Ledipasvir
Overview
Description
Ledipasvir is a direct-acting antiviral agent used primarily for the treatment of chronic hepatitis C virus (HCV) infections. It was developed by Gilead Sciences and is commonly used in combination with sofosbuvir under the brand name Harvoni. This compound targets the non-structural protein 5A (NS5A) of the hepatitis C virus, which is essential for viral replication and assembly .
Preparation Methods
Synthetic Routes and Reaction Conditions
The synthesis of Ledipasvir involves multiple steps, including the preparation of key intermediates. One method involves the preparation of a high-purity intermediate, (1R, 3S, 4S)-N-tertbutyloxycarbonyl-2-azabicyclo[2.2.1]heptane-3-carboxylic acid, through enzymatic hydrolysis . Another method involves late-stage cyclopropanation and fluorination processes, which provide a novel and efficient route for the preparation of this compound with a total yield of 20% over eight linear steps .
Industrial Production Methods
Industrial production of this compound focuses on optimizing yield, purity, and cost-effectiveness. The process typically involves the use of high-purity intermediates and environmentally friendly reaction conditions. The methods are designed to be scalable for large-scale production, ensuring high selectivity and reduced production costs .
Chemical Reactions Analysis
Types of Reactions
Ledipasvir undergoes various chemical reactions, including:
Reduction: Reduction reactions are less common for this compound.
Substitution: Substitution reactions, particularly involving halogen atoms, are more relevant in the synthesis of this compound
Common Reagents and Conditions
Common reagents used in the synthesis and reactions of this compound include acetonitrile, acetic acid, and isopropyl ether. Reaction conditions often involve elevated temperatures and controlled environments to ensure high yield and purity .
Major Products Formed
The major product formed from the reactions involving this compound is the final active pharmaceutical ingredient used in the treatment of hepatitis C. Other intermediates and by-products are typically removed through purification processes .
Scientific Research Applications
Treatment of Hepatitis C
Ledipasvir is approved for use in combination with sofosbuvir in various treatment regimens:
- 8-week regimen : For treatment-naive patients without cirrhosis and low baseline HCV RNA levels (<6 million IU/mL).
- 12-week regimen : For a broader range of patients, including those with cirrhosis or prior treatment experience.
Efficacy Data:
- In clinical trials, sustained virologic response (SVR) rates have been reported as high as 95%-100% across different populations and treatment durations. For instance, a study involving 2,099 participants showed SVR12 rates of 96% for the 8-week regimen and 97% for the 12-week regimen .
Real-World Effectiveness
Real-world studies have corroborated clinical trial findings, demonstrating high SVR rates in diverse patient populations:
- A multicenter observational study reported SVR12 rates of 96% among patients treated with this compound-sofosbuvir for 8 weeks .
- Factors influencing treatment outcomes included liver function indicators such as albumin levels and bilirubin levels .
Safety Profile
The safety profile of this compound has been assessed in numerous studies. Common adverse effects include:
- Headache
- Fatigue
- Nausea
Serious adverse events are rare but can occur, particularly when ribavirin is included in the treatment regimen .
Comparative Efficacy
The combination therapy of this compound and sofosbuvir has been compared to other antiviral treatments. The following table summarizes key findings from various studies:
Case Studies
Several case studies highlight the effectiveness of this compound in diverse settings:
- Egyptian Cohort Study : This study enrolled 255 patients with HCV genotype 4 and demonstrated high SVR rates (95% for the 8-week regimen and 98% for the 12-week regimen), emphasizing its efficacy even among previously treated patients .
- HCV-TARGET Study : An extensive analysis involving over 2,000 participants showed that higher baseline albumin and lower bilirubin levels were associated with better treatment outcomes .
Mechanism of Action
Ledipasvir inhibits the hepatitis C virus non-structural protein 5A (NS5A), which is crucial for viral RNA replication and assembly of HCV virions. By preventing the hyperphosphorylation of NS5A, this compound disrupts the production of viral proteins, thereby inhibiting viral replication and assembly .
Comparison with Similar Compounds
Similar Compounds
Sofosbuvir: Another direct-acting antiviral used in combination with Ledipasvir. It inhibits the NS5B polymerase of HCV.
Daclatasvir: An NS5A inhibitor similar to this compound but with different pharmacokinetic properties.
Ombitasvir: Another NS5A inhibitor used in combination with other antiviral agents for HCV treatment
Uniqueness of this compound
This compound is unique due to its high potency against multiple HCV genotypes and its ability to achieve sustained virologic response (SVR) rates of over 95% when used in combination with sofosbuvir. Its long half-life and minimal side effects make it a preferred choice for HCV treatment .
Biological Activity
Ledipasvir is a potent antiviral agent used primarily in the treatment of hepatitis C virus (HCV) infections. It functions as an NS5A inhibitor, targeting the NS5A protein crucial for HCV replication. This article provides a comprehensive overview of the biological activity of this compound, including its pharmacological properties, efficacy in clinical studies, and safety profile.
This compound inhibits HCV replication by binding to the NS5A protein, which is essential for viral RNA replication and assembly. Although the exact biochemical mechanism remains unconfirmed due to the lack of known enzymatic functions of NS5A, studies indicate that this compound effectively disrupts HCV lifecycle processes.
- In vitro Activity : this compound exhibits varying inhibitory concentrations (EC50) against different HCV genotypes:
Pharmacokinetics
This compound is characterized by high plasma protein binding (>99.8%) and demonstrates a wide volume of distribution. Following administration, it shows minimal metabolism with over 98% of systemic exposure attributed to the parent compound. The pharmacokinetic profile indicates a long mean residence time, supporting its efficacy in sustained viral response (SVR) outcomes .
Efficacy in Clinical Trials
Numerous clinical trials have evaluated the efficacy of this compound, often in combination with sofosbuvir (LDV/SOF). The following table summarizes key findings from prominent studies:
| Study | Treatment Duration | Population | SVR12 Rate |
|---|---|---|---|
| LONESTAR Study | 8 weeks | Treatment-naïve GT1 | 95% |
| LONESTAR Study | 12 weeks | Treatment-naïve GT1 | 100% |
| Phase II Trial | 12 weeks | Treatment-experienced | 95% |
| Phase II Trial | 8 weeks | Acute HCV in HIV-infected men | 100% |
| Egyptian Study | 12 weeks | Non-cirrhotic patients | ≥94% |
These results indicate that this compound combined with sofosbuvir is highly effective across various patient populations, including those with prior treatment failures and different HCV genotypes .
Safety Profile
The safety profile of this compound has been assessed in various studies, revealing that it is generally well tolerated. Common adverse events include headache and fatigue, particularly in patients receiving ribavirin alongside this compound . Serious adverse events are rare, making it a favorable option for many patients.
Case Studies
-
Case Study: Treatment-Naïve Patients
In a multicenter trial involving treatment-naïve patients with HCV genotype 4, SVR12 rates reached up to 98% after a 12-week regimen of LDV/SOF, demonstrating high effectiveness even in populations previously underrepresented in clinical trials . -
Case Study: Patients with HIV Co-Infection
A study focusing on men with HIV co-infection who received an eight-week course of LDV/SOF achieved a remarkable SVR rate of 100%, highlighting the compound's efficacy even in complex patient populations .
Q & A
Q. Basic: What are the standard analytical methods for quantifying Ledipasvir in pharmacokinetic studies?
Q: What validated chromatographic techniques are used to quantify this compound in plasma or formulations, and how are linearity and accuracy ensured? A: Reverse-phase high-performance liquid chromatography (RP-HPLC) is widely employed, with calibration curves established using absorbance spectra (200–400 nm). Linear ranges for this compound typically span 5–25 µg/mL, validated via recovery tests showing >98% accuracy . Method validation includes testing selectivity under thermal stress to confirm no peak overlap with degradation products or excipients .
Q. Basic: What are the key steps in this compound synthesis, and how have recent advancements improved efficiency?
Q: Describe the synthetic pathway for this compound and innovations that reduce step count or improve yield.
A: The synthesis involves a late-stage cyclopropanation and difluorination, enabling an 8-step process with 20% overall yield. Copper electrode-mediated multistep reactions accelerate synthesis by 20–50× compared to batch methods, reducing reaction time and resource use .
Q. Advanced: How do multivariate chemometric models enhance precision in quantifying this compound in complex matrices?
Q: What computational approaches address variability in spectrophotometric data for this compound-Sofosbuvir mixtures? A: Genetic algorithm-optimized partial least squares (GA-PLS) and artificial neural networks (ANN) improve predictive accuracy by selecting optimal wavelength combinations and reducing noise. These models achieve <2% relative error in concentration predictions for multi-component formulations .
Q. Advanced: What pharmacokinetic challenges arise from this compound’s pH-dependent solubility, and how are they mitigated?
Q: How does gastric pH affect this compound bioavailability, and what drug-interaction protocols are recommended? A: Elevated gastric pH reduces this compound solubility, lowering exposure by 30–40%. Clinical protocols mandate 4-hour separation from antacids and avoid high-dose PPIs (>20 mg omeprazole). Real-world data show proton pump inhibitor (PPI) use correlates with 5–6% lower SVR rates, necessitating dose timing adjustments .
Q. Advanced: How do contradictions between clinical trial and real-world data inform this compound-Sofosbuvir treatment duration?
Q: Why do some real-world studies report lower SVR rates for 8-week regimens despite non-inferiority in trials? A: While phase III trials showed 94–99% SVR for 8-week regimens in non-cirrhotic patients, real-world analyses identify confounding factors (e.g., cirrhosis misclassification, PPI use). Multivariate regression highlights albumin levels, bilirubin, and cirrhosis status as predictors of efficacy, supporting extended regimens for high-risk subgroups .
Q. Basic: How are stability-indicating methods validated for this compound in combination therapies?
Q: What stress conditions and parameters are used to validate this compound stability in fixed-dose combinations? A: Forced degradation studies under thermal (40–80°C), oxidative (H₂O₂), and photolytic conditions confirm method robustness. Acceptance criteria include ≤2% deviation in peak purity and resolution, with recovery rates of 98–102% across 50–150% concentration ranges .
Q. Advanced: How do multivariate analyses identify predictors of treatment failure in this compound-based regimens?
Q: What statistical methods reconcile disparate SVR outcomes in heterogeneous patient populations? A: Logistic regression of real-world cohorts (e.g., HCV-TARGET) identifies predictors like baseline HCV RNA >6 million IU/mL, hypoalbuminemia (<3.5 g/dL), and cirrhosis as risk factors. Machine learning models further stratify patients by IL28B genotype and fibrosis score to optimize regimen duration .
Q. Advanced: How do NS5A resistance-associated substitutions (RASs) impact this compound efficacy in HIV/HCV co-infection?
Q: What RASs are linked to virologic relapse, and how are they detected in resistant strains? A: Deep sequencing identifies Y93H/N RASs in NS5A, reducing this compound’s binding affinity by >100×. In co-infected patients, pre-existing RASs correlate with 2–4% relapse rates, necessitating resistance testing before salvage therapies (e.g., adding ribavirin or extending treatment to 24 weeks) .
Properties
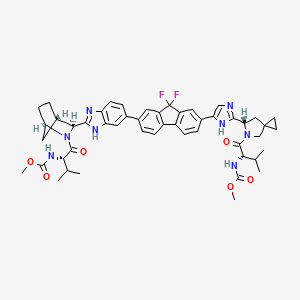
IUPAC Name |
methyl N-[(2S)-1-[(6S)-6-[5-[9,9-difluoro-7-[2-[(1R,3S,4S)-2-[(2S)-2-(methoxycarbonylamino)-3-methylbutanoyl]-2-azabicyclo[2.2.1]heptan-3-yl]-3H-benzimidazol-5-yl]fluoren-2-yl]-1H-imidazol-2-yl]-5-azaspiro[2.4]heptan-5-yl]-3-methyl-1-oxobutan-2-yl]carbamate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C49H54F2N8O6/c1-24(2)39(56-46(62)64-5)44(60)58-23-48(15-16-48)21-38(58)42-52-22-37(55-42)28-9-13-32-31-12-8-26(18-33(31)49(50,51)34(32)19-28)27-10-14-35-36(20-27)54-43(53-35)41-29-7-11-30(17-29)59(41)45(61)40(25(3)4)57-47(63)65-6/h8-10,12-14,18-20,22,24-25,29-30,38-41H,7,11,15-17,21,23H2,1-6H3,(H,52,55)(H,53,54)(H,56,62)(H,57,63)/t29-,30+,38-,39-,40-,41-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
VRTWBAAJJOHBQU-KMWAZVGDSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)C(C(=O)N1CC2(CC2)CC1C3=NC=C(N3)C4=CC5=C(C=C4)C6=C(C5(F)F)C=C(C=C6)C7=CC8=C(C=C7)N=C(N8)C9C1CCC(C1)N9C(=O)C(C(C)C)NC(=O)OC)NC(=O)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC(C)[C@@H](C(=O)N1CC2(CC2)C[C@H]1C3=NC=C(N3)C4=CC5=C(C=C4)C6=C(C5(F)F)C=C(C=C6)C7=CC8=C(C=C7)N=C(N8)[C@@H]9[C@H]1CC[C@H](C1)N9C(=O)[C@H](C(C)C)NC(=O)OC)NC(=O)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C49H54F2N8O6 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID90154829 | |
| Record name | Ledipasvir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID90154829 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
889.0 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
Ledipasvir is an inhibitor of the Hepatitis C Virus (HCV) NS5A protein required for viral RNA replication and assembly of HCV virions. Although its exact mechanism of action is unknown, it is postulated to prevent hyperphosphorylation of NS5A which is required for viral production. | |
| Record name | Ledipasvir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB09027 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
1256388-51-8 | |
| Record name | Ledipasvir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1256388-51-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Ledipasvir [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1256388518 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Ledipasvir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB09027 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Ledipasvir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID90154829 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Methyl[(2S)-1-{(6S)-6-[5-(9,9-difluoro-7-{2-[(1R,3S,4S)-2-{(2S)-2-[(methoxycarbonyl)amino]-3-methylbutanoyl}-2-azabicyclo[2.2.1]hept-3-yl]-1H-benzimidazol-6-yl}-9H-fluoren-2-yl)-1H-imidazol-2-yl]-5-azaspiro[2.4]hept-5-yl}-3-methyl-1-oxobutan-2-yl]carbamate | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | LEDIPASVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/013TE6E4WV | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Synthesis routes and methods I
Procedure details












Synthesis routes and methods II
Procedure details












Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














