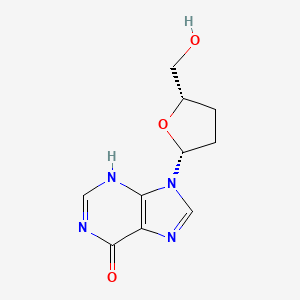
Didanosine
Descripción general
Descripción
Didanosina, también conocida como 2’,3’-dideoxiinosina, es un análogo de nucleósido sintético de la adenosina. Se utiliza principalmente como un medicamento antirretroviral para el tratamiento de la infección por el virus de la inmunodeficiencia humana (VIH). La didanosina actúa inhibiendo la enzima transcriptasa inversa, que es esencial para la replicación del VIH. Este compuesto fue descrito por primera vez en 1975 y aprobado para su uso en los Estados Unidos en 1991 .
Métodos De Preparación
Rutas Sintéticas y Condiciones de Reacción: La didanosina se sintetiza a través de un proceso de varios pasos que comienza con la inosina. Los pasos clave involucran la eliminación selectiva de los grupos hidroxilo en las posiciones 2' y 3' de la porción ribosa. Esto se logra típicamente a través de una serie de reacciones químicas que incluyen protección, desprotección y pasos de reducción selectiva.
Métodos de Producción Industrial: La producción industrial de didanosina implica síntesis a gran escala utilizando rutas químicas similares a las de la síntesis de laboratorio, pero optimizadas para mayores rendimientos y pureza. El proceso incluye medidas rigurosas de control de calidad para garantizar que el producto final cumpla con los estándares farmacéuticos.
Tipos de Reacciones:
Oxidación: La didanosina puede sufrir reacciones de oxidación, aunque estas no suelen ser relevantes para su actividad farmacológica.
Reducción: El compuesto en sí es una forma reducida de inosina, obtenida a través de pasos de reducción selectiva durante la síntesis.
Sustitución: La didanosina puede participar en reacciones de sustitución nucleofílica, particularmente en el anillo de purina.
Reactivos y Condiciones Comunes:
Oxidación: Agentes oxidantes comunes como el peróxido de hidrógeno o el permanganato de potasio.
Reducción: Agentes reductores como el borohidruro de sodio o el hidruro de litio y aluminio.
Sustitución: Nucleófilos como aminas o tioles en condiciones básicas.
Principales Productos Formados: El producto principal de estas reacciones suele ser la propia didanosina o sus derivados, dependiendo de las condiciones de reacción y los reactivos específicos utilizados.
Aplicaciones Científicas De Investigación
La didanosina tiene una amplia gama de aplicaciones en la investigación científica:
Química: Se utiliza como un compuesto modelo para estudiar análogos de nucleósidos y sus propiedades químicas.
Biología: Se utiliza en estudios de metabolismo de ácidos nucleicos e interacciones enzimáticas.
Medicina: Se utiliza ampliamente en la investigación sobre el tratamiento del VIH y los mecanismos de resistencia.
Industria: Se utiliza en el desarrollo de terapias antirretrovirales y en el estudio de interacciones medicamentosas.
Mecanismo De Acción
La didanosina se metaboliza intracelularmente a su forma activa, dideoxyadenosina trifosfato (ddATP). Este metabolito activo inhibe la enzima transcriptasa inversa del VIH al competir con el sustrato natural, deoxyadenosina trifosfato (dATP). Al incorporarse al ADN viral, el ddATP provoca la terminación de la cadena, evitando que el virus se replique .
Compuestos Similares:
Zidovudina (AZT): Otro inhibidor de la transcriptasa inversa de nucleósidos utilizado en el tratamiento del VIH.
Estavudina (d4T): Mecanismo de acción similar pero perfil de efectos secundarios diferente.
Lamivudina (3TC): A menudo se utiliza en combinación con otros antirretrovirales para obtener efectos sinérgicos.
Singularidad: La didanosina es única en sus modificaciones estructurales específicas, que confieren su actividad contra el VIH. A diferencia de otros análogos de nucleósidos, la didanosina tiene una base de hipoxantina unida al anillo de azúcar, que es crucial para su mecanismo de acción .
Comparación Con Compuestos Similares
Zidovudine (AZT): Another nucleoside reverse transcriptase inhibitor used in HIV treatment.
Stavudine (d4T): Similar mechanism of action but different side effect profile.
Lamivudine (3TC): Often used in combination with other antiretrovirals for synergistic effects.
Uniqueness: Didanosine is unique in its specific structural modifications, which confer its activity against HIV. Unlike other nucleoside analogues, this compound has a hypoxanthine base attached to the sugar ring, which is crucial for its mechanism of action .
Actividad Biológica
Didanosine (ddI), a nucleoside reverse transcriptase inhibitor (NRTI), has been primarily utilized in the treatment of HIV/AIDS. Its biological activity is characterized by its mechanism of action, pharmacokinetics, mutagenicity, and clinical efficacy. This article synthesizes findings from various studies to provide a comprehensive overview of this compound's biological activity.
This compound is metabolized intracellularly to its active form, this compound triphosphate (ddATP). The conversion involves phosphorylation through several enzymatic pathways, ultimately leading to the incorporation of ddATP into viral DNA during reverse transcription. This incorporation results in chain termination, effectively inhibiting viral replication. The intracellular half-life of ddATP is approximately 12-24 hours, suggesting that less frequent dosing may be sufficient compared to other NRTIs like zidovudine (AZT) .
Pharmacokinetics
This compound exhibits low plasma protein binding (≤ 5%) and is less lipophilic than zidovudine. Consequently, it has a different distribution profile, with lower concentrations in cerebrospinal fluid compared to plasma . Approximately 40% of this compound is excreted unchanged in urine, while the remainder is metabolized into hypoxanthine and uric acid .
Mutagenicity and Toxicity
Research indicates that this compound may exhibit clastogenic effects at high doses. In vitro studies have shown it can induce DNA damage in human lymphocytes and Chinese hamster ovary cells. However, animal studies have reported no significant increase in micronuclei formation at lower doses . Notably, this compound does not appear to induce reverse mutations in Salmonella typhimurium without metabolic activation, suggesting a limited mutagenic potential under certain conditions .
Table 1: Summary of Mutagenicity Studies on this compound
| Study Type | Results |
|---|---|
| In Vitro (Human Lymphocytes) | Clastogenic effects observed |
| In Vitro (Chinese Hamster Ovary) | Induced DNA damage |
| In Vivo (Rodent Micronucleus Assay) | No increase in micronuclei at low doses |
| Salmonella typhimurium | No reverse mutations without activation |
Clinical Efficacy
This compound has demonstrated significant antiviral activity in clinical settings. A study involving 168 HIV-1 infected patients showed that those treated with this compound experienced a median decrease in HIV-1 RNA levels by -0.56 log copies/mL compared to +0.07 log copies/mL in the placebo group after four weeks . Notably, 31% of patients on this compound achieved undetectable viral loads (<400 copies/mL), compared to only 6% in the placebo group .
Case Study: this compound in Treatment-Experienced Patients
In a cohort of treatment-experienced patients with multiple nucleoside reverse-transcriptase inhibitor-associated mutations (NAMs), this compound maintained antiviral efficacy. Patients with up to five NAMs at baseline still exhibited significant reductions in viral load when treated with this compound . This underscores its potential utility even in challenging treatment scenarios.
Q & A
Basic Research Questions
Q. What validated analytical methods are recommended for quantifying didanosine in pharmaceutical formulations?
The United States Pharmacopeia (USP) outlines a reversed-phase HPLC method for this compound quantification. A standard preparation using USP this compound RS is diluted to ~LC/900, and the assay preparation follows similar dilution. Chromatographic separation uses a 0.01 M ammonium acetate buffer (pH 6.0) and a C18 column. Peak responses are compared between standard and test solutions to calculate purity (90–110% of labeled claim) . For higher sensitivity, LC-MS with deuterated internal standards (e.g., this compound-d2) improves accuracy in pharmacokinetic studies .
Q. How should researchers handle this compound to ensure safety in laboratory settings?
this compound requires precautions to avoid inhalation, skin contact, or ingestion. Use PPE (gloves, lab coat, goggles) and work in a ventilated hood. Storage should be in sealed containers at 15–30°C to prevent degradation. In case of exposure, rinse affected areas with water for 15 minutes and seek medical evaluation due to its acute toxicity (oral LD₅₀: 4300 mg/kg in rats) and potential mutagenicity .
Q. What are the key considerations when designing in vitro studies to assess this compound’s antiviral efficacy?
Use HIV-1 reverse transcriptase (RT) inhibition assays with wild-type and mutant RT strains (e.g., L74V or K65R mutations linked to resistance). Cell-based models (e.g., MT-4 lymphocytes) should measure IC₅₀ values via plaque reduction or RT activity assays. Include controls for cytotoxicity and validate results against reference inhibitors like zidovudine .
Q. How can researchers ensure stability of this compound in solution during long-term experiments?
Prepare fresh solutions in pH-neutral buffers (e.g., PBS) and avoid prolonged exposure to light or temperatures >30°C. Degradation products (e.g., hypoxanthine) can be monitored via HPLC with UV detection at 250 nm. For multi-day assays, freeze aliquots at -20°C and validate stability over 72 hours .
Advanced Research Questions
Q. How do RT codon polymorphisms (e.g., L228H/R) influence this compound’s virological response in clinical cohorts?
Retrospective genotypic analysis of HIV-1 isolates from ART-experienced patients reveals that polymorphisms at RT codons 228 (L→H/R) correlate with reduced response to this compound. Use multivariate regression to adjust for confounding factors (e.g., prior NRTI exposure). Annotate sequences with resistance scores (e.g., ANRS algorithm) and validate findings in independent datasets to account for population-specific mutation patterns .
Q. What methodologies resolve contradictions in long-term toxicity data for this compound in preclinical models?
Mitochondrial toxicity assays (e.g., lactate/pyruvate ratios in hepatocytes) and in vivo models (e.g., transgenic mice expressing POLG mutations) can clarify mechanisms like mitochondrial DNA depletion. Combine histopathology (e.g., hepatic steatosis) with metabolomics to distinguish drug-specific effects from confounding variables (e.g., comorbidities) .
Q. How can researchers optimize genotypic resistance scoring systems for this compound in heterogeneous HIV-1 subtypes?
Develop weighted scores using machine learning (e.g., random forests) trained on clinical outcomes (viral load reduction) and subtype-specific mutation frequencies. Cross-validate against global databases (e.g., Stanford HIVDB) and include accessory mutations (e.g., T69D) that modulate resistance. Ensure scores are updated as new subtype data emerge .
Q. What experimental approaches validate this compound’s degradation pathways under accelerated storage conditions?
Conduct forced degradation studies (heat, humidity, UV light) followed by LC-MS/MS to identify major degradants. Compare fragmentation patterns with reference standards (e.g., USP Related Compound A). Quantify degradation kinetics using Arrhenius modeling to predict shelf-life under standard storage .
Q. Methodological Best Practices
- Data Contradiction Analysis : Use meta-analytic frameworks (e.g., PRISMA) to harmonize disparate findings from clinical trials and preclinical studies. Stratify by variables like dosing regimen or patient CD4+ counts .
- Human Subject Research : Align participant selection with FDA guidelines (e.g., exclusion of HLA-B*5701-positive individuals to avoid hypersensitivity). Document informed consent and adverse events per IRB protocols .
Propiedades
IUPAC Name |
9-[5-(hydroxymethyl)oxolan-2-yl]-1H-purin-6-one | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C10H12N4O3/c15-3-6-1-2-7(17-6)14-5-13-8-9(14)11-4-12-10(8)16/h4-7,15H,1-3H2,(H,11,12,16) | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
BXZVVICBKDXVGW-UHFFFAOYSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CC(OC1CO)N2C=NC3=C2N=CNC3=O | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C10H12N4O3 | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID80860902 | |
| Record name | 9-[5-(Hydroxymethyl)oxolan-2-yl]-1,9-dihydro-6H-purin-6-one | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80860902 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
236.23 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
69655-05-6 | |
| Record name | 2', hydrate | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=612049 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Descargo de responsabilidad e información sobre productos de investigación in vitro
Tenga en cuenta que todos los artículos e información de productos presentados en BenchChem están destinados únicamente con fines informativos. Los productos disponibles para la compra en BenchChem están diseñados específicamente para estudios in vitro, que se realizan fuera de organismos vivos. Los estudios in vitro, derivados del término latino "in vidrio", involucran experimentos realizados en entornos de laboratorio controlados utilizando células o tejidos. Es importante tener en cuenta que estos productos no se clasifican como medicamentos y no han recibido la aprobación de la FDA para la prevención, tratamiento o cura de ninguna condición médica, dolencia o enfermedad. Debemos enfatizar que cualquier forma de introducción corporal de estos productos en humanos o animales está estrictamente prohibida por ley. Es esencial adherirse a estas pautas para garantizar el cumplimiento de los estándares legales y éticos en la investigación y experimentación.















