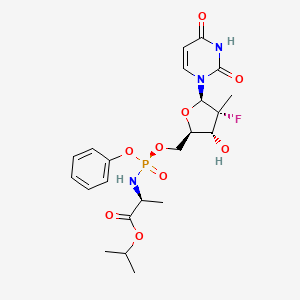
Sofosbuvir
- Click on QUICK INQUIRY to receive a quote from our team of experts.
- With the quality product at a COMPETITIVE price, you can focus more on your research.
Overview
Description
Sofosbuvir is a direct-acting antiviral medication used to treat chronic hepatitis C virus (HCV) infections. It belongs to the nucleotide analog family of medications and works by inhibiting the HCV NS5B RNA-dependent RNA polymerase, which is essential for viral replication . This compound was discovered in 2007 and approved for medical use in the United States in 2013 . It is marketed under the brand name Sovaldi and is included in the World Health Organization’s List of Essential Medicines .
Preparation Methods
The synthesis of sofosbuvir involves several steps, including the preparation of key intermediates and the final coupling reaction. One of the synthetic routes involves the use of (2’R)-2’-deoxy-2’-fluoro-2’-methyluridine as a starting material. This compound is reacted with other raw materials in an organic solvent in the presence of a Lewis acid and an alkali to produce this compound . The reaction conditions are mild, and the process is suitable for large-scale industrial production .
Another method involves a more stereoselective preparation, which allows for the production of the desired isomer without the need for intermediate separation. This method reduces production costs and is also suitable for industrialized production .
Chemical Reactions Analysis
Sofosbuvir undergoes various chemical reactions, including:
Oxidation: This compound can be oxidized to form its active metabolite, GS-461203, which is a triphosphate.
Reduction: Reduction reactions are not commonly associated with this compound.
Substitution: This compound can undergo substitution reactions, particularly involving the fluorine atom on the furanose ring.
Common reagents and conditions used in these reactions include organic solvents, Lewis acids, and alkali . The major products formed from these reactions include the active triphosphate form of this compound and other intermediates used in its synthesis .
Scientific Research Applications
Treatment of Chronic Hepatitis C
Sofosbuvir is primarily indicated for the treatment of chronic hepatitis C across all genotypes. Its effectiveness has been demonstrated in various clinical trials:
- Efficacy Across Genotypes : this compound-based regimens have shown high SVR rates, particularly in genotypes 1 and 2, with studies reporting SVR rates exceeding 90% in treatment-naïve patients .
- Challenges with Genotype 3 : Despite its overall effectiveness, genotype 3 infections present challenges, with lower SVR rates reported, especially in patients with cirrhosis .
Special Populations
This compound has been evaluated for use in special populations, including those with advanced chronic kidney disease (CKD):
- Patients with CKD : A systematic review indicated that this compound-based regimens are safe and effective for patients with stage 4–5 CKD. The pooled SVR rate was found to be 99%, highlighting its utility even in patients with significant renal impairment .
- Co-infection Scenarios : this compound is also effective in patients co-infected with HIV or those who have previously failed other DAA therapies, demonstrating its versatility in complex clinical situations .
Real-World Efficacy
A study involving a cohort of decompensated cirrhotic patients treated with this compound/Velpatasvir reported an impressive SVR12 rate of 94.4%. This highlights the drug's effectiveness even in challenging patient populations .
Phase III Trials
In a Phase III trial conducted in Japan, all participants receiving an all-oral regimen containing this compound achieved undetectable HCV levels by week four, underscoring its rapid action against the virus .
Safety Profile
This compound is generally well-tolerated, but some adverse events have been reported:
- Common Adverse Events : Fatigue, headache, and anemia are frequently observed but are typically manageable. Serious adverse events occur at a rate of approximately 9% in clinical settings .
- Monitoring Recommendations : Close monitoring of renal function is advised for patients with CKD during treatment to mitigate risks associated with potential renal impairment .
Comparative Effectiveness
A systematic review comparing this compound-based regimens with and without ribavirin found that while adding ribavirin may enhance SVR rates in some cases, it also increases the risk of serious adverse events . This underscores the importance of tailoring treatment regimens to individual patient needs.
Mechanism of Action
Sofosbuvir is a prodrug that is metabolized into its active form, GS-461203, within the body . This active form inhibits the HCV NS5B RNA-dependent RNA polymerase by acting as a defective substrate, thereby terminating the viral RNA chain and preventing replication . The inhibition of this enzyme is crucial for the suppression of HCV replication and the achievement of a sustained virologic response .
Comparison with Similar Compounds
Sofosbuvir is often compared with other direct-acting antiviral agents, such as:
Ledipasvir: Used in combination with this compound for the treatment of HCV.
Velpatasvir: Another antiviral agent used in combination with this compound for pan-genotypic treatment of HCV.
Daclatasvir: Used in combination with this compound for the treatment of HCV.
Simeprevir: Used in combination with this compound for the treatment of HCV.
Remdesivir: An antiviral agent used for the treatment of COVID-19, which also targets RNA-dependent RNA polymerase.
This compound is unique in its high barrier to resistance development and its effectiveness across multiple HCV genotypes . Its combination with other antiviral agents enhances its efficacy and broadens its application in treating various HCV genotypes .
Q & A
Basic Research Questions
Q. What validated analytical methods are available for quantifying sofosbuvir in pharmaceutical formulations or biological matrices?
- Methodology : Reverse-phase high-performance liquid chromatography (RP-HPLC) is widely used, with parameters optimized via Quality by Design (QbD) approaches. For example, a QbD-based method optimized mobile phase ratio (e.g., acetonitrile:buffer), buffer pH (e.g., phosphate buffer at pH 4.5–6.5), and column type (C18 or phenyl-hexyl) using Design-Expert® software. This method achieved retention times of 2.5–4.5 minutes, peak tailing <2, and theoretical plates >5,000 .
- Validation : Include specificity, linearity (e.g., 10–100 µg/mL), precision (RSD <2%), and robustness against pH or flow-rate variations .
Q. How are clinical trials for this compound-based regimens designed to assess efficacy against HCV genotypes?
- Design : Trials often use randomized, controlled designs with stratification by genotype (e.g., genotype 2 vs. 3), cirrhosis status, and prior treatment history. For example, the VALENCE trial extended treatment duration for genotype 3 from 12 to 24 weeks after interim analysis showed improved sustained virologic response (SVR) rates (85% vs. 56% in cirrhotic patients) .
- Endpoints : Primary endpoints include SVR at 12 weeks post-treatment (SVR12), analyzed via intention-to-treat. Secondary endpoints may address adverse events (e.g., headache, fatigue) and subgroup efficacy .
Q. What statistical tools are employed to analyze this compound clinical trial data?
- Tools : SPSS or R for descriptive statistics (mean, SD) and inferential tests (Chi-square, paired t-tests). For example, a study comparing dual (this compound + ribavirin) vs. triple therapy (with peginterferon) used a p-value ≤0.05 to define significance in SVR rates .
Advanced Research Questions
Q. How can multivariate chemometric models resolve challenges in quantifying this compound in combination therapies (e.g., with ledipasvir)?
- Approach : Genetic algorithm-partial least squares (GA-PLS) and artificial neural networks (ANN) improve precision in spectral data analysis. For instance, GA-PLS applied to UV-Vis spectra (200–400 nm) of this compound/ledipasvir mixtures achieved recovery rates of 98–102% with RSD <1.5% .
- Validation : Cross-validate models using synthetic mixtures and real-world samples, ensuring minimal interference from excipients or metabolites .
Q. What experimental designs optimize this compound-loaded lipid nanocarriers for enhanced bioavailability?
- Design : A 2³ factorial design evaluates factors like lipid concentration (X1), cholesterol content (X2), and centrifugation speed (X3). Responses such as drug entrapment efficiency (DEE >80%) and particle size (<200 nm) are modeled via Design-Expert® software. For example, high X1 and low X3 maximized DEE while minimizing particle size .
- Characterization : Use dynamic light scattering (DLS) for size distribution and in vitro release studies in simulated intestinal fluid (pH 6.8) .
Q. How do researchers address contradictory efficacy data for this compound across HCV genotypes?
- Analysis : Stratify data by genotype and cirrhosis status. Genotype 3 patients show lower SVR rates (68% with cirrhosis vs. 91% without) compared to genotype 2 (93% SVR). Meta-analyses or pooled data from trials like FISSION and NEUTRINO can reconcile discrepancies by adjusting for covariates (e.g., viral load, IL28B polymorphisms) .
Q. What advanced bioanalytical techniques quantify this compound metabolites (e.g., GS-331007) in plasma?
- Technique : Online SPE-LC coupled to Q-Exactive HRMS enables sensitive detection (LOQ: 1 ng/mL) using t-MS² or t-SIM modes. This method validated this compound’s pharmacokinetics, including its short half-life (0.4 hours) and metabolite GS-331007’s prolonged half-life (27 hours) .
Q. How can QbD principles improve robustness in this compound assay development?
- Implementation : Define critical quality attributes (CQAs) like retention time and peak symmetry. Use risk assessment (e.g., Ishikawa diagrams) to prioritize factors (mobile phase, column temperature). A 14-run experimental design identified optimal conditions (e.g., pH 5.0, 70:30 acetonitrile:buffer) with robustness confirmed via intermediate precision testing (RSD <1.5%) .
Q. Methodological Frameworks
Q. How to formulate a research question for this compound studies addressing gaps in HCV management?
- Process : Start with systematic reviews to identify gaps (e.g., optimizing regimens for genotype 4). Narrow questions using PICOT criteria: Population (HCV genotype 4 patients), Intervention (this compound/velpatasvir), Comparison (historical interferon-based therapies), Outcome (SVR12), Time (12 weeks). Sub-questions may explore resistance-associated substitutions (RASs) or cost-effectiveness in low-resource settings .
Q. What strategies mitigate bias in this compound clinical trials with open-label designs?
Properties
Key on ui mechanism of action |
Sofosbuvir is a direct-acting antiviral agent (pan-genotypic polymerase inhibitor) against the hepatitis C virus. HCV RNA replication is mediated by a membrane-associated multiprotein replication complex. The HCV polymerase (NS5B protein) is an RNA-dependent RNA polymerase (RdRp). It is the essential initiating and catalytic subunit of this replication complex and is critical for the viral replication cycle. There is no human homolog for HCV NS5B RdRp. Sofosbuvir is a monophosphorylated pyrimidine nucleotide prodrug that undergoes intracellular metabolism to form the pharmacologically active uridine analog triphosphate (GS-461203). GS-461203 competes with natural nucleotides for incorporation (by HCV NS5B) into the nascent RNA strand during replication of the viral genome. GS-461203 differs from endogenous pyrimidine nucleotides in that it has been modified at the 2' position with the addition of a methyl and a fluoro functional group. Incorporation of GS-461203 into nascent RNA strongly reduces the efficiency of further RNA elongation by RdRp, resulting in premature termination of RNA synthesis. The stopping of viral replication leads to a rapid decline of HCV viral load and clearing of HCV levels in the body. |
|---|---|
CAS No. |
1190307-88-0 |
Molecular Formula |
C22H29FN3O9P |
Molecular Weight |
529.5 g/mol |
IUPAC Name |
propan-2-yl 2-[[[(4R)-5-(2,4-dioxopyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyloxolan-2-yl]methoxy-phenoxyphosphoryl]amino]propanoate |
InChI |
InChI=1S/C22H29FN3O9P/c1-13(2)33-19(29)14(3)25-36(31,35-15-8-6-5-7-9-15)32-12-16-18(28)22(4,23)20(34-16)26-11-10-17(27)24-21(26)30/h5-11,13-14,16,18,20,28H,12H2,1-4H3,(H,25,31)(H,24,27,30)/t14?,16?,18?,20?,22-,36+/m1/s1 |
InChI Key |
TTZHDVOVKQGIBA-IECBXEDQSA-N |
SMILES |
CC(C)OC(=O)C(C)NP(=O)(OCC1C(C(C(O1)N2C=CC(=O)NC2=O)(C)F)O)OC3=CC=CC=C3 |
Isomeric SMILES |
CC(C)OC(=O)C(C)N[P@](=O)(OCC1C([C@@](C(O1)N2C=CC(=O)NC2=O)(C)F)O)OC3=CC=CC=C3 |
Canonical SMILES |
CC(C)OC(=O)C(C)NP(=O)(OCC1C(C(C(O1)N2C=CC(=O)NC2=O)(C)F)O)OC3=CC=CC=C3 |
Appearance |
white solid powder |
Color/Form |
White to off-white crystalline solid |
Purity |
>98% (or refer to the Certificate of Analysis) |
shelf_life |
>2 years if stored properly |
solubility |
Slightly soluble in water |
storage |
Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
Synonyms |
PSI-7977; PSI 7977; PSI7977; GS7977; GS-7977; GS 7977; Sofosbuvir; Sovaldi; Virunon; Vosevi; Hepcinat; Hepcvir; Resof; |
Origin of Product |
United States |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















