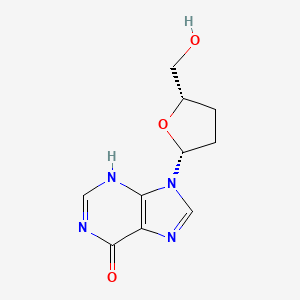
Didanosine
Übersicht
Beschreibung
Didanosine, also known as 2’,3’-dideoxyinosine, is a synthetic nucleoside analogue of adenosine. It is primarily used as an antiretroviral medication for the treatment of Human Immunodeficiency Virus (HIV) infection. This compound works by inhibiting the reverse transcriptase enzyme, which is essential for the replication of HIV. This compound was first described in 1975 and approved for use in the United States in 1991 .
Vorbereitungsmethoden
Synthetische Routen und Reaktionsbedingungen: Didanosin wird durch einen mehrstufigen Prozess aus Inosin synthetisiert. Die wichtigsten Schritte beinhalten die selektive Entfernung von Hydroxylgruppen an den 2’ und 3’ Positionen des Ribose-Moleküls. Dies wird typischerweise durch eine Reihe von chemischen Reaktionen erreicht, einschließlich Schutz, Entschützung und selektiver Reduktionsschritte.
Industrielle Produktionsmethoden: Die industrielle Produktion von Didanosin umfasst die großtechnische Synthese unter Verwendung ähnlicher chemischer Routen wie bei der Laborsynthese, die jedoch für höhere Ausbeuten und Reinheit optimiert sind. Der Prozess umfasst strenge Qualitätskontrollmaßnahmen, um sicherzustellen, dass das Endprodukt die pharmazeutischen Standards erfüllt.
Arten von Reaktionen:
Oxidation: Didanosin kann Oxidationsreaktionen unterliegen, obwohl diese typischerweise nicht für seine pharmakologische Aktivität relevant sind.
Reduktion: Die Verbindung selbst ist eine reduzierte Form von Inosin, die durch selektive Reduktionsschritte während der Synthese erzielt wird.
Substitution: Didanosin kann an nukleophilen Substitutionsreaktionen teilnehmen, insbesondere am Purinring.
Häufige Reagenzien und Bedingungen:
Oxidation: Häufige Oxidationsmittel wie Wasserstoffperoxid oder Kaliumpermanganat.
Reduktion: Reduktionsmittel wie Natriumborhydrid oder Lithiumaluminiumhydrid.
Substitution: Nukleophile wie Amine oder Thiole unter basischen Bedingungen.
Hauptprodukte, die gebildet werden: Das Hauptprodukt dieser Reaktionen ist typischerweise Didanosin selbst oder dessen Derivate, abhängig von den spezifischen Reaktionsbedingungen und den verwendeten Reagenzien.
Wissenschaftliche Forschungsanwendungen
Didanosin hat eine breite Palette von Anwendungen in der wissenschaftlichen Forschung:
Chemie: Wird als Modellverbindung zur Untersuchung von Nukleosid-Analoga und deren chemischen Eigenschaften verwendet.
Biologie: Wird in Studien zum Nukleinsäurestoffwechsel und zu Enzymwechselwirkungen eingesetzt.
Medizin: Wird in der Forschung zur HIV-Behandlung und zu Resistenzmechanismen umfassend eingesetzt.
Industrie: Wird bei der Entwicklung von antiretroviralen Therapien und bei der Untersuchung von Arzneimittelwechselwirkungen eingesetzt.
5. Wirkmechanismus
Didanosin wird intrazellulär zu seiner aktiven Form, Dideoxyadenosintriphosphat (ddATP), metabolisiert. Dieses aktive Metabolit hemmt das HIV-Reverse-Transkriptase-Enzym, indem es mit dem natürlichen Substrat, Deoxyadenosintriphosphat (dATP), konkurriert. Durch die Einlagerung in die virale DNA verursacht ddATP eine Kettenabbruchreaktion, die die Virusreplikation verhindert .
Ähnliche Verbindungen:
Zidovudin (AZT): Ein weiteres Nukleosid-Reverse-Transkriptase-Inhibitor, das in der HIV-Behandlung eingesetzt wird.
Stavudin (d4T): Ähnlicher Wirkmechanismus, aber unterschiedliches Nebenwirkungsprofil.
Lamivudin (3TC): Wird häufig in Kombination mit anderen Antiretroviralen eingesetzt, um synergistische Effekte zu erzielen.
Eindeutigkeit: Didanosin ist durch seine spezifischen Strukturmodifikationen einzigartig, die seine Aktivität gegen HIV vermitteln. Im Gegensatz zu anderen Nukleosid-Analoga hat Didanosin eine Hypoxanthin-Base, die an den Zucker-Ring gebunden ist, was für seinen Wirkmechanismus entscheidend ist .
Wirkmechanismus
Didanosine is metabolized intracellularly to its active form, dideoxyadenosine triphosphate (ddATP). This active metabolite inhibits the HIV reverse transcriptase enzyme by competing with the natural substrate, deoxyadenosine triphosphate (dATP). By incorporating into the viral DNA, ddATP causes chain termination, preventing the virus from replicating .
Vergleich Mit ähnlichen Verbindungen
Zidovudine (AZT): Another nucleoside reverse transcriptase inhibitor used in HIV treatment.
Stavudine (d4T): Similar mechanism of action but different side effect profile.
Lamivudine (3TC): Often used in combination with other antiretrovirals for synergistic effects.
Uniqueness: Didanosine is unique in its specific structural modifications, which confer its activity against HIV. Unlike other nucleoside analogues, this compound has a hypoxanthine base attached to the sugar ring, which is crucial for its mechanism of action .
Biologische Aktivität
Didanosine (ddI), a nucleoside reverse transcriptase inhibitor (NRTI), has been primarily utilized in the treatment of HIV/AIDS. Its biological activity is characterized by its mechanism of action, pharmacokinetics, mutagenicity, and clinical efficacy. This article synthesizes findings from various studies to provide a comprehensive overview of this compound's biological activity.
This compound is metabolized intracellularly to its active form, this compound triphosphate (ddATP). The conversion involves phosphorylation through several enzymatic pathways, ultimately leading to the incorporation of ddATP into viral DNA during reverse transcription. This incorporation results in chain termination, effectively inhibiting viral replication. The intracellular half-life of ddATP is approximately 12-24 hours, suggesting that less frequent dosing may be sufficient compared to other NRTIs like zidovudine (AZT) .
Pharmacokinetics
This compound exhibits low plasma protein binding (≤ 5%) and is less lipophilic than zidovudine. Consequently, it has a different distribution profile, with lower concentrations in cerebrospinal fluid compared to plasma . Approximately 40% of this compound is excreted unchanged in urine, while the remainder is metabolized into hypoxanthine and uric acid .
Mutagenicity and Toxicity
Research indicates that this compound may exhibit clastogenic effects at high doses. In vitro studies have shown it can induce DNA damage in human lymphocytes and Chinese hamster ovary cells. However, animal studies have reported no significant increase in micronuclei formation at lower doses . Notably, this compound does not appear to induce reverse mutations in Salmonella typhimurium without metabolic activation, suggesting a limited mutagenic potential under certain conditions .
Table 1: Summary of Mutagenicity Studies on this compound
| Study Type | Results |
|---|---|
| In Vitro (Human Lymphocytes) | Clastogenic effects observed |
| In Vitro (Chinese Hamster Ovary) | Induced DNA damage |
| In Vivo (Rodent Micronucleus Assay) | No increase in micronuclei at low doses |
| Salmonella typhimurium | No reverse mutations without activation |
Clinical Efficacy
This compound has demonstrated significant antiviral activity in clinical settings. A study involving 168 HIV-1 infected patients showed that those treated with this compound experienced a median decrease in HIV-1 RNA levels by -0.56 log copies/mL compared to +0.07 log copies/mL in the placebo group after four weeks . Notably, 31% of patients on this compound achieved undetectable viral loads (<400 copies/mL), compared to only 6% in the placebo group .
Case Study: this compound in Treatment-Experienced Patients
In a cohort of treatment-experienced patients with multiple nucleoside reverse-transcriptase inhibitor-associated mutations (NAMs), this compound maintained antiviral efficacy. Patients with up to five NAMs at baseline still exhibited significant reductions in viral load when treated with this compound . This underscores its potential utility even in challenging treatment scenarios.
Q & A
Basic Research Questions
Q. What validated analytical methods are recommended for quantifying didanosine in pharmaceutical formulations?
The United States Pharmacopeia (USP) outlines a reversed-phase HPLC method for this compound quantification. A standard preparation using USP this compound RS is diluted to ~LC/900, and the assay preparation follows similar dilution. Chromatographic separation uses a 0.01 M ammonium acetate buffer (pH 6.0) and a C18 column. Peak responses are compared between standard and test solutions to calculate purity (90–110% of labeled claim) . For higher sensitivity, LC-MS with deuterated internal standards (e.g., this compound-d2) improves accuracy in pharmacokinetic studies .
Q. How should researchers handle this compound to ensure safety in laboratory settings?
this compound requires precautions to avoid inhalation, skin contact, or ingestion. Use PPE (gloves, lab coat, goggles) and work in a ventilated hood. Storage should be in sealed containers at 15–30°C to prevent degradation. In case of exposure, rinse affected areas with water for 15 minutes and seek medical evaluation due to its acute toxicity (oral LD₅₀: 4300 mg/kg in rats) and potential mutagenicity .
Q. What are the key considerations when designing in vitro studies to assess this compound’s antiviral efficacy?
Use HIV-1 reverse transcriptase (RT) inhibition assays with wild-type and mutant RT strains (e.g., L74V or K65R mutations linked to resistance). Cell-based models (e.g., MT-4 lymphocytes) should measure IC₅₀ values via plaque reduction or RT activity assays. Include controls for cytotoxicity and validate results against reference inhibitors like zidovudine .
Q. How can researchers ensure stability of this compound in solution during long-term experiments?
Prepare fresh solutions in pH-neutral buffers (e.g., PBS) and avoid prolonged exposure to light or temperatures >30°C. Degradation products (e.g., hypoxanthine) can be monitored via HPLC with UV detection at 250 nm. For multi-day assays, freeze aliquots at -20°C and validate stability over 72 hours .
Advanced Research Questions
Q. How do RT codon polymorphisms (e.g., L228H/R) influence this compound’s virological response in clinical cohorts?
Retrospective genotypic analysis of HIV-1 isolates from ART-experienced patients reveals that polymorphisms at RT codons 228 (L→H/R) correlate with reduced response to this compound. Use multivariate regression to adjust for confounding factors (e.g., prior NRTI exposure). Annotate sequences with resistance scores (e.g., ANRS algorithm) and validate findings in independent datasets to account for population-specific mutation patterns .
Q. What methodologies resolve contradictions in long-term toxicity data for this compound in preclinical models?
Mitochondrial toxicity assays (e.g., lactate/pyruvate ratios in hepatocytes) and in vivo models (e.g., transgenic mice expressing POLG mutations) can clarify mechanisms like mitochondrial DNA depletion. Combine histopathology (e.g., hepatic steatosis) with metabolomics to distinguish drug-specific effects from confounding variables (e.g., comorbidities) .
Q. How can researchers optimize genotypic resistance scoring systems for this compound in heterogeneous HIV-1 subtypes?
Develop weighted scores using machine learning (e.g., random forests) trained on clinical outcomes (viral load reduction) and subtype-specific mutation frequencies. Cross-validate against global databases (e.g., Stanford HIVDB) and include accessory mutations (e.g., T69D) that modulate resistance. Ensure scores are updated as new subtype data emerge .
Q. What experimental approaches validate this compound’s degradation pathways under accelerated storage conditions?
Conduct forced degradation studies (heat, humidity, UV light) followed by LC-MS/MS to identify major degradants. Compare fragmentation patterns with reference standards (e.g., USP Related Compound A). Quantify degradation kinetics using Arrhenius modeling to predict shelf-life under standard storage .
Q. Methodological Best Practices
- Data Contradiction Analysis : Use meta-analytic frameworks (e.g., PRISMA) to harmonize disparate findings from clinical trials and preclinical studies. Stratify by variables like dosing regimen or patient CD4+ counts .
- Human Subject Research : Align participant selection with FDA guidelines (e.g., exclusion of HLA-B*5701-positive individuals to avoid hypersensitivity). Document informed consent and adverse events per IRB protocols .
Eigenschaften
IUPAC Name |
9-[5-(hydroxymethyl)oxolan-2-yl]-1H-purin-6-one | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C10H12N4O3/c15-3-6-1-2-7(17-6)14-5-13-8-9(14)11-4-12-10(8)16/h4-7,15H,1-3H2,(H,11,12,16) | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
BXZVVICBKDXVGW-UHFFFAOYSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CC(OC1CO)N2C=NC3=C2N=CNC3=O | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C10H12N4O3 | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID80860902 | |
| Record name | 9-[5-(Hydroxymethyl)oxolan-2-yl]-1,9-dihydro-6H-purin-6-one | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80860902 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
236.23 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
69655-05-6 | |
| Record name | 2', hydrate | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=612049 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Haftungsausschluss und Informationen zu In-Vitro-Forschungsprodukten
Bitte beachten Sie, dass alle Artikel und Produktinformationen, die auf BenchChem präsentiert werden, ausschließlich zu Informationszwecken bestimmt sind. Die auf BenchChem zum Kauf angebotenen Produkte sind speziell für In-vitro-Studien konzipiert, die außerhalb lebender Organismen durchgeführt werden. In-vitro-Studien, abgeleitet von dem lateinischen Begriff "in Glas", beinhalten Experimente, die in kontrollierten Laborumgebungen unter Verwendung von Zellen oder Geweben durchgeführt werden. Es ist wichtig zu beachten, dass diese Produkte nicht als Arzneimittel oder Medikamente eingestuft sind und keine Zulassung der FDA für die Vorbeugung, Behandlung oder Heilung von medizinischen Zuständen, Beschwerden oder Krankheiten erhalten haben. Wir müssen betonen, dass jede Form der körperlichen Einführung dieser Produkte in Menschen oder Tiere gesetzlich strikt untersagt ist. Es ist unerlässlich, sich an diese Richtlinien zu halten, um die Einhaltung rechtlicher und ethischer Standards in Forschung und Experiment zu gewährleisten.
















