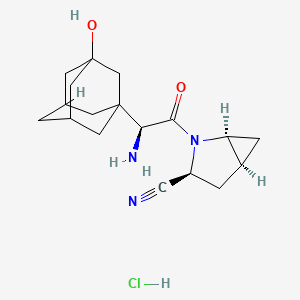
Saxagliptin hydrochloride
Overview
Description
Saxagliptin Hydrochloride is an orally active hypoglycemic drug used in the management of type 2 diabetes mellitus. It belongs to the class of dipeptidyl peptidase-4 (DPP-4) inhibitors, which work by increasing the levels of incretin hormones in the body. These hormones help regulate blood sugar levels by increasing insulin production and decreasing glucose production by the liver .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of Saxagliptin Hydrochloride involves several key steps. One common method includes the coupling of two amino acid derivatives in the presence of a coupling reagent. The amide coupling of (S)-(+)-p-toluenesulfonamide, titanium(IV) ethoxide, and adamantane-1-carboxaldehyde to obtain hydroxybenzotriazole and EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) is a crucial step .
Industrial Production Methods: Industrial production of this compound often involves the use of high-performance liquid chromatography (HPLC) for the quantification and validation of the compound. This method ensures the purity and quality of the final product .
Chemical Reactions Analysis
Types of Reactions: Saxagliptin Hydrochloride undergoes various chemical reactions, including:
Oxidation: This reaction involves the addition of oxygen or the removal of hydrogen.
Reduction: This reaction involves the addition of hydrogen or the removal of oxygen.
Substitution: This reaction involves the replacement of one atom or group of atoms with another.
Common Reagents and Conditions: Common reagents used in these reactions include oxidizing agents like potassium permanganate, reducing agents like sodium borohydride, and substitution reagents like halogens. The conditions for these reactions typically involve controlled temperatures and pH levels to ensure the desired outcome .
Major Products Formed: The major products formed from these reactions include various intermediates that are crucial for the synthesis of this compound. These intermediates are then further processed to obtain the final compound .
Scientific Research Applications
Primary Use in Type 2 Diabetes Mellitus
Saxagliptin is primarily indicated for use as an adjunct to diet and exercise in adults with type 2 diabetes. It has shown effectiveness in lowering blood sugar levels, improving glycemic control as measured by reductions in glycated hemoglobin (HbA1c) levels. Clinical studies have demonstrated that saxagliptin can lead to significant improvements in fasting plasma glucose and postprandial glucose levels across diverse patient populations, including those with varying cardiovascular risk factors .
Combination Therapy
Recent studies have explored the efficacy of saxagliptin when used in combination with other treatments:
- With Insulin : Saxagliptin has been shown to be effective in patients already on insulin therapy, providing additional glycemic control .
- With Vitamin D : A study indicated that saxagliptin combined with vitamin D could help preserve beta-cell function in adult-onset type 1 diabetes, demonstrating a potential role beyond type 2 diabetes management .
Cardiovascular Implications
Research has suggested that saxagliptin may have beneficial effects on cardiovascular health. In various clinical trials, it was found to be well-tolerated and associated with improved glycemic control without significant adverse cardiovascular events, making it a suitable option for diabetic patients with cardiovascular concerns .
Renal Impairment Considerations
Saxagliptin's pharmacokinetics are affected by renal function. Studies indicate that dosage adjustments may be necessary for patients with renal impairment to avoid increased exposure and potential side effects. This highlights the importance of individualized treatment plans based on renal function .
Clinical Trials Overview
A comprehensive review of clinical trials involving saxagliptin reveals consistent findings regarding its efficacy and safety:
Side Effects
While generally well-tolerated, saxagliptin can cause side effects such as:
- Headache
- Nasopharyngitis
- Gastrointestinal issues
- Rarely, pancreatitis or severe allergic reactions .
Case Study: Saxagliptin and Beta-Cell Function Preservation
A multicenter randomized trial evaluated the impact of saxagliptin combined with vitamin D on beta-cell function in patients with adult-onset type 1 diabetes. The study demonstrated that this combination therapy led to a statistically significant preservation of C-peptide levels over a 24-month period compared to conventional therapy alone, suggesting potential benefits for patients beyond standard diabetes management .
Observational Study: Hospitalized Heart Failure Risk
An observational study assessed the risk of hospitalized heart failure among new users of saxagliptin compared to other DPP-4 inhibitors. The findings indicated no significant increase in heart failure risk among saxagliptin users, reinforcing its safety profile in diabetic patients with pre-existing heart conditions .
Mechanism of Action
Saxagliptin Hydrochloride works by inhibiting the enzyme dipeptidyl peptidase-4 (DPP-4). This inhibition increases the levels of incretin hormones such as glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). These hormones help lower blood sugar levels by increasing insulin secretion and decreasing glucagon secretion from the liver .
Comparison with Similar Compounds
Metformin: Another antidiabetic drug that improves insulin sensitivity.
Semaglutide: A GLP-1 receptor agonist that mimics the effects of incretin hormones.
Comparison:
Saxagliptin Hydrochloride vs. Metformin: this compound works by inhibiting DPP-4, while Metformin improves insulin sensitivity.
This compound vs. Semaglutide: Both drugs increase the levels of incretin hormones, but Semaglutide is a GLP-1 receptor agonist, while this compound is a DPP-4 inhibitor.
This compound stands out due to its specific mechanism of action and its effectiveness in combination therapies for type 2 diabetes mellitus.
Biological Activity
Saxagliptin hydrochloride is a dipeptidyl peptidase-4 (DPP-4) inhibitor primarily used in the management of type 2 diabetes mellitus (T2DM). Its biological activity is characterized by its ability to enhance glycemic control through various mechanisms, including the modulation of incretin hormones. This article explores the biological activity of saxagliptin, supported by data tables, case studies, and detailed research findings.
Saxagliptin acts by inhibiting the DPP-4 enzyme, which is responsible for the degradation of incretin hormones such as glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). By preventing the breakdown of these hormones, saxagliptin increases their levels in circulation, leading to:
- Increased Insulin Secretion : Saxagliptin enhances insulin release from pancreatic beta cells in response to elevated blood glucose levels.
- Decreased Glucagon Secretion : It reduces glucagon secretion from pancreatic alpha cells, which helps lower hepatic glucose production.
- Improved Glycemic Control : The overall effect is a reduction in fasting plasma glucose and postprandial glucose levels.
The selectivity of saxagliptin for DPP-4 over other DPP enzymes (DPP-8 and DPP-9) is notable, with a 400-fold and 950-fold selectivity, respectively . This selectivity contributes to its favorable safety profile, minimizing adverse effects associated with non-selective DPP inhibition.
Pharmacokinetics
Saxagliptin exhibits first-order kinetics with a median time to peak plasma concentration (Tmax) of approximately 2 hours after oral administration. The elimination half-life is about 2.5 hours for saxagliptin and 3.1 hours for its active metabolite M2 . Key pharmacokinetic parameters are summarized in Table 1.
| Parameter | Value |
|---|---|
| Bioavailability | 67% |
| Volume of Distribution | 151 L |
| Protein Binding | <10% |
| Metabolism | CYP3A4/5 |
| Elimination | Renal and hepatic |
| Half-life | Saxagliptin: 2.5 hours; M2: 3.1 hours |
Clinical Efficacy
Saxagliptin has demonstrated significant efficacy in improving glycemic control in various clinical settings:
- Monotherapy : In clinical trials, saxagliptin reduced HbA1c levels by approximately 0.7% to 0.9% compared to placebo .
- Combination Therapy : When used in combination with metformin or sulfonylureas, saxagliptin showed enhanced efficacy without increasing the risk of hypoglycemia .
Case Study Findings
A recent multi-center randomized controlled trial investigated the effects of saxagliptin combined with vitamin D on β-cell preservation in T2DM patients. Results indicated that participants receiving saxagliptin plus vitamin D had a significant preservation of β-cell function compared to those on conventional therapy alone:
- C-peptide AUC Change at 24 Months :
- Saxagliptin + Vitamin D: -276 pmol/L
- Saxagliptin Alone: -314 pmol/L
- Conventional Therapy: -419 pmol/L
The proportion of participants showing a ΔC-peptide response was significantly higher in the saxagliptin plus vitamin D group (57.6%) compared to conventional therapy (37.2%) .
Safety Profile
Saxagliptin is generally well-tolerated, with a low incidence of hypoglycemia and weight neutrality observed across clinical trials. Adverse effects are comparable to placebo, making it a favorable option for T2DM management .
Q & A
Q. How can researchers optimize analytical methods for simultaneous quantification of Saxagliptin hydrochloride in combination therapies (e.g., with Metformin or Dapagliflozin)?
Answer:
Researchers should prioritize chromatographic techniques such as HPTLC or RP-HPLC due to their specificity and reproducibility. For HPTLC, a validated method using silica gel plates with a mobile phase of acetonitrile:1% ammonium acetate (9:1 v/v) and detection at 210 nm enables simultaneous analysis of Saxagliptin (SAX), Metformin (MET), and Dapagliflozin (DAP) with linear ranges of 0.25–10 μg/band (SAX/DAP) and 0.25–25 μg/band (MET) . For HPLC, a C18 column with phosphate buffer (pH 4.5) and acetonitrile gradient elution achieves baseline separation of SAX and MET, with validation per ICH guidelines for precision (RSD <2%) and accuracy (98–102%) .
Q. What safety protocols are critical for handling this compound in laboratory settings?
Answer:
this compound poses risks of skin sensitization and respiratory irritation. Key protocols include:
- Personal Protective Equipment (PPE): Nitrile gloves, lab coats, and safety goggles to avoid direct contact .
- Ventilation: Use fume hoods for powder handling to prevent inhalation of dust/particulates .
- Storage: Keep in airtight containers at 2–8°C, away from moisture and incompatible substances (e.g., strong oxidizers) .
- Spill Management: Neutralize spills with inert absorbents (e.g., vermiculite) and dispose of as hazardous waste .
Q. What experimental design considerations are essential for developing controlled-release formulations of this compound?
Answer:
Preformulation studies should focus on:
- Solubility and Stability: Assess pH-dependent solubility (optimal at 4.5–6.5) and photostability under ICH Q1B guidelines .
- Excipient Compatibility: Screen with osmotic agents (e.g., cellulose acetate) and plasticizers (e.g., PEG 400) for bilayer tablets combining immediate-release SAX and controlled-release MET .
- In Vitro Release Testing: Use USP Apparatus II (paddle) at 50 rpm in 0.1N HCl for 2 hours followed by pH 6.8 buffer to simulate gastrointestinal conditions .
Q. How can researchers resolve contradictions between preclinical and clinical pharmacokinetic data for this compound?
Answer:
Discrepancies often arise from species-specific DPP-4 enzyme affinity or metabolite activity. Strategies include:
- Metabolite Profiling: SAX’s active metabolite (M2) contributes 50% of DPP-4 inhibition in humans but exhibits lower potency in rodents .
- Dose Adjustments: Use allometric scaling based on body surface area and enzyme expression levels in target tissues .
- Population Pharmacokinetics: Apply nonlinear mixed-effects modeling (NONMEM) to account for inter-individual variability in clinical trials .
Q. What methodological approaches validate this compound’s selectivity for DPP-4 inhibition over related proteases?
Answer:
- Enzyme Assays: Compare IC₅₀ values against DPP-8 and DPP-9 using fluorogenic substrates (e.g., H-Gly-Pro-AMC). SAX shows >400-fold selectivity for DPP-4 (Ki = 0.6–1.3 nM) .
- Structural Analysis: Molecular docking studies reveal SAX’s cyanopyrrolidine group forms hydrogen bonds with DPP-4’s Glu205 and Tyr547 residues, unlike DPP-8/9 .
Q. What stability challenges arise in this compound formulations, and how are they mitigated?
Answer:
- Hydrolysis: SAX degrades in acidic conditions (e.g., gastric pH). Use enteric coatings (e.g., Eudragit L100) for delayed release .
- Oxidation: Include antioxidants (e.g., ascorbic acid) in lyophilized parenteral formulations to prevent degradation during storage .
- Photodegradation: Protect tablets with opaque packaging (e.g., aluminum blisters) .
Q. How should clinical trials be designed to assess Saxagliptin’s long-term cardiovascular outcomes in type 2 diabetes?
Answer:
- Endpoint Selection: Composite endpoints (e.g., MACE: cardiovascular death, nonfatal MI/stroke) aligned with FDA guidance .
- Comparator Arms: Use active controls (e.g., Sitagliptin) and adjust for baseline HbA1c variability .
- Follow-Up Duration: Minimum 2 years to detect treatment-emergent effects (e.g., heart failure risk) .
Properties
IUPAC Name |
(1S,3S,5S)-2-[(2S)-2-amino-2-(3-hydroxy-1-adamantyl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile;hydrochloride | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C18H25N3O2.ClH/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17;/h10-15,23H,1-7,9,20H2;1H/t10?,11?,12-,13+,14+,15-,17?,18?;/m1./s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
TUAZNHHHYVBVBR-NHKADLRUSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1C2CC2N(C1C#N)C(=O)C(C34CC5CC(C3)CC(C5)(C4)O)N.Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C1[C@@H]2C[C@@H]2N([C@@H]1C#N)C(=O)[C@H](C34CC5CC(C3)CC(C5)(C4)O)N.Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C18H26ClN3O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID50991191 | |
| Record name | Saxagliptin hydrochloride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID50991191 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
351.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
709031-78-7 | |
| Record name | Saxagliptin hydrochloride | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0709031787 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Saxagliptin hydrochloride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID50991191 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (1S,3S,5S)-2-[(2S)-2-Amino-2-(3-hydroxyadamantan-1-yl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile hydrochloride | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | SAXAGLIPTIN HYDROCHLORIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/Z8J84YIX6L | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















