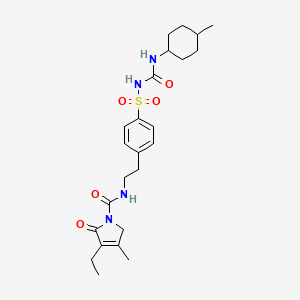
Glimepiride
Vue d'ensemble
Description
Le glimépiride est un médicament antidiabétique appartenant à la classe des sulfonylurées, principalement prescrit pour la prise en charge du diabète de type 2. Il est considéré comme une option de deuxième intention par rapport à la metformine, en raison de la sécurité et de l'efficacité bien établies de la metformine . Le glimépiride agit principalement en augmentant la quantité d'insuline libérée par le pancréas . Il a été breveté en 1979 et approuvé pour une utilisation médicale en 1995 .
Méthodes De Préparation
Voies de synthèse et conditions de réaction
Le glimépiride est synthétisé par un processus en plusieurs étapes impliquant plusieurs intermédiaires. Les étapes clés comprennent la formation du cycle pyrrole et la fixation du groupe sulfonylurée. Les conditions de réaction impliquent généralement l'utilisation de solvants organiques, de catalyseurs et de températures contrôlées pour garantir les transformations chimiques souhaitées .
Méthodes de production industrielle
La production industrielle de glimépiride implique une synthèse à grande échelle utilisant des conditions de réaction optimisées pour maximiser le rendement et la pureté. Le processus comprend des étapes de purification telles que la cristallisation et la filtration pour obtenir le produit final sous une forme adaptée à un usage pharmaceutique .
Analyse Des Réactions Chimiques
Types de réactions
Le glimépiride subit diverses réactions chimiques, notamment :
Oxydation : Implique l'ajout d'oxygène ou l'élimination d'hydrogène.
Réduction : Implique l'ajout d'hydrogène ou l'élimination d'oxygène.
Substitution : Implique le remplacement d'un groupe fonctionnel par un autre.
Réactifs et conditions courants
Les réactifs courants utilisés dans ces réactions comprennent des agents oxydants comme le permanganate de potassium, des agents réducteurs comme le borohydrure de sodium et divers catalyseurs pour faciliter les réactions .
Principaux produits formés
Les principaux produits formés à partir de ces réactions dépendent des conditions et des réactifs spécifiques utilisés. Par exemple, l'oxydation du glimépiride peut conduire à la formation de sulfoxydes ou de sulfones .
Applications de la recherche scientifique
Le glimépiride a un large éventail d'applications de recherche scientifique :
Chimie : Utilisé comme composé modèle pour étudier le comportement des sulfonylurées et de leurs dérivés.
Biologie : Étudié pour ses effets sur les processus cellulaires et la sécrétion d'insuline.
Industrie : Utilisé dans le développement de nouvelles formulations pharmaceutiques et systèmes d'administration de médicaments.
Mécanisme d'action
Le glimépiride agit comme un sécrétagogue d'insuline. Il stimule la libération d'insuline par les cellules bêta pancréatiques en bloquant les canaux potassiques sensibles à l'ATP, ce qui provoque une dépolarisation des cellules bêta et une sécrétion d'insuline subséquente . De plus, il augmente l'activité des récepteurs intracellulaires de l'insuline, améliorant la réponse de l'organisme à l'insuline .
Applications De Recherche Scientifique
Glimepiride is a second-generation sulfonylurea drug used to manage type 2 diabetes mellitus (T2DM) by improving glycemic control . It stimulates the secretion of insulin from pancreatic beta cells and enhances the sensitivity of peripheral tissues to insulin, increasing glucose uptake and reducing blood glucose and glycated hemoglobin (HbA1c) levels .
Clinical Applications and Efficacy
Monotherapy: this compound is effective as a monotherapy for patients with T2DM who have not achieved adequate glycemic control through diet alone . A randomized, placebo-controlled trial demonstrated that this compound (1–8 mg) significantly reduced fasting plasma glucose (FPG) by 46 mg/dL, post-prandial glucose (PPG) by 72 mg/dL, and HbA1c by 1.4% compared to placebo . In this study, 69% of patients taking this compound achieved good glycemic control (HbA1c ≤ 7.2%) compared to 32% on placebo .
Combination Therapy: this compound can be used in combination with other antidiabetic medications, such as metformin, to improve glycemic control .
Cardiovascular Benefits: Long-term use of this compound is associated with better survival rates and reduced hospitalizations for heart failure, acute myocardial infarction, and stroke in patients with T2DM and congestive heart failure (CHF) . High-dose this compound may offer greater cardiovascular protection compared to low-dose regimens . The cardiovascular protective effect of this compound may be related to increased levels of epoxyeicosatrienoic acid (EET) through soluble epoxide hydrolase (sEH) inhibition .
Comparison with Other Sulfonylureas: Studies suggest that this compound may have a beneficial effect on insulin resistance compared to glibenclamide . this compound treatment was associated with increased adiponectin concentrations and reduced waist circumference, while glibenclamide showed an inverse correlation between changes in serum adiponectin and HOMA-IR .
Dosing and Administration
This compound is typically administered orally once daily, with dosages ranging from 1 to 8 mg . Some studies have explored the pharmacokinetics and pharmacodynamics of once- versus twice-daily dosing, with varying results . One study found lower glucose levels throughout the day with once-daily dosing compared to twice-daily, while another observed a significant decrease in FPG with twice-daily administration .
Safety and Tolerability
Mécanisme D'action
Glimepiride acts as an insulin secretagogue. It stimulates the release of insulin from pancreatic beta cells by blocking ATP-sensitive potassium channels, causing depolarization of the beta cells and subsequent insulin secretion . Additionally, it increases the activity of intracellular insulin receptors, enhancing the body’s response to insulin .
Comparaison Avec Des Composés Similaires
Composés similaires
- Glipizide
- Glyburide
- Metformin
- Sémaglutide
Unicité
Le glimépiride est unique parmi les sulfonylurées en raison de sa durée d'action plus longue et de son risque moindre d'hypoglycémie par rapport aux autres sulfonylurées de deuxième génération . Il a également moins d'effets cardiovasculaires, ce qui en fait une option plus sûre pour les patients ayant des problèmes cardiovasculaires .
Activité Biologique
Glimepiride is an oral antidiabetic medication belonging to the sulfonylurea class, primarily used in the management of type 2 diabetes mellitus (T2DM). Its primary mechanism of action involves stimulating insulin secretion from pancreatic beta cells, thereby lowering blood glucose levels. This article delves into the biological activity of this compound, supported by various studies, case reports, and clinical trials.
This compound functions by binding to ATP-sensitive potassium channels (K_ATP channels) on the plasma membrane of pancreatic beta cells. This binding leads to cell membrane depolarization, which opens voltage-dependent calcium channels. The influx of calcium ions triggers the exocytosis of insulin granules into the bloodstream. The overall process can be summarized as follows:
- Binding to K_ATP Channels : this compound inhibits these channels, preventing potassium from exiting the cell.
- Depolarization : This inhibition causes depolarization of the beta cell membrane.
- Calcium Influx : Voltage-dependent calcium channels open, allowing calcium to enter the cell.
- Insulin Secretion : Increased intracellular calcium stimulates insulin release.
Additionally, this compound enhances peripheral insulin sensitivity and promotes glucose uptake in muscle and adipose tissues by increasing the translocation of GLUT4 transporters .
Pharmacokinetics
This compound is well-absorbed after oral administration, with peak plasma concentrations typically occurring within 2-3 hours. It undergoes hepatic metabolism primarily via the cytochrome P450 enzyme CYP2C9, producing active metabolites that contribute to its pharmacological effects. The elimination half-life is approximately 5-9 hours .
Glycemic Control
Several clinical studies have demonstrated the efficacy of this compound in controlling blood glucose levels:
- In a randomized controlled trial involving 249 patients with T2DM, this compound significantly reduced fasting plasma glucose (FPG) by an average of 46 mg/dL and hemoglobin A1c (HbA1c) by 1.4% compared to placebo .
- A meta-analysis indicated that this compound treatment led to a reduction in postprandial glucose levels by 72 mg/dL more than placebo .
| Study | Population | This compound Dose | FPG Reduction | HbA1c Reduction |
|---|---|---|---|---|
| Goldberg et al. (2012) | 304 T2DM patients | 1-8 mg/day | 43-74 mg/dL | 1.2%-1.9% |
| Placebo-Controlled Trial (1998) | 249 patients | 1-8 mg/day | 46 mg/dL | 1.4% |
Extrapancreatic Effects
This compound also exhibits extrapancreatic effects that enhance insulin sensitivity and promote glucose utilization in peripheral tissues. In vitro studies suggest that it may potentiate lipogenesis and glycogenesis, although its clinical relevance remains uncertain .
Safety Profile
The safety profile of this compound has been extensively studied:
- A large-scale study indicated that the incidence of hypoglycemia was low (0.9%) among patients using this compound over an extended period .
- Cardiovascular safety has been supported by trials such as CARMELINA and CAROLINA, which showed no significant increase in major adverse cardiovascular events compared to other antidiabetic agents .
Case Studies and Observations
Case studies have highlighted the effectiveness of this compound in specific patient populations:
- Elderly Patients : In older adults with T2DM, this compound has been shown to effectively lower blood glucose levels without significant adverse effects.
- Patients with Comorbidities : In patients with concurrent heart failure and diabetes, this compound use was associated with reduced all-cause mortality and hospitalizations .
Q & A
Basic Research Questions
Q. What validated analytical methods are recommended for detecting glimepiride in biological matrices?
To ensure reproducibility, use gas chromatography-mass spectrometry (GC-MS) with derivatization. Derivatize this compound using agents like N-methyl-N-(trimethylsilyl) trifluoroacetamide (MSTFA) to improve thermal stability and volatility. Validate the method using a Plackett-Burman experimental design (8-run, 4 variables) to assess robustness and ruggedness, with peak area as the response variable .
Q. How should dissolution rate studies for hydrophobic this compound formulations be designed?
Employ the slurry method to enhance dissolution. Validate dissolution profiles using high-performance liquid chromatography (HPLC) per International Council for Harmonisation (ICH) guidelines. Characterize formulations via X-ray diffraction (XRD) to confirm crystalline/amorphous transitions (2θ range: 2°–40°, step width 2°/min) .
Q. What statistical approaches are suitable for analyzing this compound pharmacokinetic data?
Use one-tailed t-tests (Type 2 error) and ANOVA for robustness testing. For multi-variable interactions, apply Plackett-Burman designs to isolate significant factors (e.g., pH, temperature) affecting analytical responses like peak area .
Advanced Research Questions
Q. How can contradictions in this compound-protein binding affinity data be resolved across studies?
Perform frontal analysis using high-performance affinity chromatography (HPAC) with human serum albumin (HSA) columns. Compare one-site vs. two-site binding models using residual plots and F-tests. For example, this compound binding at 0.5–50 μM concentrations showed deviations at low concentrations, favoring a two-site model .
Q. What experimental strategies optimize this compound polymorph synthesis and characterization?
Synthesize novel polymorphs via solvent evaporation or crystallization. Characterize using ultra-performance liquid chromatography (UPLC) for thermodynamic solubility and XRD for structural analysis. Rod-shaped crystalline structures (observed at 10X–100X magnification) indicate distinct polymorphic forms .
Q. How does this compound modulate neurodegenerative pathways in prion disease models?
In vitro, this compound activates glycosylphosphatidylinositol-specific phospholipase C (GPI-PLC), shedding cellular prion protein (PrP<sup>C</sup>) and reducing PrP<sup>Sc</sup> formation. Use neuronal cell lines exposed to PrP82–146 for survival assays and cPLA2 activity measurements to validate neuroprotective effects .
Q. What methodologies address this compound’s multi-site interactions in complex biological systems?
Combine HPAC with competitive binding assays. For example, frontal analysis of this compound-HSA interactions revealed two binding sites with average relative standard deviations of ±0.04–3.2%. Validate using equilibrium dialysis or surface plasmon resonance (SPR) for kinetic parameter estimation .
Q. Methodological Considerations
- Data Imputation in Clinical Trials: For longitudinal studies (e.g., 104-week HbA1c comparisons), apply multiple imputation to handle missing data (e.g., 13.9% imputed for this compound arms). Use ANCOVA with last observation carried forward (LOCF) to maintain statistical power .
- Reference Standards: Source certified this compound reference compounds (e.g., Related Compounds A, B, C) for impurity profiling. Store at controlled temperatures (±2°C) to prevent degradation .
Propriétés
IUPAC Name |
4-ethyl-3-methyl-N-[2-[4-[(4-methylcyclohexyl)carbamoylsulfamoyl]phenyl]ethyl]-5-oxo-2H-pyrrole-1-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C24H34N4O5S/c1-4-21-17(3)15-28(22(21)29)24(31)25-14-13-18-7-11-20(12-8-18)34(32,33)27-23(30)26-19-9-5-16(2)6-10-19/h7-8,11-12,16,19H,4-6,9-10,13-15H2,1-3H3,(H,25,31)(H2,26,27,30) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
WIGIZIANZCJQQY-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCC1=C(CN(C1=O)C(=O)NCCC2=CC=C(C=C2)S(=O)(=O)NC(=O)NC3CCC(CC3)C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C24H34N4O5S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID5040675, DTXSID20861130 | |
| Record name | Glimepiride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5040675 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 3-Ethyl-2,5-dihydro-4-methyl-N-[2-[4-[[[[(4-methylcyclohexyl)amino]carbonyl]amino]sulfonyl]phenyl]ethyl]-2-oxo-1H-pyrrole-1-carboxamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID20861130 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
490.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Glimepiride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014367 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
>73.6 [ug/mL] (The mean of the results at pH 7.4), Partly miscible, 3.84e-02 g/L | |
| Record name | SID49648856 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Glimepiride | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00222 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Glimepiride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014367 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
ATP-sensitive potassium channels on pancreatic beta cells that are gated by intracellular ATP and ADP. The hetero-octomeric complex of the channel is composed of four pore-forming Kir6.2 subunits and four regulatory sulfonylurea receptor (SUR) subunits. Alternative splicing allows the formation of channels composed of varying subunit isoforms expressed at different concentrations in different tissues. In pancreatic beta cells, ATP-sensitive potassium channels play a role as essential metabolic sensors and regulators that couple membrane excitability with glucose-stimulated insulin secretion (GSIS). When there is a decrease in the ATP:ADP ratio, the channels are activated and open, leading to K+ efflux from the cell, membrane hyperpolarization, and suppression of insulin secretion. In contrast, increased uptake of glucose into the cell leads to elevated intracellular ATP:ADP ratio, leading to the closure of channels and membrane depolarization. Depolarization leads to activation and opening of the voltage-dependent Ca2+ channels and consequently an influx of calcium ions into the cell. Elevated intracellular calcium levels causes the contraction of the filaments of actomyosin responsible for the exocytosis of insulin granules stored in vesicles. Glimepiride blocks the ATP-sensitive potassium channel by binding non-specifically to the B sites of both sulfonylurea receptor-1 (SUR1) and sulfonylurea receptor-2A (SUR2A) subunits as well as the A site of SUR1 subunit of the channel to promote insulin secretion from the beta cell. | |
| Record name | Glimepiride | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00222 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
261361-60-8, 93479-97-1, 684286-46-2 | |
| Record name | 3-Ethyl-2,5-dihydro-4-methyl-N-[2-[4-[[[[(4-methylcyclohexyl)amino]carbonyl]amino]sulfonyl]phenyl]ethyl]-2-oxo-1H-pyrrole-1-carboxamide | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=261361-60-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Glimepiride [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0093479971 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Glimepiride, cis- | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0684286462 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Glimepiride | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00222 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | glimepiride | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759809 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Glimepiride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5040675 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 3-Ethyl-2,5-dihydro-4-methyl-N-[2-[4-[[[[(4-methylcyclohexyl)amino]carbonyl]amino]sulfonyl]phenyl]ethyl]-2-oxo-1H-pyrrole-1-carboxamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID20861130 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Glimepiride | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | GLIMEPIRIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/6KY687524K | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | GLIMEPIRIDE, CIS- | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/24T6XIR2MZ | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Glimepiride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014367 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
207 °C | |
| Record name | Glimepiride | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00222 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Glimepiride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014367 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Avertissement et informations sur les produits de recherche in vitro
Veuillez noter que tous les articles et informations sur les produits présentés sur BenchChem sont destinés uniquement à des fins informatives. Les produits disponibles à l'achat sur BenchChem sont spécifiquement conçus pour des études in vitro, qui sont réalisées en dehors des organismes vivants. Les études in vitro, dérivées du terme latin "in verre", impliquent des expériences réalisées dans des environnements de laboratoire contrôlés à l'aide de cellules ou de tissus. Il est important de noter que ces produits ne sont pas classés comme médicaments et n'ont pas reçu l'approbation de la FDA pour la prévention, le traitement ou la guérison de toute condition médicale, affection ou maladie. Nous devons souligner que toute forme d'introduction corporelle de ces produits chez les humains ou les animaux est strictement interdite par la loi. Il est essentiel de respecter ces directives pour assurer la conformité aux normes légales et éthiques en matière de recherche et d'expérimentation.
















