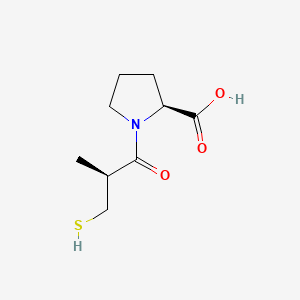
Captopril
概述
描述
卡托普利是一种强效的血管紧张素转换酶 (ACE) 竞争性抑制剂,它负责将血管紧张素 I 转换为血管紧张素 II。血管紧张素 II 是血压的关键调节剂,也是肾素-血管紧张素-醛固酮系统 (RAAS) 的重要组成部分。 卡托普利主要用于治疗原发性或肾血管性高血压、充血性心力衰竭、心肌梗死后的左心室功能障碍和肾病 .
准备方法
合成路线和反应条件: 卡托普利可以通过多种方法合成。一种常用的方法是,在三乙胺等碱存在的情况下,使 L-脯氨酸与 2-甲基-3-巯基丙酰氯反应。 反应通常在二氯甲烷等有机溶剂中于低温下进行,以防止副反应 .
工业生产方法: 卡托普利的工业生产通常涉及使用大型反应器,在其中严格控制反应条件,以确保高产率和纯度。该过程包括重结晶等步骤,以纯化最终产物。 使用连续流动反应器等先进技术可以提高生产过程的效率和可扩展性 .
化学反应分析
反应类型: 卡托普利会发生各种化学反应,包括氧化、还原和取代反应。 例如,卡托普利中的硫醇基可以被氧化生成二硫化物,而羧基可以参与酯化反应 .
常用试剂和条件: 涉及卡托普利的反应中常用的试剂包括过氧化氢等氧化剂(用于氧化反应)和硼氢化钠等还原剂(用于还原反应)。 反应通常在温和条件下进行,以防止化合物降解 .
主要生成产物: 这些反应产生的主要产物包括来自氧化的卡托普利二硫化物和来自酯化反应的各种酯。 这些衍生物可能具有不同的药理特性,并且经常被研究以寻找其潜在的治疗应用 .
科学研究应用
卡托普利具有广泛的科学研究应用。在化学领域,它被用作研究酶抑制和反应机理的模型化合物。在生物学领域,卡托普利被用于研究肾素-血管紧张素-醛固酮系统在各种生理过程中的作用。 在医学领域,卡托普利被广泛研究,用于治疗高血压、心力衰竭和糖尿病肾病的治疗效果 .
作用机制
卡托普利通过抑制血管紧张素转换酶发挥作用,从而阻止血管紧张素 I 转换为血管紧张素 II。这导致血管扩张、血压降低和心脏负荷减轻。 抑制血管紧张素 II 的形成还会减少醛固酮的释放,从而减少钠和水的潴留 .
相似化合物的比较
卡托普利经常与其他 ACE 抑制剂(如依那普利、赖诺普利和雷米普利)进行比较。虽然所有这些化合物都抑制相同的酶,但卡托普利由于其硫醇基而独一无二,该基团使其具有高效力和快速起效。 该硫醇基也可能引起皮肤疹和味觉障碍等副作用,这些副作用在其他 ACE 抑制剂中不太常见 .
类似化合物列表:- 依那普利
- 赖诺普利
- 雷米普利
- 贝那普利
尽管卡托普利可能存在副作用,但其独特的结构和快速作用使其成为一种有价值的治疗剂。 它的发现和发展极大地推动了心血管疾病的治疗 .
生物活性
Captopril, an angiotensin-converting enzyme (ACE) inhibitor, is primarily recognized for its role in managing hypertension and heart failure. However, its biological activities extend beyond these applications, showcasing a range of effects on cellular processes and immune responses. This article delves into the diverse biological activities of this compound, supported by data tables, case studies, and detailed research findings.
This compound functions by inhibiting the ACE, which is crucial in the renin-angiotensin-aldosterone system (RAAS). This inhibition leads to decreased levels of angiotensin II, resulting in vasodilation, reduced blood pressure, and decreased workload on the heart. Additionally, this compound has been shown to modulate immune responses and apoptosis in various cell types.
Inhibition of Apoptosis
Recent studies have highlighted this compound's ability to inhibit apoptosis in T cells. A significant study demonstrated that this compound blocked activation-induced apoptosis in murine T cell hybridomas by inhibiting interleukin-2 (IL-2) synthesis and caspase-3-like activity. The results indicated that this compound effectively reduced cell death upon T cell activation with anti-CD3 antibodies.
Study Findings
- Cell Proliferation : this compound inhibited T cell proliferation significantly when stimulated with anti-CD3 antibodies.
- DNA Fragmentation : Activation-induced DNA fragmentation was reduced by this compound in a dose-dependent manner.
- Fas Expression : this compound also modulated Fas and Fas ligand expression on T cells, contributing to its anti-apoptotic effects .
Impact on Systemic Inflammation
This compound's role extends into the modulation of systemic inflammation. A randomized controlled trial assessed the effects of this compound combined with telmisartan on inflammation markers in hemodialysis patients. The study found no statistically significant differences between treatment groups regarding demographic variables or treatment adherence, indicating a consistent response across participants .
Clinical Efficacy in Hypertension Management
This compound has been extensively studied for its efficacy in managing hypertension. A multicenter surveillance study involving 5,000 patients revealed that this compound effectively lowered blood pressure while maintaining a low incidence of adverse effects like rash and dysgeusia. The study reported a discontinuation rate due to side effects of only 5.5% over one year .
| Study Parameter | This compound Group | Placebo Group | Statistical Significance |
|---|---|---|---|
| Clinical Improvement | 80% | 27% | p < 0.001 |
| Ejection Fraction | 0.22 ± 0.02 | 0.18 ± 0.002 | p < 0.05 |
| Exercise Tolerance | 614 ± 27 seconds | 483 ± 43 seconds | p < 0.01 |
Effects on Heart Failure
In patients with refractory chronic congestive heart failure, this compound has shown significant clinical benefits. A placebo-controlled trial indicated that patients receiving this compound exhibited substantial improvements in functional class and exercise tolerance compared to those receiving placebo .
Key Findings
- Patient Cohort : The study included patients resistant to digitalis and diuretics.
- Efficacy : this compound resulted in a mean improvement in functional class value from 2.8 to 2.3 compared to minimal changes in the placebo group.
- Safety Profile : The treatment was well-tolerated with minimal adverse events reported.
常见问题
Basic Research Questions
Q. What structural and mechanistic principles guided the rational design of captopril as an ACE inhibitor?
this compound was developed using a receptor-based design approach targeting the zinc-binding active site of angiotensin-converting enzyme (ACE). Its sulfhydryl group mimics the carboxyl group of the ACE substrate, enabling competitive inhibition. This design was informed by studies on the venom peptide teprotide, which revealed ACE's catalytic mechanism . Methodologically, crystallographic data and structure-activity relationship (SAR) studies were critical for optimizing binding affinity and selectivity .
Q. How can Quality by Design (QbD) principles be applied to optimize this compound formulations for controlled release?
QbD methodologies involve systematic experimentation with variables like polymer composition (e.g., HPMC K15M and ethylcellulose) to achieve target dissolution profiles. For example, a central composite design (CCD) can model the impact of excipient ratios on drug release kinetics, ensuring robustness in sustained-release formulations. Dissolution testing remains the critical quality attribute (CQA) for validation .
Q. What validated analytical methods are recommended for quantifying this compound in biological matrices?
Reverse-phase high-performance liquid chromatography (RP-HPLC) with UV detection is widely used, offering retention times of ~3.1 minutes under optimized conditions (acetonitrile: 28–36% v/v, pH 2.8–3.6) . For higher sensitivity, LC-HRMS with a mass window of 1 ppm provides selective quantification in dried blood spots, minimizing matrix interference (e.g., m/z 218.0845 for this compound) .
Advanced Research Questions
Q. How do experimental variables (e.g., age, sex, dosage) influence the reproducibility of this compound's therapeutic outcomes in preclinical models?
Studies in renal injury models highlight confounding factors like mixed-sex cohorts and age ranges (3–12 months in rats), which increase data variability. To mitigate this, stratified randomization and covariance analysis (adjusting for age/sex) are recommended. For example, this compound’s efficacy in reducing HW/BW ratios in cardiomyopathic rats was significant only after controlling for these variables .
Q. What mechanisms explain this compound's antiangiogenic effects, and how can these be reconciled with its primary ACE inhibition role?
this compound inhibits endothelial cell migration and metalloproteinases (72-/92-kDa) via zinc chelation, independent of ACE. This biphasic dose-response (effective at <10 µM and millimolar ranges) suggests dual pathways: ACE-dependent vasodilation and ACE-independent antiangiogenesis. In vitro scratch assays and corneal neovascularization models are key for mechanistic validation .
Q. Why do contradictory results arise in studies investigating this compound's metabolic effects (e.g., insulin sensitivity, lipid profiles)?
In SHRSP-ZF rats, this compound failed to improve glucose, insulin, or triglyceride levels despite its antihypertensive action. This discrepancy may stem from tissue-specific ACE expression or compensatory pathways (e.g., RAAS upregulation). Dual inhibition strategies (e.g., combining this compound with PPAR-γ agonists) and tracer-based metabolic flux analysis are proposed to clarify these interactions .
Q. How can novel delivery systems (e.g., transdermal films, biosensors) enhance this compound's therapeutic applicability?
Niosomal-loaded transdermal films improve bioavailability by encapsulating this compound in non-ionic surfactants, achieving sustained release (>12 hours) with minimal dose requirements. For real-time monitoring, smartphone-integrated biosensors using N-CQDs-MnO₂ nanotubes enable dual-mode detection of this compound in plasma (LOD: 0.1 µM) .
Q. Data Contradiction Analysis
Q. How should researchers interpret conflicting data on this compound's lifespan-extending effects in murine models?
The ITP study reported increased median lifespan in mice, but male results were inconclusive due to experimental artifacts. Replication with higher doses (planned by ITP) and strain-specific genetic profiling (e.g., C57BL/6 vs. BALB/c) are advised. Notably, mice lack hypertension-related mortality, necessitating alternative models (e.g., senescence-accelerated mice) .
Q. What methodological refinements address variability in this compound's pharmacokinetic (PK) studies?
Interspecies differences in absorption (e.g., low Caco-2 permeability vs. in vivo bioavailability) require physiologically based pharmacokinetic (PBPK) modeling. Parameterization with plasma protein binding (103.2% recovery in LC-MS) and enterohepatic recirculation data improves predictive accuracy .
Q. Tables for Key Findings
属性
IUPAC Name |
(2S)-1-[(2S)-2-methyl-3-sulfanylpropanoyl]pyrrolidine-2-carboxylic acid | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C9H15NO3S/c1-6(5-14)8(11)10-4-2-3-7(10)9(12)13/h6-7,14H,2-5H2,1H3,(H,12,13)/t6-,7+/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
FAKRSMQSSFJEIM-RQJHMYQMSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(CS)C(=O)N1CCCC1C(=O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@H](CS)C(=O)N1CCC[C@H]1C(=O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C9H15NO3S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
1253948-36-5 | |
| Record name | L-Proline, 1-[(2S)-3-mercapto-2-methyl-1-oxopropyl]-, homopolymer | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1253948-36-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
DSSTOX Substance ID |
DTXSID1037197 | |
| Record name | Captopril | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1037197 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
217.29 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Captopril | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015328 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Freely soluble, Freely soluble in water (approximately 160 mg/mL), Freely soluble in alcohol, chloroform, methylene chloride; sparingly soluble in ethyl acetate, 4.52e+00 g/L | |
| Record name | Captopril | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01197 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Captopril | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6527 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Captopril | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015328 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
There are two isoforms of ACE: the somatic isoform, which exists as a glycoprotein comprised of a single polypeptide chain of 1277; and the testicular isoform, which has a lower molecular mass and is thought to play a role in sperm maturation and binding of sperm to the oviduct epithelium. Somatic ACE has two functionally active domains, N and C, which arise from tandem gene duplication. Although the two domains have high sequence similarity, they play distinct physiological roles. The C-domain is predominantly involved in blood pressure regulation while the N-domain plays a role in hematopoietic stem cell differentiation and proliferation. ACE inhibitors bind to and inhibit the activity of both domains, but have much greater affinity for and inhibitory activity against the C-domain. Captopril, one of the few ACE inhibitors that is not a prodrug, competes with ATI for binding to ACE and inhibits and enzymatic proteolysis of ATI to ATII. Decreasing ATII levels in the body decreases blood pressure by inhibiting the pressor effects of ATII as described in the Pharmacology section above. Captopril also causes an increase in plasma renin activity likely due to a loss of feedback inhibition mediated by ATII on the release of renin and/or stimulation of reflex mechanisms via baroreceptors. Captopril’s affinity for ACE is approximately 30,000 times greater than that of ATI., The local role of the renin angiotensin system (RAS) was documented recently beside its conventional systemic functions. Studies showed that the effector angiotensin II (AngII) alters bone health, while inhibition of the angiotensin converting enzyme (ACE-1) preserved these effects. The newly identified Ang1-7 exerts numerous beneficial effects opposing the AngII. Thus, the current study examines the role of Ang1-7 in mediating the osteo-preservative effects of ACEI (captopril) through the G-protein coupled Mas receptor using an ovariectomized (OVX) rat model of osteoporosis. 8 weeks after the surgical procedures, captopril was administered orally (40 mg/kg/day), while the specific Mas receptor blocker (A-779) was delivered at infusion rate of 400 ng/kg/1 min for 6 weeks. Bone metabolic markers were measured in serum and urine. Minerals concentrations were quantified in serum, urine and femoral bones by inductive coupled plasma mass spectroscopy (ICP-MS). Trabecular and cortical morphometry was analyzed in the right distal femurs using micro-CT. Finally, the expressions of RAS peptides, enzymes and receptors along with the receptor activator of NF-kappaB ligand (RANKL) and osteoprotegerin (OPG) were determined femurs heads. OVX animals markedly showed altered bone metabolism and mineralization along with disturbed bone micro-structure. Captopril significantly restored the metabolic bone bio-markers and corrected Ca2+ and P values in urine and bones of estrogen deficient rats. Moreover, the trabecular and cortical morphometric features were repaired by captopril in OVX groups. Captopril also improved the expressions of ACE-2, Ang1-7, Mas and OPG, while abolished OVX-induced up-regulation of ACE-1, AngII, Ang type 1 receptor (AT1R) and RANKL. Inhibition of Ang1-7 cascade by A-779 significantly eradicated captopril protective effects on bone metabolism, mineralization and micro-structure. A-779 also restored OVX effects on RANKL expression and ACE-1/AngII/AT1R cascade and down-regulated OPG expression and ACE-2/Ang1-7/Mas pathway. In line with the clinical observations of the bone-preservative properties following ACE-1 inhibition, local activation of ACE-2/Ang1-7/Mas signaling and suppressed osteoclastogenesis seem responsible for the osteo-preservative effect of captopril, which could offers a potential therapeutic value in treatment of disabling bone and skeletal muscular diseases. | |
| Record name | Captopril | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01197 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Captopril | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6527 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White to off-white, crystalline powder, Crystals from ethyl acetate/hexane | |
CAS No. |
62571-86-2 | |
| Record name | Captopril | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=62571-86-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Captopril [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0062571862 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Captopril | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01197 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | captopril | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757419 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Captopril | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1037197 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Captopril | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.057.806 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CAPTOPRIL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/9G64RSX1XD | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Captopril | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6527 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Captopril | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015328 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
103-104, 103-104 °C, 106 °C | |
| Record name | Captopril | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01197 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Captopril | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6527 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Captopril | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015328 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
体外研究产品的免责声明和信息
请注意,BenchChem 上展示的所有文章和产品信息仅供信息参考。 BenchChem 上可购买的产品专为体外研究设计,这些研究在生物体外进行。体外研究,源自拉丁语 "in glass",涉及在受控实验室环境中使用细胞或组织进行的实验。重要的是要注意,这些产品没有被归类为药物或药品,他们没有得到 FDA 的批准,用于预防、治疗或治愈任何医疗状况、疾病或疾病。我们必须强调,将这些产品以任何形式引入人类或动物的身体都是法律严格禁止的。遵守这些指南对确保研究和实验的法律和道德标准的符合性至关重要。













