This compound undergoes hepatic and renal metabolism primarily via enzymatic oxidation and glucuronidation.
Table 2: Metabolic Pathways and Enzymatic Targets
- Key Findings :
Stability and Degradation
This compound demonstrates pH-dependent stability and susceptibility to photodegradation.
Table 3: Stability Under Controlled Conditions
- Formulation Stability : Recrystallization from methanol or N-methyl-2-pyrrolidone/water yields >99.5% pure product .
Drug-Drug Interaction Chemistry
Scientific Research Applications
Efficacy in Clinical Trials
Phase 3 Studies
Bictegravir has been evaluated in several Phase 3 clinical trials. In these studies, it demonstrated non-inferiority compared to other standard regimens, such as dolutegravir combined with emtricitabine and tenofovir alafenamide. For instance, one study reported that after 96 weeks, 84% of participants on this compound achieved an HIV-1 RNA level of less than 50 copies/mL, comparable to 86% in the dolutegravir group .
Long-Term Effectiveness
The BICSTaR (this compound Single Tablet Regimen) study provides real-world evidence supporting the long-term effectiveness of this compound. Data from this observational cohort study showed that after 12 months of treatment, HIV-1 RNA was suppressed to less than 50 copies/mL in 94% of treatment-naïve participants and 97% of treatment-experienced participants .
Safety Profile
This compound's safety profile has been extensively documented. In clinical trials, drug-related adverse events were reported in approximately 20% of participants receiving this compound, which is lower compared to the dolutegravir group where adverse events were reported in about 28% . The most common side effects included gastrointestinal issues and headache, with serious adverse events being rare .
Real-World Applications
Diverse Populations
Research indicates that this compound is effective across diverse populations. A study focusing on Asian cohorts found that after 12 months of treatment with this compound/emtricitabine/tenofovir alafenamide, 98.2% of treatment-naïve participants achieved viral suppression . This highlights this compound's applicability in various demographic groups.
Combination Therapies
this compound is also being explored in combination with other antiretroviral therapies for enhanced efficacy. For example, ongoing research into nanoparticle formulations combining this compound with tenofovir alafenamide aims to develop long-acting formulations that could improve adherence and outcomes in HIV prevention strategies .
Case Studies
Case Study: BICSTaR Study
In the BICSTaR study involving over 1500 participants from multiple countries, researchers assessed the effectiveness and safety of this compound/emtricitabine/tenofovir alafenamide. The results confirmed high rates of viral suppression and favorable tolerability over a year-long follow-up period .
Case Study: Long-Acting Formulations
A novel approach using a nanoformulation combining this compound with tenofovir alafenamide has shown promise in preclinical studies for long-acting HIV prevention. This formulation aims to provide sustained drug release, potentially reducing the frequency of dosing required for effective prophylaxis against HIV .
Mechanism of Action
Bictegravir inhibits the strand transfer of viral DNA into the human genome by targeting the HIV-1 integrase enzyme. This prevents the integration of the HIV-1 virus into the host DNA, thereby blocking the replication and propagation of the virus . This compound has shown powerful antiviral activity against various subtypes of HIV-1 and HIV-2 in vitro .
Comparison with Similar Compounds
Bictegravir belongs to the class of integrase strand transfer inhibitors, which also includes dolutegravir, cabotegravir, elvitegravir, and raltegravir . Compared to these compounds, this compound has unique pharmacokinetic and pharmacodynamic properties, such as a long plasma half-life and minimal drug-drug interactions . This compound, dolutegravir, and raltegravir are recommended components of initial regimens for most people with HIV in the US adult and adolescent HIV treatment guidelines .
Biological Activity
Bictegravir (BIC) is an integrase strand transfer inhibitor (INSTI) utilized in the treatment of HIV-1 infection. It is part of the combination therapy Biktarvy, which includes emtricitabine (FTC) and tenofovir alafenamide (TAF). BIC exhibits high potency and selectivity against HIV-1, demonstrating a favorable pharmacokinetic profile and resistance characteristics compared to other INSTIs like raltegravir (RAL) and elvitegravir (EVG) .
This compound functions by inhibiting the HIV integrase enzyme, which is crucial for the viral replication cycle. Specifically, BIC binds to the integrase active site, blocking the strand transfer step necessary for viral DNA integration into the host genome. This action effectively halts viral replication and contributes to viral load reduction in patients .
Key Features of this compound's Mechanism:
- High Potency : BIC shows superior antiviral activity against multiple HIV-1 variants.
- Resistance Profile : It has a longer dissociation half-life from integrase/DNA complexes than RAL and EVG, making it less susceptible to resistance mutations .
- Synergistic Activity : In vitro studies indicate that BIC, when combined with FTC and TAF, exhibits synergistic anti-HIV activity without antagonism .
Pharmacokinetics
This compound is characterized by its favorable pharmacokinetic properties:
- Bioavailability : High oral bioavailability allows for once-daily dosing.
- Half-life : Extended half-life supports sustained antiviral effects.
- Drug Interactions : BIC is a substrate for P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP), necessitating caution when co-administered with drugs that inhibit these transporters .
Table 1: Pharmacokinetic Parameters of this compound
| Parameter | Value |
|---|---|
| Bioavailability | High |
| Half-life | Approximately 18 hours |
| Protein Binding | ~90% |
| Metabolism | Minimal hepatic metabolism; primarily renal excretion |
Clinical Efficacy
Clinical trials have demonstrated that this compound-containing regimens are highly effective in achieving viral suppression in both treatment-naïve and treatment-experienced patients.
Case Studies
- Long-term Efficacy in Women and Older Adults :
- Nanoencapsulation Study :
Safety Profile
The safety profile of this compound is generally favorable, with no significant overlapping toxicities observed when used in combination with FTC and TAF. Studies indicate no evidence of genotoxicity or carcinogenicity associated with this compound, further supporting its clinical use .
Common Adverse Effects:
- Gastrointestinal disturbances
- Fatigue
- Headache
Q & A
Basic Research Questions
Q. What experimental models are most appropriate for evaluating bictegravir’s efficacy against HIV-1 in mucosal tissue?
Methodological Answer: Non-human primate (NHP) models are critical for studying this compound’s mucosal penetration and efficacy, as human trials cannot directly measure drug concentrations in penile, rectal, or urethral tissues. Use high-performance liquid chromatography (HPLC) or mass spectrometry to quantify this compound levels in mucosal swabs and correlate them with viral suppression rates in NHPs. Include control groups receiving emtricitabine/tenofovir to isolate this compound’s contribution .
Q. How does this compound’s antiviral activity profile compare to dolutegravir in INSTI-resistant HIV-1 strains?
Methodological Answer: Conduct in vitro susceptibility assays using patient-derived HIV-1 isolates with integrase strand transfer inhibitor (INSTI) resistance-associated mutations (RAMs). Measure fold-change (FC) in EC50 values relative to wild-type virus for both drugs. Focus on mutations like G140S/Q148R/H/K, where this compound shows greater conformational flexibility and slower dissociation from the integrase-DNA complex compared to dolutegravir. Use statistical tools (e.g., ANOVA) to validate potency differences .
Q. What pharmacokinetic (PK) parameters should be prioritized when designing studies on this compound in obese populations?
Methodological Answer: Use physiologically based pharmacokinetic (PBPK) modeling to predict AUC (area under the curve) and trough concentrations (Cτ) in obese vs. non-obese cohorts. Validate models with sparse sampling in clinical trials, focusing on tissue distribution and protein-binding differences. Monitor viral suppression rates to assess clinical relevance of PK changes .
Advanced Research Questions
Q. How can researchers resolve contradictions between in vitro and clinical data on this compound’s efficacy against INSTI-resistant variants?
Methodological Answer: Perform systematic reviews of in vitro selection studies (e.g., VIKING trial data) and real-world clinical outcomes. Use meta-analysis to quantify discrepancies, such as higher failure rates in patients with Q148 + ≥2 RAMs despite favorable in vitro this compound activity. Investigate confounding factors like adherence, drug-drug interactions, and host genetics .
Q. What experimental designs optimize this compound dosing for event-driven HIV prevention strategies?
Methodological Answer: Design randomized crossover trials comparing steady-state vs. on-demand this compound dosing in NHPs. Measure drug concentrations in mucosal tissues and plasma at multiple timepoints. Pair with ex vivo challenge assays using HIV-1 pseudoviruses to model prevention efficacy. Employ Bayesian adaptive designs to refine dosing intervals .
Q. How do this compound’s pharmacodynamic properties influence combination therapy outcomes in patients with pre-existing M184V/I mutations?
Methodological Answer: Retrospectively analyze clinical trial data (e.g., FDA submission studies) to compare viral suppression rates in patients with M184V/I receiving this compound/emtricitabine/tenofovir vs. dolutegravir-based regimens. Use deep sequencing to detect emergent INSTI RAMs and quantify viral fitness costs. Apply logistic regression to identify predictors of virologic failure .
Q. What methodologies best assess this compound’s penetration into immune sanctuaries (e.g., lymph nodes, CNS)?
Methodological Answer: Use radiolabeled this compound in animal models to track tissue distribution via PET-CT imaging. Corrogate findings with LC-MS/MS measurements in biopsy samples. For human studies, pair lumbar punctures (for CSF) and fine-needle aspirates (lymph nodes) with population PK modeling to estimate compartment-specific exposure .
Guidance for Formulating Rigorous Research Questions
- PICO Framework : Structure questions around Population (e.g., INSTI-experienced patients), Intervention (this compound dosing), Comparison (dolutegravir), and Outcome (virologic failure rate) .
- FINER Criteria : Ensure questions are Feasible (e.g., accessible biospecimens), Novel (e.g., mucosal PK in underrepresented populations), and Relevant to NIH HIV Cure research priorities .
Properties
IUPAC Name |
(1S,11R,13R)-5-hydroxy-3,6-dioxo-N-[(2,4,6-trifluorophenyl)methyl]-12-oxa-2,9-diazatetracyclo[11.2.1.02,11.04,9]hexadeca-4,7-diene-7-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C21H18F3N3O5/c22-9-3-14(23)12(15(24)4-9)6-25-20(30)13-7-26-8-16-27(10-1-2-11(5-10)32-16)21(31)17(26)19(29)18(13)28/h3-4,7,10-11,16,29H,1-2,5-6,8H2,(H,25,30)/t10-,11+,16+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
SOLUWJRYJLAZCX-LYOVBCGYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CC2CC1N3C(O2)CN4C=C(C(=O)C(=C4C3=O)O)C(=O)NCC5=C(C=C(C=C5F)F)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C1C[C@@H]2C[C@H]1N3[C@H](O2)CN4C=C(C(=O)C(=C4C3=O)O)C(=O)NCC5=C(C=C(C=C5F)F)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C21H18F3N3O5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID701027937 | |
| Record name | Bictegravir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID701027937 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
449.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Boiling Point |
682.5±55.0 °C at 760 mmHg | |
| Record name | Bictegravir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11799 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
This single dose medication inhibits the strand transfer of viral DNA into the human genome, preventing HIV-1 virus replication and propagation. In vitro, bictegravir has shown powerful antiviral activity against HIV-2 and various subtypes of HIV-1. It has shown synergistic effects when combined with other ARVs, including tenofovir alafenamide (TAF), emtricitabine (FTC), and darunavir (DRV). The three components of the first USA approved medication ( trade name: Biktarvy ) are as follows: Bictegravir: integrase strand transfer inhibitor; INSTI), an HIV-1 encoded enzyme necessary for viral replication. Inhibition of the integrase enzyme prevents the integration of HIV-1 into host DNA, blocking the conversion of the HIV-1 provirus and progression of the virus [FDA LABEL]. Emtricitabine: FTC, is phosphorylated by cellular enzymes to form emtricitabine 5'-triphosphate. Emtricitabine is phosphorylated to form emtricitabine 5'-triphosphate intracellularly. This metabolite inhibits the activity of human immunodeficiency virus (HIV) reverse transcriptase by competing with the substrate deoxycytidine 5'-triphosphate and by incorporating itself into viral DNA preventing DNA chain elongation [FDA LABEL]. Tenofovir Alafenamide: TAF is a phosphonamidate prodrug of tenofovir (2′-deoxyadenosine monophosphate analog). Plasma exposure to TAF leads to leakage into cells and then TAF is intracellularly converted to tenofovir by hydrolysis by cathepsin. Tenofovir is subsequently phosphorylated by cellular kinases to the metabolite tenofovir diphosphate, which is the active form of the drug. Tenofovir diphosphate inhibits HIV-1 replication by incorporating into viral DNA by the HIV reverse transcriptase, resulting in DNA chain-termination. Tenofovir diphosphate also weakly inhibits mammalian DNA polymerases [FDA LABEL]. | |
| Record name | Bictegravir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11799 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
1611493-60-7 | |
| Record name | (2R,5S,13aR)-2,3,4,5,7,9,13,13a-Octahydro-8-hydroxy-7,9-dioxo-N-[(2,4,6-trifluorophenyl)methyl]-2,5-methanopyrido[1′,2′:4,5]pyrazino[2,1-b][1,3]oxazepine-10-carboxamide | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1611493-60-7 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Bictegravir [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1611493607 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Bictegravir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11799 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Bictegravir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID701027937 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | BICTEGRAVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/8GB79LOJ07 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















