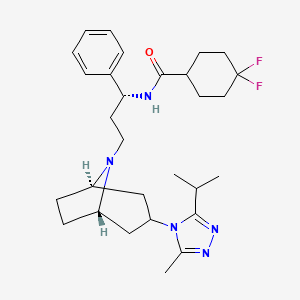
Maraviroc
Descripción general
Descripción
Maraviroc es un antagonista del receptor de quimiocinas desarrollado por Pfizer, comercializado bajo los nombres comerciales Selzentry en los Estados Unidos y Celsentri en la Unión Europea . Se utiliza principalmente como un medicamento antirretroviral para el tratamiento de la infección por VIH-1 tropa CCR5 . This compound actúa bloqueando el receptor CCR5 en la superficie de ciertas células humanas, lo que evita que el virus del VIH ingrese a estas células .
Métodos De Preparación
Maraviroc puede sintetizarse utilizando varios métodos. Este método implica la alquilación directa de una amina con un alcohol en condiciones tecnológicamente aceptables . El proceso incluye pasos de aislamiento y purificación mejorados para obtener this compound de alta pureza . Los métodos de producción industrial suelen seguir rutas sintéticas similares, pero se optimizan para la fabricación a gran escala para garantizar la coherencia y la eficiencia .
Análisis De Reacciones Químicas
Maraviroc sufre varios tipos de reacciones químicas, que incluyen:
Oxidación: this compound puede oxidarse en condiciones específicas, lo que lleva a la formación de varios productos de oxidación.
Reducción: Las reacciones de reducción pueden modificar los grupos funcionales dentro de this compound, alterando sus propiedades químicas.
Sustitución: This compound puede sufrir reacciones de sustitución en las que un grupo funcional es reemplazado por otro. Los reactivos comunes utilizados en estas reacciones incluyen agentes oxidantes, agentes reductores y varios catalizadores.
Aplicaciones Científicas De Investigación
Antiretroviral Therapy
Efficacy in HIV Treatment
Maraviroc is primarily indicated for treatment-experienced patients infected with R5-tropic HIV-1. Clinical trials, such as the MOTIVATE studies, have demonstrated its efficacy in reducing viral loads and improving immunological responses. In these studies, patients treated with this compound showed significantly greater reductions in HIV-1 RNA levels compared to those receiving placebo, with durable responses observed over extended follow-ups .
Long-Term Safety Profile
A five-year safety evaluation indicated that this compound was generally well-tolerated among patients, with low rates of adverse events and no significant increase in serious clinical outcomes like hepatic failure or malignancy . This safety profile supports its continued use in long-term antiretroviral therapy.
Microbicide Development
Topical Application for HIV Prevention
Recent research has explored this compound's potential as a topical microbicide. A study demonstrated that a this compound gel formulation provided complete protection against vaginal HIV-1 challenges in humanized mice models . This finding suggests that this compound could be developed as an effective microbicide to prevent sexual transmission of HIV, particularly in populations at high risk.
| Study | Model | Outcome |
|---|---|---|
| PLOS One (2011) | Humanized mice | Complete protection against vaginal HIV-1 challenge with this compound gel |
| Nature (2020) | Patient cohort | Reactivation of latent HIV with prolonged this compound administration |
Reactivation of Latent HIV
Role as a Latency-Reversing Agent
This compound has been investigated for its ability to reactivate latent HIV reservoirs. A study indicated that prolonged administration of this compound led to increased levels of viral RNA expression in patients previously on suppressive antiretroviral therapy, suggesting its potential role as a latency-reversing agent . This application is crucial for strategies aimed at achieving a functional cure for HIV.
Immunological Effects
Impact on T-cell Activation
Research has shown that this compound intensification can unexpectedly increase T-cell activation within peripheral blood and rectal mucosa during treated HIV infection. This phenomenon raises questions about the drug's effects on immune system dynamics and its potential implications for long-term treatment strategies .
Neuroprotection Research
Exploration in Neuroinflammatory Conditions
Recent studies have examined this compound's effects beyond HIV treatment, particularly regarding neuroprotection. It has been shown to reduce inflammation in vitro without exerting direct cytotoxic effects on astroglial cells, indicating potential applications in neuroinflammatory diseases . Although these findings are preliminary, they suggest a broader therapeutic scope for this compound.
Mecanismo De Acción
Maraviroc funciona como un inhibidor de la entrada al unirse selectivamente al receptor CCR5 en la superficie de las células humanas . Esta unión evita que la proteína gp120 del VIH-1 se asocie con el receptor CCR5, bloqueando así la entrada del virus a la célula huésped . El fármaco actúa como un modulador alostérico negativo del receptor CCR5, induciendo un cambio conformacional que inhibe la interacción entre el receptor y el virus .
Comparación Con Compuestos Similares
Maraviroc es único entre los agentes antirretrovirales, ya que se dirige a un receptor humano en lugar del virus en sí . Los compuestos similares incluyen otros antagonistas de CCR5, como:
Vicriviroc: Otro antagonista de CCR5 con mecanismos de acción similares pero diferentes propiedades farmacocinéticas.
Actividad Biológica
Maraviroc, also known as UK-427857, is a selective antagonist of the CCR5 receptor, which plays a crucial role in the entry of HIV-1 into host cells. This compound represents the first drug in a new class of antiretroviral agents that target host proteins rather than viral components. Its unique mechanism of action and pharmacological properties have made it a significant focus of research in the treatment of HIV-1 infections.
This compound inhibits HIV-1 entry by binding to the CCR5 receptor on the surface of CD4+ T cells. This binding prevents the viral envelope protein gp120 from interacting with CCR5, effectively blocking the fusion process necessary for viral entry into host cells. The geometric mean 90% inhibitory concentration (IC90) for this compound against various CCR5-tropic HIV-1 strains is approximately 2.0 nM, demonstrating its potent antiviral activity against a wide range of viral isolates .
In Vitro Studies
In vitro studies have shown that this compound exhibits:
- Potent Anti-HIV Activity : Effective against 200 clinically derived HIV-1 envelope-recombinant pseudoviruses, including those resistant to other drug classes.
- Selectivity for CCR5 : this compound has been confirmed to be highly selective for the CCR5 receptor, with no significant effects on other receptors or enzymes, including hERG ion channels, indicating a favorable safety profile .
Pharmacokinetics
This compound is characterized by its oral bioavailability and pharmacokinetic properties that support once or twice daily dosing. Preclinical models predict human pharmacokinetics consistent with these dosing regimens, allowing for convenient administration in clinical settings .
Clinical Efficacy
Clinical trials have demonstrated that this compound effectively reduces viral load in patients with CCR5-tropic HIV-1. It has been shown to improve immune function and reduce the risk of disease progression in treated individuals. The drug's efficacy is further supported by studies highlighting its ability to maintain viral suppression over extended periods .
Table 1: Summary of this compound's Biological Activity
| Property | Value |
|---|---|
| Chemical Name | 4,4-Difluoro-N-{(1S)-3-[3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]oct-8-yl]-1-phenylpropyl}cyclohexanecarboxamide |
| IC90 Against CCR5-tropic HIV-1 | 2.0 nM |
| Selectivity Index (hERG) | >10 μM |
| Oral Bioavailability | Yes |
| Recommended Dosing | Once or twice daily |
Table 2: Clinical Trial Results for this compound
| Study Phase | Participants | Outcome |
|---|---|---|
| Phase II | 200 | Significant reduction in viral load |
| Phase III | 500 | Improved CD4+ T cell counts |
| Long-term follow-up | 300 | Sustained viral suppression |
Case Study 1: Efficacy in Treatment-Experienced Patients
A study involving treatment-experienced patients showed that those receiving this compound as part of their regimen experienced a significant decline in plasma HIV RNA levels compared to those on standard therapy alone. This highlights this compound's potential as an effective option for patients with limited treatment choices due to resistance .
Case Study 2: Safety Profile Assessment
In a long-term safety assessment involving over 1000 participants, this compound demonstrated a favorable safety profile with minimal adverse effects reported. The most common side effects were mild and included headaches and gastrointestinal disturbances. Importantly, no significant cardiovascular events were noted during the trial period .
Propiedades
IUPAC Name |
4,4-difluoro-N-[3-[3-(3-methyl-5-propan-2-yl-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octan-8-yl]-1-phenylpropyl]cyclohexane-1-carboxamide | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C29H41F2N5O/c1-19(2)27-34-33-20(3)36(27)25-17-23-9-10-24(18-25)35(23)16-13-26(21-7-5-4-6-8-21)32-28(37)22-11-14-29(30,31)15-12-22/h4-8,19,22-26H,9-18H2,1-3H3,(H,32,37) | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
GSNHKUDZZFZSJB-UHFFFAOYSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=NN=C(N1C2CC3CCC(C2)N3CCC(C4=CC=CC=C4)NC(=O)C5CCC(CC5)(F)F)C(C)C | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C29H41F2N5O | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
513.7 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Descargo de responsabilidad e información sobre productos de investigación in vitro
Tenga en cuenta que todos los artículos e información de productos presentados en BenchChem están destinados únicamente con fines informativos. Los productos disponibles para la compra en BenchChem están diseñados específicamente para estudios in vitro, que se realizan fuera de organismos vivos. Los estudios in vitro, derivados del término latino "in vidrio", involucran experimentos realizados en entornos de laboratorio controlados utilizando células o tejidos. Es importante tener en cuenta que estos productos no se clasifican como medicamentos y no han recibido la aprobación de la FDA para la prevención, tratamiento o cura de ninguna condición médica, dolencia o enfermedad. Debemos enfatizar que cualquier forma de introducción corporal de estos productos en humanos o animales está estrictamente prohibida por ley. Es esencial adherirse a estas pautas para garantizar el cumplimiento de los estándares legales y éticos en la investigación y experimentación.















