Clindamycin
概要
説明
クリンダマイシンは、さまざまな細菌感染症の治療に使用されるリンコサミド系抗生物質です。特に、嫌気性菌や特定のグラム陽性菌(ブドウ球菌や連鎖球菌など)に対して有効です。 クリンダマイシンは、骨髄炎、骨盤内炎症性疾患、咽頭炎、肺炎、皮膚感染症などの感染症の治療に一般的に使用されます 。 経口カプセル、外用クリーム、静脈注射液など、さまざまな形態で入手できます .
2. 製法
クリンダマイシンは、天然に存在する抗生物質であるリンコマイシンから合成されます。 合成には、リンコマイシンの7位のヒドロキシル基を塩素原子に置換する塩素化が伴います 。このプロセスには、いくつかの段階が含まれます。
シリコン保護基の付加: リンコマイシンは、最初にシリコン基を使用して保護されます。
選択的脱保護: 保護されたリンコマイシンは、選択的脱保護を受けます。
ミツノブ反応: 脱保護されたリンコマイシンは、ミツノブ反応を受けます。
加水分解反応: その後、生成物は加水分解されて7-エピメリンコマイシンが得られます。
クリンダマイシン塩酸塩の工業生産には、塩素化、加水分解、抽出、濃縮の段階が含まれ、遊離アルカリ形態が得られます。その後、塩形成と脱アルコール化を行い、クリンダマイシン塩酸塩が得られます .
3. 化学反応の分析
クリンダマイシンは、さまざまな化学反応を起こし、それらには以下が含まれます。
酸化: クリンダマイシンは、酸化されてスルホキシドとN-脱メチル化代謝物を形成することができます。
還元: 還元反応はそれほど一般的ではありませんが、特定の条件下で起こる可能性があります。
これらの反応に使用される一般的な試薬には、塩素化剤、酸化剤、還元剤が含まれます。 これらの反応から生成される主な生成物は、クリンダマイシン塩酸塩とその代謝物です .
4. 科学研究への応用
クリンダマイシンは、幅広い科学研究に利用されています。
化学: 抗生物質の合成と改変を研究するためのモデル化合物として使用されます。
生物学: クリンダマイシンは、細菌のタンパク質合成と耐性メカニズムの研究に使用されます。
準備方法
Clindamycin is synthesized from lincomycin, a naturally occurring antibiotic. The synthesis involves the chlorination of lincomycin to replace the hydroxyl group at position 7 with a chlorine atom . The process includes several steps:
Silicon Protecting Group Application: Lincomycin is first protected using a silicon group.
Selective Deprotection: The protected lincomycin undergoes selective deprotection.
Mitsunobu Substitution Reaction: The deprotected lincomycin is subjected to a Mitsunobu substitution reaction.
Hydrolysis Reaction: The product is then hydrolyzed to obtain 7-epime lincomycin.
Chlorination Reaction: Finally, the 7-epime lincomycin is chlorinated to produce this compound.
Industrial production of this compound hydrochloride involves chlorination, hydrolysis, extraction, and concentration steps to obtain the free alkali form, followed by salt formation and dealcoholation to yield this compound hydrochloride .
化学反応の分析
Clindamycin undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized to form sulfoxide and N-demethylated metabolites.
Reduction: Reduction reactions are less common but can occur under specific conditions.
Substitution: The Mitsunobu substitution reaction is a key step in its synthesis.
Common reagents used in these reactions include chlorinating agents, oxidizing agents, and reducing agents. The major products formed from these reactions are this compound hydrochloride and its metabolites .
科学的研究の応用
Clindamycin has a wide range of scientific research applications:
Chemistry: It is used as a model compound in studying antibiotic synthesis and modification.
Biology: this compound is used to study bacterial protein synthesis and resistance mechanisms.
Medicine: It is widely used to treat bacterial infections, including those caused by methicillin-resistant Staphylococcus aureus (MRSA) and anaerobic bacteria
作用機序
クリンダマイシンは、細菌の50Sリボソームサブユニットに結合することにより、タンパク質合成を阻害して作用します。 この作用は、翻訳中のペプチド鎖の伸長を阻害し、細菌の増殖を効果的に停止します 。 クリンダマイシンは細菌のリボソームを標的とし、トランスペプチダーゼ反応を阻害し、初期の鎖伸長を阻害します .
類似化合物との比較
クリンダマイシンは、多くの場合、以下のような他の抗生物質と比較されます。
リンコマイシン: クリンダマイシンは、リンコマイシンの塩素化誘導体であり、より優れた特性を持っています。
アモキシシリン: 同様の感染症に用いられるペニシリン系抗生物質ですが、作用機序が異なります。
ドキシサイクリン: より幅広い活性を持つテトラサイクリン系抗生物質です
クリンダマイシンは、嫌気性菌に対する高い有効性と骨や膿瘍への浸透能力が高く、骨髄炎やその他の深在性感染症の治療に特に有効である点が特徴です .
生物活性
Clindamycin is an antibiotic belonging to the lincosamide class, primarily effective against anaerobic bacteria and certain protozoa. It is widely used in clinical settings for treating various infections, including skin and soft tissue infections, respiratory tract infections, and certain types of bone infections. This article delves into the biological activity of this compound, supported by data tables, case studies, and relevant research findings.
This compound exerts its antibacterial effects by inhibiting bacterial protein synthesis. It binds to the 50S ribosomal subunit of susceptible bacteria, thereby blocking peptide bond formation during translation. This mechanism is similar to that of macrolides but differs in its binding site preference, making this compound effective against a range of Gram-positive cocci and anaerobic bacteria.
Key Points:
- Target : 50S ribosomal subunit.
- Effect : Inhibition of protein synthesis.
- Spectrum : Effective against anaerobes and some protozoa.
Clinical Applications
This compound is indicated for various infections, particularly those caused by anaerobic bacteria. Its effectiveness is highlighted in several case studies:
Case Study Highlights:
- Skin and Soft Tissue Infections : this compound has shown significant efficacy in treating cellulitis and abscesses caused by Staphylococcus aureus, including methicillin-resistant strains (MRSA).
- Bone Infections : In osteomyelitis cases, this compound demonstrated favorable outcomes when combined with surgical intervention.
- Periodontal Disease : A study indicated that this compound could improve glycemic control in diabetic patients with periodontal disease, showing a mean reduction in HbA1c levels .
Efficacy Against Specific Pathogens
This compound's activity against various pathogens can be summarized in the following table:
Resistance Patterns
Resistance to this compound can occur through various mechanisms, including:
- Methylation of adenine residues in the 23S rRNA, which alters the binding site.
- Efflux pumps that expel the antibiotic from bacterial cells.
Monitoring resistance patterns is crucial, especially in hospital settings where resistant strains may emerge.
Adverse Effects and Considerations
While this compound is generally well-tolerated, it can lead to side effects such as gastrointestinal disturbances and a risk of C. difficile-associated diarrhea. The incidence of C. difficile infection has been noted to increase with this compound use, necessitating careful patient monitoring .
Important Considerations:
- Caution in prescribing for patients with a history of gastrointestinal disorders.
- Monitoring for signs of C. difficile infection during treatment.
Q & A
Basic Research Questions
Q. How can researchers design bioequivalence trials for generic clindamycin formulations, and what statistical criteria ensure validity?
- Methodological Answer : Bioequivalence studies should follow CHMP guidelines, using a randomized, crossover design with 90% confidence intervals for AUC0-inf and Cmax within 0.8–1.24. Linear pharmacokinetics (150–600 mg dose range) justify dose selection (e.g., 300 mg). Analytical methods (e.g., HPLC) and adherence to EMEA/CHMP/EWP/40326/2006 ensure reproducibility .
Q. What experimental methods are recommended to detect inducible this compound resistance in Staphylococcus aureus?
- Methodological Answer : Use the D-zone test: place erythromycin (15 µg) and this compound (2 µg) discs 15 mm apart on Mueller-Hinton agar. Flattening of the this compound inhibition zone near erythromycin indicates inducible resistance. Confirm with CLSI M100 standards and statistical tools (e.g., SPSS) for data analysis .
Q. How should systematic reviews assess this compound’s efficacy in preventing post-surgical infections?
- Methodological Answer : Employ PRISMA guidelines, extract data from RCTs using tools like the Cochrane Risk of Bias Tool (e.g., randomization, blinding). Pool data via meta-analysis (fixed/random effects models) and address heterogeneity with sensitivity analysis. Focus on outcomes like infection rates in third molar extractions .
Q. What are key considerations for designing in vitro susceptibility testing of this compound against drug-resistant Staphylococci?
- Methodological Answer : Use microdilution methods to determine MICs, adhering to CLSI-M100 standards. Include positive controls (e.g., ciprofloxacin) and analyze data with Fisher’s exact test to compare sensitivity across strains. Account for regional resistance patterns in study design .
Advanced Research Questions
Q. How can physiologically based pharmacokinetic (PBPK) models optimize this compound dosing in pediatric populations?
- Methodological Answer : Develop PBPK models using adult PK data (extracted via Plot Digitizer®) and scale parameters (e.g., organ weights, enzyme expression) using ontogeny functions. Validate with opportunistic pediatric data and software like Simcyp®. Address variability in CYP3A4 maturation .
Q. What molecular dynamics (MD) approaches elucidate this compound resistance mechanisms in bacterial ribosomes?
- Methodological Answer : Simulate this compound binding to wild-type (WT) and mutant (A2058G) 23S rRNA ribosome fragments in explicit solvent. Analyze conformational flexibility (RMSD, RMSF) and stacking interactions (e.g., G2505-U2506) using GROMACS/AMBER. Correlate findings with in vitro resistance data .
Q. How can factorial design optimize this compound-loaded nanogel formulations for enhanced delivery?
- Methodological Answer : Apply 3<sup>2</sup> full factorial design to evaluate independent variables (e.g., polymer concentration, cross-linker ratio). Use DOE software to analyze responses (entrapment efficiency, release kinetics). Validate with in vitro characterization (e.g., TEM, DSC) .
Q. What pharmacovigilance strategies address this compound-associated C. difficile colitis in clinical trials?
- Methodological Answer : Monitor diarrhea incidence (>20% systemic cases) and confirm pseudomembranous colitis via toxin PCR. Exclude high-risk patients (e.g., prior C. difficile history) and analyze covariates (e.g., age, concomitant antibiotics) using logistic regression .
Q. How do cross-resistance patterns between this compound and macrolides inform combination therapy design?
- Methodological Answer : Test for erm/mef resistance genes via PCR in isolates with MLSB phenotypes. Use checkerboard assays to quantify synergism (FIC index ≤0.5) with erythromycin. Model PK/PD interactions (e.g., AUC/MIC) to optimize dosing .
Q. What statistical methods resolve contradictions in this compound’s efficacy for bacterial vaginosis trials?
特性
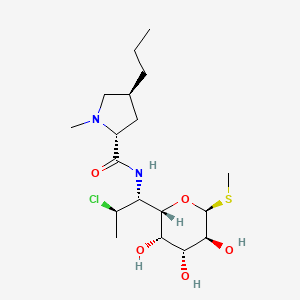
IUPAC Name |
(2S,4R)-N-[(1S,2S)-2-chloro-1-[(2R,3R,4S,5R,6R)-3,4,5-trihydroxy-6-methylsulfanyloxan-2-yl]propyl]-1-methyl-4-propylpyrrolidine-2-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C18H33ClN2O5S/c1-5-6-10-7-11(21(3)8-10)17(25)20-12(9(2)19)16-14(23)13(22)15(24)18(26-16)27-4/h9-16,18,22-24H,5-8H2,1-4H3,(H,20,25)/t9-,10+,11-,12+,13-,14+,15+,16+,18+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
KDLRVYVGXIQJDK-AWPVFWJPSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCCC1CC(N(C1)C)C(=O)NC(C2C(C(C(C(O2)SC)O)O)O)C(C)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCC[C@@H]1C[C@H](N(C1)C)C(=O)N[C@@H]([C@@H]2[C@@H]([C@@H]([C@H]([C@H](O2)SC)O)O)O)[C@H](C)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C18H33ClN2O5S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
21462-39-5 (mono-hydrochloride), 58207-19-5 (mono-HCl, mono-hydrate) | |
| Record name | Clindamycin [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0018323449 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID2022836 | |
| Record name | Clindamycin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2022836 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
425.0 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
Clindamycin may be bacteriostatic or bactericidal in action, depending on the concentration of the drug attained at the site of infection and the susceptibility of the infecting organism. Clindamycin palmitate hydrochloride and clindamycin phosphate are inactive until hydrolyzed to free clindamycin. This hydrolysis occurs rapidly in vivo. Clindamycin appears to inhibit protein synthesis in susceptible organisms by binding to 50S ribosomal subunits; the primary effect is inhibition of peptide bond formation. The site of action appears to be the same as that of erythromycin, chloramphenicol, and lincomycin., Clindamycin binds exclusively to the 50S subunit of bacterial ribosomes and suppresses protein synthesis., ... Clindamycin is not a substrate for macrolide efflux pumps, and strains that are resistant to macrolides by this mechanism are susceptible to clindamycin. | |
| Record name | CLINDAMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3037 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Yellow, amorphous solid | |
CAS No. |
18323-44-9 | |
| Record name | Clindamycin | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=18323-44-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Clindamycin [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0018323449 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Clindamycin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2022836 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Clindamycin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.038.357 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CLINDAMYCIN | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/3U02EL437C | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | CLINDAMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3037 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。















