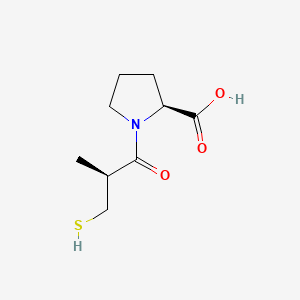
Captopril
概要
説明
カプトプリルは、アンジオテンシンIをアンジオテンシンIIに変換する役割を担う、アンジオテンシン変換酵素(ACE)の強力な競合阻害剤です。アンジオテンシンIIは血圧の主要な調節因子であり、レニン・アンジオテンシン・アルドステロン系(RAAS)の重要な構成要素です。 カプトプリルは主に、本態性高血圧または腎血管性高血圧、うっ血性心不全、心筋梗塞後の左室機能不全、および腎症の治療に使用されます .
準備方法
合成経路と反応条件: カプトプリルはいくつかの方法で合成することができます。一般的な方法の1つは、L-プロリンと2-メチル-3-メルカプトプロピオニルクロリドをトリエチルアミンなどの塩基の存在下で反応させる方法です。 この反応は通常、ジクロロメタンなどの有機溶媒中で低温で行われ、副反応を防ぎます .
工業的製造方法: カプトプリルの工業的製造では、反応条件を厳密に制御して高収率と高純度を確保する、大型反応器の使用が一般的です。このプロセスには、最終生成物を精製するための再結晶化などの工程が含まれます。 連続フロー反応器などの高度な技術を使用すると、製造プロセスの効率とスケーラビリティを向上させることができます .
化学反応の分析
反応の種類: カプトプリルは、酸化、還元、置換など、さまざまな化学反応を起こします。 たとえば、カプトプリルのチオール基は酸化されてジスルフィドを形成することができます。一方、カルボキシル基はエステル化反応に関与することができます .
一般的な試薬と条件: カプトプリルを含む反応で使用される一般的な試薬には、酸化反応用の過酸化水素などの酸化剤と、還元反応用の水素化ホウ素ナトリウムなどの還元剤があります。 反応は通常、化合物の分解を防ぐために、穏やかな条件下で行われます .
生成される主な生成物: これらの反応から生成される主な生成物には、酸化によるカプトプリルジスルフィドと、エステル化反応によるさまざまなエステルが含まれます。 これらの誘導体は、異なる薬理学的特性を持つことがあり、その潜在的な治療用途について研究されています .
科学研究への応用
カプトプリルは、幅広い科学研究に利用されています。化学分野では、酵素阻害と反応機構を研究するためのモデル化合物として使用されます。生物学分野では、カプトプリルはさまざまな生理学的プロセスにおけるレニン・アンジオテンシン・アルドステロン系の役割を調査するために使用されています。 医学分野では、カプトプリルは高血圧、心不全、糖尿病性腎症の治療における治療効果について広く研究されています .
科学的研究の応用
Captopril has a wide range of scientific research applications. In chemistry, it is used as a model compound for studying enzyme inhibition and reaction mechanisms. In biology, this compound is used to investigate the role of the renin-angiotensin-aldosterone system in various physiological processes. In medicine, this compound is extensively studied for its therapeutic effects in treating hypertension, heart failure, and diabetic nephropathy .
作用機序
カプトプリルは、アンジオテンシン変換酵素を阻害することで効果を発揮し、アンジオテンシンIからアンジオテンシンIIへの変換を防ぎます。これにより、血管拡張、血圧低下、心臓への負担軽減が起こります。 アンジオテンシンII形成の阻害は、アルドステロンの放出も抑制し、ナトリウムと水の貯留減少につながります .
類似化合物との比較
カプトプリルは、エナラプリル、リシノプリル、ラミプリルなどの他のACE阻害剤と比較されることがよくあります。これらの化合物はすべて同じ酵素を阻害しますが、カプトプリルは、その高い効力と急速な作用開始に寄与するチオール基が特徴です。 このチオール基は、皮膚の発疹や味覚障害などの副作用を引き起こす可能性がありますが、これらの副作用は他のACE阻害剤ではそれほど一般的ではありません .
類似化合物のリスト:- エナラプリル
- リシノプリル
- ラミプリル
- ベナゼプリル
カプトプリルの独特の構造と急速な作用は、潜在的な副作用があるものの、貴重な治療薬となっています。 カプトプリルの発見と開発は、心臓血管疾患の治療を大きく進歩させました .
生物活性
Captopril, an angiotensin-converting enzyme (ACE) inhibitor, is primarily recognized for its role in managing hypertension and heart failure. However, its biological activities extend beyond these applications, showcasing a range of effects on cellular processes and immune responses. This article delves into the diverse biological activities of this compound, supported by data tables, case studies, and detailed research findings.
This compound functions by inhibiting the ACE, which is crucial in the renin-angiotensin-aldosterone system (RAAS). This inhibition leads to decreased levels of angiotensin II, resulting in vasodilation, reduced blood pressure, and decreased workload on the heart. Additionally, this compound has been shown to modulate immune responses and apoptosis in various cell types.
Inhibition of Apoptosis
Recent studies have highlighted this compound's ability to inhibit apoptosis in T cells. A significant study demonstrated that this compound blocked activation-induced apoptosis in murine T cell hybridomas by inhibiting interleukin-2 (IL-2) synthesis and caspase-3-like activity. The results indicated that this compound effectively reduced cell death upon T cell activation with anti-CD3 antibodies.
Study Findings
- Cell Proliferation : this compound inhibited T cell proliferation significantly when stimulated with anti-CD3 antibodies.
- DNA Fragmentation : Activation-induced DNA fragmentation was reduced by this compound in a dose-dependent manner.
- Fas Expression : this compound also modulated Fas and Fas ligand expression on T cells, contributing to its anti-apoptotic effects .
Impact on Systemic Inflammation
This compound's role extends into the modulation of systemic inflammation. A randomized controlled trial assessed the effects of this compound combined with telmisartan on inflammation markers in hemodialysis patients. The study found no statistically significant differences between treatment groups regarding demographic variables or treatment adherence, indicating a consistent response across participants .
Clinical Efficacy in Hypertension Management
This compound has been extensively studied for its efficacy in managing hypertension. A multicenter surveillance study involving 5,000 patients revealed that this compound effectively lowered blood pressure while maintaining a low incidence of adverse effects like rash and dysgeusia. The study reported a discontinuation rate due to side effects of only 5.5% over one year .
| Study Parameter | This compound Group | Placebo Group | Statistical Significance |
|---|---|---|---|
| Clinical Improvement | 80% | 27% | p < 0.001 |
| Ejection Fraction | 0.22 ± 0.02 | 0.18 ± 0.002 | p < 0.05 |
| Exercise Tolerance | 614 ± 27 seconds | 483 ± 43 seconds | p < 0.01 |
Effects on Heart Failure
In patients with refractory chronic congestive heart failure, this compound has shown significant clinical benefits. A placebo-controlled trial indicated that patients receiving this compound exhibited substantial improvements in functional class and exercise tolerance compared to those receiving placebo .
Key Findings
- Patient Cohort : The study included patients resistant to digitalis and diuretics.
- Efficacy : this compound resulted in a mean improvement in functional class value from 2.8 to 2.3 compared to minimal changes in the placebo group.
- Safety Profile : The treatment was well-tolerated with minimal adverse events reported.
Q & A
Basic Research Questions
Q. What structural and mechanistic principles guided the rational design of captopril as an ACE inhibitor?
this compound was developed using a receptor-based design approach targeting the zinc-binding active site of angiotensin-converting enzyme (ACE). Its sulfhydryl group mimics the carboxyl group of the ACE substrate, enabling competitive inhibition. This design was informed by studies on the venom peptide teprotide, which revealed ACE's catalytic mechanism . Methodologically, crystallographic data and structure-activity relationship (SAR) studies were critical for optimizing binding affinity and selectivity .
Q. How can Quality by Design (QbD) principles be applied to optimize this compound formulations for controlled release?
QbD methodologies involve systematic experimentation with variables like polymer composition (e.g., HPMC K15M and ethylcellulose) to achieve target dissolution profiles. For example, a central composite design (CCD) can model the impact of excipient ratios on drug release kinetics, ensuring robustness in sustained-release formulations. Dissolution testing remains the critical quality attribute (CQA) for validation .
Q. What validated analytical methods are recommended for quantifying this compound in biological matrices?
Reverse-phase high-performance liquid chromatography (RP-HPLC) with UV detection is widely used, offering retention times of ~3.1 minutes under optimized conditions (acetonitrile: 28–36% v/v, pH 2.8–3.6) . For higher sensitivity, LC-HRMS with a mass window of 1 ppm provides selective quantification in dried blood spots, minimizing matrix interference (e.g., m/z 218.0845 for this compound) .
Advanced Research Questions
Q. How do experimental variables (e.g., age, sex, dosage) influence the reproducibility of this compound's therapeutic outcomes in preclinical models?
Studies in renal injury models highlight confounding factors like mixed-sex cohorts and age ranges (3–12 months in rats), which increase data variability. To mitigate this, stratified randomization and covariance analysis (adjusting for age/sex) are recommended. For example, this compound’s efficacy in reducing HW/BW ratios in cardiomyopathic rats was significant only after controlling for these variables .
Q. What mechanisms explain this compound's antiangiogenic effects, and how can these be reconciled with its primary ACE inhibition role?
this compound inhibits endothelial cell migration and metalloproteinases (72-/92-kDa) via zinc chelation, independent of ACE. This biphasic dose-response (effective at <10 µM and millimolar ranges) suggests dual pathways: ACE-dependent vasodilation and ACE-independent antiangiogenesis. In vitro scratch assays and corneal neovascularization models are key for mechanistic validation .
Q. Why do contradictory results arise in studies investigating this compound's metabolic effects (e.g., insulin sensitivity, lipid profiles)?
In SHRSP-ZF rats, this compound failed to improve glucose, insulin, or triglyceride levels despite its antihypertensive action. This discrepancy may stem from tissue-specific ACE expression or compensatory pathways (e.g., RAAS upregulation). Dual inhibition strategies (e.g., combining this compound with PPAR-γ agonists) and tracer-based metabolic flux analysis are proposed to clarify these interactions .
Q. How can novel delivery systems (e.g., transdermal films, biosensors) enhance this compound's therapeutic applicability?
Niosomal-loaded transdermal films improve bioavailability by encapsulating this compound in non-ionic surfactants, achieving sustained release (>12 hours) with minimal dose requirements. For real-time monitoring, smartphone-integrated biosensors using N-CQDs-MnO₂ nanotubes enable dual-mode detection of this compound in plasma (LOD: 0.1 µM) .
Q. Data Contradiction Analysis
Q. How should researchers interpret conflicting data on this compound's lifespan-extending effects in murine models?
The ITP study reported increased median lifespan in mice, but male results were inconclusive due to experimental artifacts. Replication with higher doses (planned by ITP) and strain-specific genetic profiling (e.g., C57BL/6 vs. BALB/c) are advised. Notably, mice lack hypertension-related mortality, necessitating alternative models (e.g., senescence-accelerated mice) .
Q. What methodological refinements address variability in this compound's pharmacokinetic (PK) studies?
Interspecies differences in absorption (e.g., low Caco-2 permeability vs. in vivo bioavailability) require physiologically based pharmacokinetic (PBPK) modeling. Parameterization with plasma protein binding (103.2% recovery in LC-MS) and enterohepatic recirculation data improves predictive accuracy .
Q. Tables for Key Findings
特性
IUPAC Name |
(2S)-1-[(2S)-2-methyl-3-sulfanylpropanoyl]pyrrolidine-2-carboxylic acid | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C9H15NO3S/c1-6(5-14)8(11)10-4-2-3-7(10)9(12)13/h6-7,14H,2-5H2,1H3,(H,12,13)/t6-,7+/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
FAKRSMQSSFJEIM-RQJHMYQMSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(CS)C(=O)N1CCCC1C(=O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@H](CS)C(=O)N1CCC[C@H]1C(=O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C9H15NO3S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
1253948-36-5 | |
| Record name | L-Proline, 1-[(2S)-3-mercapto-2-methyl-1-oxopropyl]-, homopolymer | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1253948-36-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
DSSTOX Substance ID |
DTXSID1037197 | |
| Record name | Captopril | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1037197 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
217.29 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Captopril | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015328 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Freely soluble, Freely soluble in water (approximately 160 mg/mL), Freely soluble in alcohol, chloroform, methylene chloride; sparingly soluble in ethyl acetate, 4.52e+00 g/L | |
| Record name | Captopril | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01197 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Captopril | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6527 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Captopril | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015328 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
There are two isoforms of ACE: the somatic isoform, which exists as a glycoprotein comprised of a single polypeptide chain of 1277; and the testicular isoform, which has a lower molecular mass and is thought to play a role in sperm maturation and binding of sperm to the oviduct epithelium. Somatic ACE has two functionally active domains, N and C, which arise from tandem gene duplication. Although the two domains have high sequence similarity, they play distinct physiological roles. The C-domain is predominantly involved in blood pressure regulation while the N-domain plays a role in hematopoietic stem cell differentiation and proliferation. ACE inhibitors bind to and inhibit the activity of both domains, but have much greater affinity for and inhibitory activity against the C-domain. Captopril, one of the few ACE inhibitors that is not a prodrug, competes with ATI for binding to ACE and inhibits and enzymatic proteolysis of ATI to ATII. Decreasing ATII levels in the body decreases blood pressure by inhibiting the pressor effects of ATII as described in the Pharmacology section above. Captopril also causes an increase in plasma renin activity likely due to a loss of feedback inhibition mediated by ATII on the release of renin and/or stimulation of reflex mechanisms via baroreceptors. Captopril’s affinity for ACE is approximately 30,000 times greater than that of ATI., The local role of the renin angiotensin system (RAS) was documented recently beside its conventional systemic functions. Studies showed that the effector angiotensin II (AngII) alters bone health, while inhibition of the angiotensin converting enzyme (ACE-1) preserved these effects. The newly identified Ang1-7 exerts numerous beneficial effects opposing the AngII. Thus, the current study examines the role of Ang1-7 in mediating the osteo-preservative effects of ACEI (captopril) through the G-protein coupled Mas receptor using an ovariectomized (OVX) rat model of osteoporosis. 8 weeks after the surgical procedures, captopril was administered orally (40 mg/kg/day), while the specific Mas receptor blocker (A-779) was delivered at infusion rate of 400 ng/kg/1 min for 6 weeks. Bone metabolic markers were measured in serum and urine. Minerals concentrations were quantified in serum, urine and femoral bones by inductive coupled plasma mass spectroscopy (ICP-MS). Trabecular and cortical morphometry was analyzed in the right distal femurs using micro-CT. Finally, the expressions of RAS peptides, enzymes and receptors along with the receptor activator of NF-kappaB ligand (RANKL) and osteoprotegerin (OPG) were determined femurs heads. OVX animals markedly showed altered bone metabolism and mineralization along with disturbed bone micro-structure. Captopril significantly restored the metabolic bone bio-markers and corrected Ca2+ and P values in urine and bones of estrogen deficient rats. Moreover, the trabecular and cortical morphometric features were repaired by captopril in OVX groups. Captopril also improved the expressions of ACE-2, Ang1-7, Mas and OPG, while abolished OVX-induced up-regulation of ACE-1, AngII, Ang type 1 receptor (AT1R) and RANKL. Inhibition of Ang1-7 cascade by A-779 significantly eradicated captopril protective effects on bone metabolism, mineralization and micro-structure. A-779 also restored OVX effects on RANKL expression and ACE-1/AngII/AT1R cascade and down-regulated OPG expression and ACE-2/Ang1-7/Mas pathway. In line with the clinical observations of the bone-preservative properties following ACE-1 inhibition, local activation of ACE-2/Ang1-7/Mas signaling and suppressed osteoclastogenesis seem responsible for the osteo-preservative effect of captopril, which could offers a potential therapeutic value in treatment of disabling bone and skeletal muscular diseases. | |
| Record name | Captopril | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01197 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Captopril | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6527 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White to off-white, crystalline powder, Crystals from ethyl acetate/hexane | |
CAS No. |
62571-86-2 | |
| Record name | Captopril | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=62571-86-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Captopril [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0062571862 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Captopril | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01197 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | captopril | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757419 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Captopril | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1037197 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Captopril | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.057.806 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CAPTOPRIL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/9G64RSX1XD | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Captopril | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6527 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Captopril | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015328 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
103-104, 103-104 °C, 106 °C | |
| Record name | Captopril | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01197 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Captopril | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6527 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Captopril | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015328 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。













