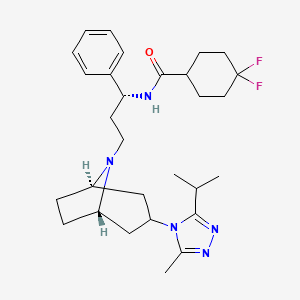
Maraviroc
Vue d'ensemble
Description
Maraviroc est un antagoniste du récepteur des chimiokines développé par Pfizer, commercialisé sous les noms de marque Selzentry aux États-Unis et Celsentri dans l'Union européenne . Il est principalement utilisé comme médicament antirétroviral pour le traitement de l'infection à VIH-1 tropique pour le CCR5 . This compound agit en bloquant le récepteur CCR5 à la surface de certaines cellules humaines, empêchant le virus du VIH de pénétrer dans ces cellules .
Méthodes De Préparation
Maraviroc peut être synthétisé à l'aide de diverses méthodes. Cette méthode implique l'alkylation directe d'une amine avec un alcool dans des conditions technologiquement acceptables . Le procédé comprend des étapes d'isolement et de purification améliorées pour obtenir du this compound de haute pureté . Les méthodes de production industrielle suivent généralement des voies de synthèse similaires mais sont optimisées pour une fabrication à grande échelle afin d'assurer la cohérence et l'efficacité .
Analyse Des Réactions Chimiques
Maraviroc subit plusieurs types de réactions chimiques, notamment:
Oxydation: this compound peut être oxydé dans des conditions spécifiques, conduisant à la formation de divers produits d'oxydation.
Réduction: Les réactions de réduction peuvent modifier les groupes fonctionnels dans this compound, modifiant ses propriétés chimiques.
Substitution: This compound peut subir des réactions de substitution où un groupe fonctionnel est remplacé par un autre. Les réactifs couramment utilisés dans ces réactions comprennent les agents oxydants, les agents réducteurs et divers catalyseurs.
Applications De Recherche Scientifique
Antiretroviral Therapy
Efficacy in HIV Treatment
Maraviroc is primarily indicated for treatment-experienced patients infected with R5-tropic HIV-1. Clinical trials, such as the MOTIVATE studies, have demonstrated its efficacy in reducing viral loads and improving immunological responses. In these studies, patients treated with this compound showed significantly greater reductions in HIV-1 RNA levels compared to those receiving placebo, with durable responses observed over extended follow-ups .
Long-Term Safety Profile
A five-year safety evaluation indicated that this compound was generally well-tolerated among patients, with low rates of adverse events and no significant increase in serious clinical outcomes like hepatic failure or malignancy . This safety profile supports its continued use in long-term antiretroviral therapy.
Microbicide Development
Topical Application for HIV Prevention
Recent research has explored this compound's potential as a topical microbicide. A study demonstrated that a this compound gel formulation provided complete protection against vaginal HIV-1 challenges in humanized mice models . This finding suggests that this compound could be developed as an effective microbicide to prevent sexual transmission of HIV, particularly in populations at high risk.
| Study | Model | Outcome |
|---|---|---|
| PLOS One (2011) | Humanized mice | Complete protection against vaginal HIV-1 challenge with this compound gel |
| Nature (2020) | Patient cohort | Reactivation of latent HIV with prolonged this compound administration |
Reactivation of Latent HIV
Role as a Latency-Reversing Agent
This compound has been investigated for its ability to reactivate latent HIV reservoirs. A study indicated that prolonged administration of this compound led to increased levels of viral RNA expression in patients previously on suppressive antiretroviral therapy, suggesting its potential role as a latency-reversing agent . This application is crucial for strategies aimed at achieving a functional cure for HIV.
Immunological Effects
Impact on T-cell Activation
Research has shown that this compound intensification can unexpectedly increase T-cell activation within peripheral blood and rectal mucosa during treated HIV infection. This phenomenon raises questions about the drug's effects on immune system dynamics and its potential implications for long-term treatment strategies .
Neuroprotection Research
Exploration in Neuroinflammatory Conditions
Recent studies have examined this compound's effects beyond HIV treatment, particularly regarding neuroprotection. It has been shown to reduce inflammation in vitro without exerting direct cytotoxic effects on astroglial cells, indicating potential applications in neuroinflammatory diseases . Although these findings are preliminary, they suggest a broader therapeutic scope for this compound.
Mécanisme D'action
Maraviroc functions as an entry inhibitor by selectively binding to the CCR5 receptor on the surface of human cells . This binding prevents the HIV-1 gp120 protein from associating with the CCR5 receptor, thereby blocking the virus from entering the host cell . The drug acts as a negative allosteric modulator of the CCR5 receptor, inducing a conformational change that inhibits the interaction between the receptor and the virus .
Comparaison Avec Des Composés Similaires
Maraviroc est unique parmi les agents antirétroviraux car il cible un récepteur humain plutôt que le virus lui-même . Des composés similaires comprennent d'autres antagonistes du CCR5, tels que:
Vicriviroc: Un autre antagoniste du CCR5 avec des mécanismes d'action similaires mais des propriétés pharmacocinétiques différentes.
Activité Biologique
Maraviroc, also known as UK-427857, is a selective antagonist of the CCR5 receptor, which plays a crucial role in the entry of HIV-1 into host cells. This compound represents the first drug in a new class of antiretroviral agents that target host proteins rather than viral components. Its unique mechanism of action and pharmacological properties have made it a significant focus of research in the treatment of HIV-1 infections.
This compound inhibits HIV-1 entry by binding to the CCR5 receptor on the surface of CD4+ T cells. This binding prevents the viral envelope protein gp120 from interacting with CCR5, effectively blocking the fusion process necessary for viral entry into host cells. The geometric mean 90% inhibitory concentration (IC90) for this compound against various CCR5-tropic HIV-1 strains is approximately 2.0 nM, demonstrating its potent antiviral activity against a wide range of viral isolates .
In Vitro Studies
In vitro studies have shown that this compound exhibits:
- Potent Anti-HIV Activity : Effective against 200 clinically derived HIV-1 envelope-recombinant pseudoviruses, including those resistant to other drug classes.
- Selectivity for CCR5 : this compound has been confirmed to be highly selective for the CCR5 receptor, with no significant effects on other receptors or enzymes, including hERG ion channels, indicating a favorable safety profile .
Pharmacokinetics
This compound is characterized by its oral bioavailability and pharmacokinetic properties that support once or twice daily dosing. Preclinical models predict human pharmacokinetics consistent with these dosing regimens, allowing for convenient administration in clinical settings .
Clinical Efficacy
Clinical trials have demonstrated that this compound effectively reduces viral load in patients with CCR5-tropic HIV-1. It has been shown to improve immune function and reduce the risk of disease progression in treated individuals. The drug's efficacy is further supported by studies highlighting its ability to maintain viral suppression over extended periods .
Table 1: Summary of this compound's Biological Activity
| Property | Value |
|---|---|
| Chemical Name | 4,4-Difluoro-N-{(1S)-3-[3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]oct-8-yl]-1-phenylpropyl}cyclohexanecarboxamide |
| IC90 Against CCR5-tropic HIV-1 | 2.0 nM |
| Selectivity Index (hERG) | >10 μM |
| Oral Bioavailability | Yes |
| Recommended Dosing | Once or twice daily |
Table 2: Clinical Trial Results for this compound
| Study Phase | Participants | Outcome |
|---|---|---|
| Phase II | 200 | Significant reduction in viral load |
| Phase III | 500 | Improved CD4+ T cell counts |
| Long-term follow-up | 300 | Sustained viral suppression |
Case Study 1: Efficacy in Treatment-Experienced Patients
A study involving treatment-experienced patients showed that those receiving this compound as part of their regimen experienced a significant decline in plasma HIV RNA levels compared to those on standard therapy alone. This highlights this compound's potential as an effective option for patients with limited treatment choices due to resistance .
Case Study 2: Safety Profile Assessment
In a long-term safety assessment involving over 1000 participants, this compound demonstrated a favorable safety profile with minimal adverse effects reported. The most common side effects were mild and included headaches and gastrointestinal disturbances. Importantly, no significant cardiovascular events were noted during the trial period .
Propriétés
IUPAC Name |
4,4-difluoro-N-[3-[3-(3-methyl-5-propan-2-yl-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octan-8-yl]-1-phenylpropyl]cyclohexane-1-carboxamide | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C29H41F2N5O/c1-19(2)27-34-33-20(3)36(27)25-17-23-9-10-24(18-25)35(23)16-13-26(21-7-5-4-6-8-21)32-28(37)22-11-14-29(30,31)15-12-22/h4-8,19,22-26H,9-18H2,1-3H3,(H,32,37) | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
GSNHKUDZZFZSJB-UHFFFAOYSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=NN=C(N1C2CC3CCC(C2)N3CCC(C4=CC=CC=C4)NC(=O)C5CCC(CC5)(F)F)C(C)C | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C29H41F2N5O | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
513.7 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Avertissement et informations sur les produits de recherche in vitro
Veuillez noter que tous les articles et informations sur les produits présentés sur BenchChem sont destinés uniquement à des fins informatives. Les produits disponibles à l'achat sur BenchChem sont spécifiquement conçus pour des études in vitro, qui sont réalisées en dehors des organismes vivants. Les études in vitro, dérivées du terme latin "in verre", impliquent des expériences réalisées dans des environnements de laboratoire contrôlés à l'aide de cellules ou de tissus. Il est important de noter que ces produits ne sont pas classés comme médicaments et n'ont pas reçu l'approbation de la FDA pour la prévention, le traitement ou la guérison de toute condition médicale, affection ou maladie. Nous devons souligner que toute forme d'introduction corporelle de ces produits chez les humains ou les animaux est strictement interdite par la loi. Il est essentiel de respecter ces directives pour assurer la conformité aux normes légales et éthiques en matière de recherche et d'expérimentation.
















