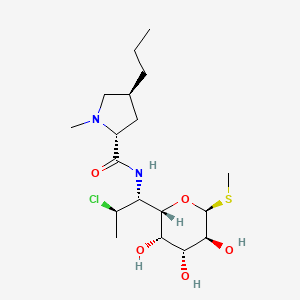
Clindamycin
描述
克林霉素是一种林可霉素类抗生素,用于治疗多种细菌感染。它对厌氧菌和某些革兰氏阳性菌,包括葡萄球菌和链球菌,特别有效。 克林霉素通常用于治疗骨髓炎、盆腔炎、链球菌性咽炎、肺炎和皮肤感染等感染 . 它有多种剂型,包括口服胶囊、外用乳膏和静脉注射液 .
准备方法
克林霉素是由林可霉素合成而来,林可霉素是一种天然抗生素。 合成过程包括对林可霉素进行氯化,用氯原子取代7位羟基 . 过程包括以下几个步骤:
硅保护基团应用: 林可霉素首先使用硅基团进行保护。
选择性脱保护: 受保护的林可霉素进行选择性脱保护。
米氏反应: 脱保护的林可霉素进行米氏反应。
水解反应: 然后将产物水解得到7-差向异构体林可霉素。
氯化反应: 最后,将7-差向异构体林可霉素进行氯化,得到克林霉素.
化学反应分析
克林霉素会发生各种化学反应,包括:
氧化: 克林霉素可以被氧化形成亚砜和N-脱甲基代谢物。
还原: 还原反应不太常见,但可能在特定条件下发生。
取代: 米氏取代反应是其合成过程中的关键步骤.
这些反应中常用的试剂包括氯化剂、氧化剂和还原剂。 这些反应的主要产物是克林霉素盐酸盐及其代谢物 .
科学研究应用
克林霉素具有广泛的科学研究应用:
化学: 它被用作研究抗生素合成和修饰的模型化合物。
生物学: 克林霉素被用于研究细菌蛋白质合成和抗药性机制。
医学: 它被广泛用于治疗细菌感染,包括由耐甲氧西林金黄色葡萄球菌 (MRSA) 和厌氧菌引起的感染
工业: 克林霉素用于制药行业生产各种抗生素制剂.
作用机制
克林霉素通过与细菌的50S核糖体亚基结合,抑制蛋白质合成来发挥作用。 这种作用阻止了翻译过程中肽链的延伸,有效地阻止了细菌生长 . 克林霉素靶向细菌核糖体,破坏转肽反应,抑制早期链延伸 .
相似化合物的比较
克林霉素通常与其他抗生素进行比较,例如:
林可霉素: 克林霉素是林可霉素的氯化衍生物,具有改进的特性。
阿莫西林: 一种青霉素类抗生素,用于治疗类似的感染,但作用机制不同。
多西环素: 一种四环素类抗生素,具有更广谱的活性
生物活性
Clindamycin is an antibiotic belonging to the lincosamide class, primarily effective against anaerobic bacteria and certain protozoa. It is widely used in clinical settings for treating various infections, including skin and soft tissue infections, respiratory tract infections, and certain types of bone infections. This article delves into the biological activity of this compound, supported by data tables, case studies, and relevant research findings.
This compound exerts its antibacterial effects by inhibiting bacterial protein synthesis. It binds to the 50S ribosomal subunit of susceptible bacteria, thereby blocking peptide bond formation during translation. This mechanism is similar to that of macrolides but differs in its binding site preference, making this compound effective against a range of Gram-positive cocci and anaerobic bacteria.
Key Points:
- Target : 50S ribosomal subunit.
- Effect : Inhibition of protein synthesis.
- Spectrum : Effective against anaerobes and some protozoa.
Clinical Applications
This compound is indicated for various infections, particularly those caused by anaerobic bacteria. Its effectiveness is highlighted in several case studies:
Case Study Highlights:
- Skin and Soft Tissue Infections : this compound has shown significant efficacy in treating cellulitis and abscesses caused by Staphylococcus aureus, including methicillin-resistant strains (MRSA).
- Bone Infections : In osteomyelitis cases, this compound demonstrated favorable outcomes when combined with surgical intervention.
- Periodontal Disease : A study indicated that this compound could improve glycemic control in diabetic patients with periodontal disease, showing a mean reduction in HbA1c levels .
Efficacy Against Specific Pathogens
This compound's activity against various pathogens can be summarized in the following table:
Resistance Patterns
Resistance to this compound can occur through various mechanisms, including:
- Methylation of adenine residues in the 23S rRNA, which alters the binding site.
- Efflux pumps that expel the antibiotic from bacterial cells.
Monitoring resistance patterns is crucial, especially in hospital settings where resistant strains may emerge.
Adverse Effects and Considerations
While this compound is generally well-tolerated, it can lead to side effects such as gastrointestinal disturbances and a risk of C. difficile-associated diarrhea. The incidence of C. difficile infection has been noted to increase with this compound use, necessitating careful patient monitoring .
Important Considerations:
- Caution in prescribing for patients with a history of gastrointestinal disorders.
- Monitoring for signs of C. difficile infection during treatment.
常见问题
Basic Research Questions
Q. How can researchers design bioequivalence trials for generic clindamycin formulations, and what statistical criteria ensure validity?
- Methodological Answer : Bioequivalence studies should follow CHMP guidelines, using a randomized, crossover design with 90% confidence intervals for AUC0-inf and Cmax within 0.8–1.24. Linear pharmacokinetics (150–600 mg dose range) justify dose selection (e.g., 300 mg). Analytical methods (e.g., HPLC) and adherence to EMEA/CHMP/EWP/40326/2006 ensure reproducibility .
Q. What experimental methods are recommended to detect inducible this compound resistance in Staphylococcus aureus?
- Methodological Answer : Use the D-zone test: place erythromycin (15 µg) and this compound (2 µg) discs 15 mm apart on Mueller-Hinton agar. Flattening of the this compound inhibition zone near erythromycin indicates inducible resistance. Confirm with CLSI M100 standards and statistical tools (e.g., SPSS) for data analysis .
Q. How should systematic reviews assess this compound’s efficacy in preventing post-surgical infections?
- Methodological Answer : Employ PRISMA guidelines, extract data from RCTs using tools like the Cochrane Risk of Bias Tool (e.g., randomization, blinding). Pool data via meta-analysis (fixed/random effects models) and address heterogeneity with sensitivity analysis. Focus on outcomes like infection rates in third molar extractions .
Q. What are key considerations for designing in vitro susceptibility testing of this compound against drug-resistant Staphylococci?
- Methodological Answer : Use microdilution methods to determine MICs, adhering to CLSI-M100 standards. Include positive controls (e.g., ciprofloxacin) and analyze data with Fisher’s exact test to compare sensitivity across strains. Account for regional resistance patterns in study design .
Advanced Research Questions
Q. How can physiologically based pharmacokinetic (PBPK) models optimize this compound dosing in pediatric populations?
- Methodological Answer : Develop PBPK models using adult PK data (extracted via Plot Digitizer®) and scale parameters (e.g., organ weights, enzyme expression) using ontogeny functions. Validate with opportunistic pediatric data and software like Simcyp®. Address variability in CYP3A4 maturation .
Q. What molecular dynamics (MD) approaches elucidate this compound resistance mechanisms in bacterial ribosomes?
- Methodological Answer : Simulate this compound binding to wild-type (WT) and mutant (A2058G) 23S rRNA ribosome fragments in explicit solvent. Analyze conformational flexibility (RMSD, RMSF) and stacking interactions (e.g., G2505-U2506) using GROMACS/AMBER. Correlate findings with in vitro resistance data .
Q. How can factorial design optimize this compound-loaded nanogel formulations for enhanced delivery?
- Methodological Answer : Apply 3<sup>2</sup> full factorial design to evaluate independent variables (e.g., polymer concentration, cross-linker ratio). Use DOE software to analyze responses (entrapment efficiency, release kinetics). Validate with in vitro characterization (e.g., TEM, DSC) .
Q. What pharmacovigilance strategies address this compound-associated C. difficile colitis in clinical trials?
- Methodological Answer : Monitor diarrhea incidence (>20% systemic cases) and confirm pseudomembranous colitis via toxin PCR. Exclude high-risk patients (e.g., prior C. difficile history) and analyze covariates (e.g., age, concomitant antibiotics) using logistic regression .
Q. How do cross-resistance patterns between this compound and macrolides inform combination therapy design?
- Methodological Answer : Test for erm/mef resistance genes via PCR in isolates with MLSB phenotypes. Use checkerboard assays to quantify synergism (FIC index ≤0.5) with erythromycin. Model PK/PD interactions (e.g., AUC/MIC) to optimize dosing .
Q. What statistical methods resolve contradictions in this compound’s efficacy for bacterial vaginosis trials?
属性
IUPAC Name |
(2S,4R)-N-[(1S,2S)-2-chloro-1-[(2R,3R,4S,5R,6R)-3,4,5-trihydroxy-6-methylsulfanyloxan-2-yl]propyl]-1-methyl-4-propylpyrrolidine-2-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C18H33ClN2O5S/c1-5-6-10-7-11(21(3)8-10)17(25)20-12(9(2)19)16-14(23)13(22)15(24)18(26-16)27-4/h9-16,18,22-24H,5-8H2,1-4H3,(H,20,25)/t9-,10+,11-,12+,13-,14+,15+,16+,18+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
KDLRVYVGXIQJDK-AWPVFWJPSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCCC1CC(N(C1)C)C(=O)NC(C2C(C(C(C(O2)SC)O)O)O)C(C)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCC[C@@H]1C[C@H](N(C1)C)C(=O)N[C@@H]([C@@H]2[C@@H]([C@@H]([C@H]([C@H](O2)SC)O)O)O)[C@H](C)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C18H33ClN2O5S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
21462-39-5 (mono-hydrochloride), 58207-19-5 (mono-HCl, mono-hydrate) | |
| Record name | Clindamycin [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0018323449 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID2022836 | |
| Record name | Clindamycin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2022836 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
425.0 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
Clindamycin may be bacteriostatic or bactericidal in action, depending on the concentration of the drug attained at the site of infection and the susceptibility of the infecting organism. Clindamycin palmitate hydrochloride and clindamycin phosphate are inactive until hydrolyzed to free clindamycin. This hydrolysis occurs rapidly in vivo. Clindamycin appears to inhibit protein synthesis in susceptible organisms by binding to 50S ribosomal subunits; the primary effect is inhibition of peptide bond formation. The site of action appears to be the same as that of erythromycin, chloramphenicol, and lincomycin., Clindamycin binds exclusively to the 50S subunit of bacterial ribosomes and suppresses protein synthesis., ... Clindamycin is not a substrate for macrolide efflux pumps, and strains that are resistant to macrolides by this mechanism are susceptible to clindamycin. | |
| Record name | CLINDAMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3037 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Yellow, amorphous solid | |
CAS No. |
18323-44-9 | |
| Record name | Clindamycin | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=18323-44-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Clindamycin [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0018323449 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Clindamycin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2022836 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Clindamycin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.038.357 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CLINDAMYCIN | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/3U02EL437C | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | CLINDAMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3037 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
体外研究产品的免责声明和信息
请注意,BenchChem 上展示的所有文章和产品信息仅供信息参考。 BenchChem 上可购买的产品专为体外研究设计,这些研究在生物体外进行。体外研究,源自拉丁语 "in glass",涉及在受控实验室环境中使用细胞或组织进行的实验。重要的是要注意,这些产品没有被归类为药物或药品,他们没有得到 FDA 的批准,用于预防、治疗或治愈任何医疗状况、疾病或疾病。我们必须强调,将这些产品以任何形式引入人类或动物的身体都是法律严格禁止的。遵守这些指南对确保研究和实验的法律和道德标准的符合性至关重要。















