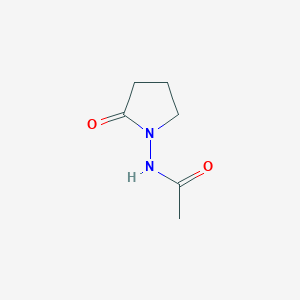
Piracetam
概要
説明
ピラセタムは、ラセタム系に属するノオトロピック薬であり、化学名は2-オキソ-1-ピロリジンアセトアミドです。それは、神経伝達物質γ-アミノ酪酸(GABA)の環状誘導体です。ピラセタムは1971年にUCB Pharmaによって最初に販売され、認知増強作用で知られています。これは、さまざまな認知障害、めまい、皮質ミオクローヌス、失読症、鎌状赤血球貧血で使用されています .
準備方法
合成経路と反応条件
ピラセタムは、いくつかの方法で合成できます。一般的な方法の1つは、塩基の存在下で2-ピロリドンをクロロ酢酸エチルと反応させ、続いて加水分解してピラセタムを得る方法です。別の方法は、γ-アミノ酪酸(GABA)を無水酢酸と環化させる方法です .
工業生産方法
ピラセタムの工業生産では、通常、前述の方法を用いた大規模合成が行われます。このプロセスは、高収率と高純度を実現するように最適化されており、最終製品が医薬品の基準を満たすことが保証されています。生産プロセスには、純粋な化合物を得るための結晶化、ろ過、乾燥などの工程が含まれます .
化学反応の分析
反応の種類
ピラセタムは、以下を含むさまざまな化学反応を受けます。
酸化: ピラセタムは酸化されて2-ピロリドン-5-カルボン酸を生成します。
還元: ピラセタムの還元により、2-ピロリドンが生成されます。
一般的な試薬と条件
酸化: 一般的な酸化剤には、過マンガン酸カリウムや過酸化水素などがあります。
還元: 水素化リチウムアルミニウムなどの還元剤を使用できます。
生成される主な生成物
酸化: 2-ピロリドン-5-カルボン酸
還元: 2-ピロリドン
置換: さまざまなアセトアミド誘導体
科学研究における用途
ピラセタムは、幅広い科学研究用途を持っています。
化学: 環状アミドとその誘導体の特性を研究するためのモデル化合物として使用されます。
生物学: 細胞膜の流動性と神経保護に対する影響が調査されています。
医学: 認知障害、ミオクローヌス、鎌状赤血球貧血の治療に使用されます。認知症、めまい、失読症などの症状に対する潜在的な利点についても研究されています。
科学的研究の応用
Piracetam has a wide range of scientific research applications:
Chemistry: Used as a model compound for studying the properties of cyclic amides and their derivatives.
Biology: Investigated for its effects on cell membrane fluidity and neuroprotection.
Medicine: Used in the treatment of cognitive disorders, myoclonus, and sickle cell anemia. It has also been studied for its potential benefits in conditions like dementia, vertigo, and dyslexia.
Industry: Utilized in the development of nootropic supplements and cognitive enhancers .
作用機序
ピラセタムの作用機序は完全に解明されていませんが、いくつかの経路が関与していると考えられています。
神経伝達物質の調節: ピラセタムは、コリン作動性およびグルタミン酸作動性システムにおける神経伝達を調節します。
神経保護: 神経保護作用があり、神経可塑性を強化し、低酸素状態から保護します。
類似の化合物との比較
ピラセタムは、以下を含む他のラセタムと比較されることがよくあります。
オキシラセタム: 刺激作用で知られており、認知増強のために使用されます。
アニラセタム: 不安解消作用があり、不安障害や認知障害に使用されます。
プラミラセタム: ピラセタムよりも強力であり、外傷性脳損傷に関連する認知障害に使用されます。
フェニルピラセタム: より強力であり、認知増強や身体能力の向上など、より幅広い適応症に使用されます .
ピラセタムは、その安全性の高いプロファイルと幅広い用途で独特であり、研究と臨床の両方において汎用性の高い化合物となっています。
類似化合物との比較
Piracetam is often compared with other racetams, including:
Oxiracetam: Known for its stimulating effects and used for cognitive enhancement.
Aniracetam: Has anxiolytic properties and is used for anxiety and cognitive disorders.
Pramiracetam: More potent than this compound, used for cognitive deficits associated with traumatic brain injuries.
Phenylthis compound: More potent and used for a wider range of indications, including cognitive enhancement and physical performance .
This compound is unique in its well-documented safety profile and broad range of applications, making it a versatile compound in both research and clinical settings.
生物活性
Piracetam, a nootropic compound first synthesized in the 1960s, is widely recognized for its cognitive-enhancing properties. It belongs to the racetam family of drugs and has been extensively studied for its effects on brain function, neuroprotection, and various neurological disorders. This article delves into the biological activity of this compound, highlighting its mechanisms of action, therapeutic applications, and relevant research findings.
This compound exhibits several mechanisms that contribute to its biological activity:
- Neurotransmitter Modulation : this compound modulates neurotransmission by enhancing cholinergic, serotonergic, noradrenergic, and glutamatergic pathways. It increases the density of postsynaptic receptors without binding strongly to them (Ki > 10μM) . This modulation facilitates improved cognitive processes such as learning and memory.
- Membrane Fluidity : The compound interacts with phospholipid membranes, promoting membrane fluidity and stability. This action is crucial for maintaining the structure and function of transmembrane proteins involved in neurotransmission .
- Neuroprotection : this compound has demonstrated neuroprotective effects against various forms of neuronal injury, including oxidative stress induced by lipopolysaccharides (LPS). It reduces intracellular reactive oxygen species (ROS) and nitric oxide (NO), which are implicated in neuroinflammation and cell death .
Therapeutic Applications
This compound has been investigated for a range of clinical applications:
- Cognitive Enhancement : Numerous studies have reported improvements in cognitive functions such as memory, attention, and consciousness in both healthy individuals and those with cognitive impairments .
- Neurodegenerative Disorders : Research suggests that this compound may have potential benefits in conditions like Alzheimer's disease and vascular dementia by enhancing synaptic plasticity and protecting neurons from damage .
- Anticonvulsant Properties : this compound has been shown to possess anticonvulsant properties, making it a candidate for treating epilepsy .
Case Studies and Clinical Trials
A summary of key studies investigating the biological activity of this compound is presented in Table 1:
| Study | Findings | Dosage | Population |
|---|---|---|---|
| Ahmed & Oswal (2010) | Continuous positive allosteric modulation on AMPA receptors; enhances neuronal excitation | 200 mg/kg | Rats |
| Colucci et al. (2012) | Improves acetylcholine functions via muscarinic receptors | 400 mg/kg | Rats |
| Braz. J. Pharm. Sci. (2022) | Reduces LPS-induced oxidative stress; improves SOD levels | 200-400 mg/kg | Mice |
Neuroprotective Effects
A notable study highlighted this compound's capacity to protect against LPS-induced neuronal toxicity. The treatment significantly reduced levels of inflammatory cytokines like IL-6 and amyloid-beta, indicating its potential role in mitigating neuroinflammation . Additionally, this compound was found to enhance antioxidant enzyme activities such as catalase and superoxide dismutase (SOD), further supporting its neuroprotective profile.
Q & A
Basic Research Questions
Q. What are the primary neuropharmacological mechanisms of action for Piracetam, and how are they experimentally validated?
this compound enhances cell membrane fluidity, modulates neurotransmission (particularly cholinergic and glutamatergic systems), and increases oxygen consumption via adenylate kinase activity in the brain . These mechanisms are validated through in vivo models assessing acetylcholine release, membrane permeability assays, and behavioral tests in rodents. For example, studies measuring synaptic plasticity changes or oxygen utilization in ischemic brains provide direct evidence .
Q. What experimental evidence supports this compound’s neuroprotective efficacy in preclinical stroke models?
Meta-analyses of animal stroke models show this compound and derivatives improve outcomes by 30.2% (95% CI: 16.1–44.4), primarily via infarct size reduction and neurological score improvements. However, only two studies directly tested this compound in stroke models, with median quality scores of 4/10, highlighting methodological limitations . Key assays include histopathological analysis of infarct regions and Morris water maze tests for functional recovery .
Q. How is this compound’s dosage optimized in experimental settings, and what factors influence dose-response relationships?
Preclinical studies use dose-ranging protocols (e.g., 80–100 mg/kg/day in rodents) to establish efficacy and safety thresholds. For instance, LD50 values in mice (455.5 mg/kg) inform toxicity limits . Dose-response linearity is observed in epilepsy trials, where 24 g/day in humans showed superior efficacy compared to lower doses . Optimization requires pharmacokinetic profiling (e.g., half-life, blood-brain barrier penetration) and endpoint-specific titration .
Advanced Research Questions
Q. How do contradictory findings in this compound’s clinical efficacy for cognitive impairment arise, and what methodological strategies resolve them?
Meta-analyses report odds ratios of 3.43–3.55 for global improvement in dementia but note heterogeneity (I² > 50%) and publication bias . Contradictions stem from:
- Study design : Crossover trials with unanalyzed first-period data inflate effect sizes .
- Population diversity : Mixed cohorts (Alzheimer’s, vascular dementia) obscure subtype-specific responses . Solutions include stratified randomization, intention-to-treat analysis, and adherence to PRISMA guidelines for systematic reviews .
Q. What critiques apply to the translational gap between this compound’s preclinical promise and clinical trial outcomes?
Preclinical data for stroke were published 10 years after clinical trials began, suggesting animal models were not foundational to trial design . Discrepancies arise from:
- Timing : Animal studies administer this compound ≤6 hours post-stroke, whereas trials allowed 12-hour delays .
- Outcome measures : Rodent infarct size vs. human functional independence scales (e.g., modified Rankin) lack alignment. Bridging this gap requires co-development of preclinical-clinical protocols and biomarker validation (e.g., neuroimaging correlates) .
Q. Why does this compound fail to enhance cognition in Down syndrome despite efficacy in other neurodevelopmental models?
A randomized crossover trial (N=25) found no cognitive improvement in Down syndrome, with adverse effects (e.g., agitation) . Potential explanations:
- Pathophysiological divergence : Down syndrome’s trisomy 21 may alter drug targets (e.g., GABAergic pathways).
- Dosing mismatch : 80–100 mg/kg/day may inadequately penetrate the blood-brain barrier in this population. Mechanistic studies comparing this compound’s effects on Shank3 vs. DYRK1A pathways could clarify etiology-specific responses .
Q. Methodological Recommendations
- For preclinical studies : Use the STAIR criteria for stroke models, including blinded outcome assessment and sample size calculations .
- For clinical trials : Prioritize adaptive designs to refine dosing (e.g., Bayesian response-adaptive randomization) .
- For meta-analyses : Apply GRADE criteria to evaluate evidence certainty and adjust for unpublished data via trim-and-fill analyses .
特性
IUPAC Name |
2-(2-oxopyrrolidin-1-yl)acetamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C6H10N2O2/c7-5(9)4-8-3-1-2-6(8)10/h1-4H2,(H2,7,9) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
GMZVRMREEHBGGF-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CC(=O)N(C1)CC(=O)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C6H10N2O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID5044491 | |
| Record name | 2-Oxo-1-Pyrrolidineacetamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5044491 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
142.16 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Boiling Point |
Decomposes | |
| Record name | Piracetam | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB09210 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Piracetam interacts with the polar heads in the phospholipids membrane and the resulting mobile drug-lipid complexes are thought to reorganize the lipids and influence membrane function and fluidity. Such interaction has been reported in a study that investigated the effects of neuronal outgrowth induced by beta amyloid peptides; while amyloid peptides cause lipid disorganization within the cell membranes leading to neuronal death, piracetam demonstrated to decrease the destabilizing effects of amyloid peptide. The authors suggest that piracetam induces a positive curvature of the membrane by occupying the polar groups in the phospholipids to counteract the negative curvature induced by amyloid peptides , which in turn would decrease the likelihood of membrane fusion. This mechanism of action is thought to improve membrane stability, allowing the membrane and transmembrane proteins to maintain and recover the three-dimensional structure or folding for normal function such as membrane transport, chemical secretion, and receptor binding and stimulation. Through restored membrane fluidity, piracetam promotes restored neurotransmission such as glutamatergic and cholinergic systems, enhances neuroplasticity and mediates neuroprotective and anticonvulsant effects at the neuronal level. It is also demonstrated that piracetam also improves the fluidity of platelet membranes. At the vascular level, piracetam decreases adhesion of erythrocytes to cell wall and reduces vasospasm which in turn improves microcirculation including cerebral and renal blood flow., It was found that a drug of the nootropic nature piracetam possessing pronounced antihypoxic properties eliminates calcium chloride-induced disturbances of the cardiac rhythm and significantly raises the threshold of atrial fibrillation during electrical stimulation. The drug's antiarrhythmic effect is followed by a decrease of the rhythm rate and an increase of the contraction amplitude. The animals treated with piracetam in a dose when its antiarrhythmic effects (300 mg/kg) exhibited a decrease of the membrane potential of erythrocytes as compared with control. Similar effects occurred in the animals treated with lidocaine. It can be concluded that in certain types of arrhythmias the use of piracetam restores the normal rhythm of contractions that is perhaps connected with its positive influence on metabolic processes in the myocardium. | |
| Record name | Piracetam | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB09210 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PIRACETAM | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7529 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from isopropanol | |
CAS No. |
7491-74-9 | |
| Record name | Piracetam | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=7491-74-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Piracetam [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0007491749 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Piracetam | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB09210 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | piracetam | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758191 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 2-Oxo-1-Pyrrolidineacetamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5044491 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Piracetam | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.028.466 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PIRACETAM | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/ZH516LNZ10 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | PIRACETAM | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7529 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Melting Point |
151.5 - 152.5 °C | |
| Record name | Piracetam | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB09210 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PIRACETAM | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7529 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
試験管内研究製品の免責事項と情報
BenchChemで提示されるすべての記事および製品情報は、情報提供を目的としています。BenchChemで購入可能な製品は、生体外研究のために特別に設計されています。生体外研究は、ラテン語の "in glass" に由来し、生物体の外で行われる実験を指します。これらの製品は医薬品または薬として分類されておらず、FDAから任何の医療状態、病気、または疾患の予防、治療、または治癒のために承認されていません。これらの製品を人間または動物に体内に導入する形態は、法律により厳格に禁止されています。これらのガイドラインに従うことは、研究と実験において法的および倫理的な基準の遵守を確実にするために重要です。
















