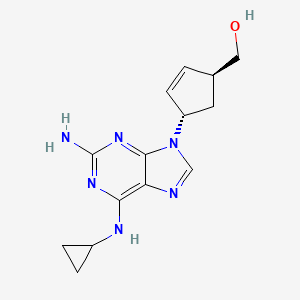
Abacavir
Overview
Description
Abacavir is a synthetic carbocyclic nucleoside analog reverse transcriptase inhibitor used primarily in the treatment of Human Immunodeficiency Virus (HIV) and Acquired Immunodeficiency Syndrome (AIDS). It is marketed under the brand name Ziagen among others. This compound is typically used in combination with other antiretroviral medications and is not recommended for use alone . It is known for its ability to inhibit the replication of HIV by targeting the reverse transcriptase enzyme, which is crucial for the virus’s replication process .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of abacavir involves several steps, starting from a suitable di-halo aminopyrimidine compound. The key steps include:
Reaction with Aminoalcohol: The di-halo aminopyrimidine is reacted with an aminoalcohol to form an intermediate compound.
Cyclization: This intermediate undergoes cyclization to form a key intermediate.
Introduction of Cyclopropylamine: The chlorine atom in the intermediate is displaced with cyclopropylamine to yield this compound as a free base.
Industrial Production Methods: Industrial production of this compound often involves optimizing the reaction conditions to ensure high yield and purity. This includes controlling the temperature, pH, and the use of specific catalysts to facilitate the reactions. The process may also involve purification steps such as crystallization and chromatography to isolate the final product .
Types of Reactions:
Oxidation: this compound undergoes oxidative degradation, which can be studied using electrochemical methods.
Reduction: While specific reduction reactions of this compound are less documented, it is possible that under certain conditions, this compound could undergo reduction.
Substitution: The synthesis of this compound itself involves substitution reactions, particularly the displacement of chlorine with cyclopropylamine.
Common Reagents and Conditions:
Oxidation: Hydrogen peroxide, platinum electrodes, boron-doped diamond electrodes.
Substitution: Cyclopropylamine, various solvents and catalysts.
Major Products:
Scientific Research Applications
Clinical Efficacy in HIV Treatment
Abacavir is FDA-approved for use in adults and children over three months old as part of combination antiretroviral therapy (ART). It is often administered alongside other agents such as lamivudine and dolutegravir.
Clinical Trials and Outcomes
- A pivotal study compared this compound with zidovudine combined with lamivudine. At week 48, 70% of patients on this compound achieved plasma HIV-1 RNA levels of ≤50 copies/mL, similar to the zidovudine group (69%) .
- The median increase in CD4+ cell counts was significantly higher in the this compound group (209 cells/mm³) compared to the zidovudine group (155 cells/mm³), indicating a robust immunological response .
Pharmacokinetics
This compound exhibits a two-compartment pharmacokinetic model with high oral bioavailability. Key pharmacokinetic parameters include:
- Clearance : Increased from a mean of 3.33 to 5.86 L/h/7 kg from day 1 to day 14 in severely malnourished children .
- Central Nervous System Penetration : Studies indicate that this compound penetrates the central nervous system effectively, with a CSF/plasma concentration ratio of approximately 31% to 44% .
Safety Profile and Side Effects
While this compound is generally well-tolerated, it has been associated with hypersensitivity reactions, particularly in individuals with the HLA-B*57:01 allele. This genetic predisposition can lead to severe allergic reactions upon exposure to the drug.
Cardiovascular Risks
Recent analyses have linked this compound use to an increased risk of major adverse cardiovascular events (MACE). A meta-analysis indicated that current or past use of this compound may elevate cardiovascular disease risk among HIV patients .
Off-Label Uses
This compound has been explored for various off-label applications, including:
- HIV Treatment in Special Populations : Research has demonstrated its efficacy in severely malnourished children, showing that WHO weight-band dosing recommendations are appropriate for this demographic .
- Potential Role in Cancer Therapy : Emerging studies are investigating the immunomodulatory effects of this compound that may enhance responses to cancer therapies.
Case Studies and Research Findings
Several studies have documented the diverse applications and outcomes associated with this compound:
Mechanism of Action
Abacavir is converted intracellularly to its active metabolite, carbovir triphosphate. This metabolite is an analog of deoxyguanosine-5’-triphosphate (dGTP) and competes for incorporation into viral DNA by the HIV reverse transcriptase enzyme. Once incorporated, it acts as a chain terminator, preventing the elongation of the viral DNA and thereby inhibiting the replication of the virus .
Comparison with Similar Compounds
Lamivudine: Another nucleoside reverse transcriptase inhibitor used in combination with abacavir.
Zidovudine: An older nucleoside reverse transcriptase inhibitor with a similar mechanism of action.
Tenofovir: A nucleotide reverse transcriptase inhibitor with a different chemical structure but similar antiviral activity.
Comparison:
Uniqueness: this compound is unique in its structure as a carbocyclic nucleoside analog. It has a distinct cyclopropylamine group that differentiates it from other nucleoside analogs.
Safety Profile: this compound has a well-documented safety profile, though it is associated with a risk of hypersensitivity reactions and potential cardiovascular risks.
Biological Activity
Abacavir is a nucleoside reverse transcriptase inhibitor (NRTI) primarily used in the treatment of HIV infection. Its biological activity is characterized by its mechanism of action, pharmacokinetics, therapeutic efficacy, safety profile, and potential side effects, including hypersensitivity reactions. This article synthesizes findings from various studies and case reports to provide a comprehensive overview of this compound's biological activity.
This compound is metabolized intracellularly to its active form, carbovir triphosphate (CBV-TP), which competes with natural deoxyguanosine triphosphate for incorporation into viral DNA. This incorporation results in chain termination during reverse transcription, effectively inhibiting viral replication. This compound has demonstrated potency against HIV strains resistant to other NRTIs, making it a valuable option in antiretroviral therapy (ART) regimens .
Pharmacokinetics
The pharmacokinetic profile of this compound reveals significant insights into its absorption, distribution, metabolism, and excretion:
- Absorption : this compound is rapidly absorbed after oral administration, with peak plasma concentrations occurring within 1.5 to 2 hours.
- Distribution : The drug penetrates the central nervous system (CNS) effectively, with cerebrospinal fluid (CSF) concentrations significantly exceeding the inhibitory concentration for HIV .
- Metabolism : this compound is primarily metabolized by the liver through non-CYP450 pathways, reducing the risk of drug-drug interactions commonly associated with other antiretrovirals .
- Excretion : The drug is eliminated mainly via renal clearance, with minimal hepatic metabolism .
Efficacy in Clinical Studies
This compound has been evaluated in numerous clinical trials demonstrating its efficacy as part of ART regimens:
- In a comparative study with zidovudine and lamivudine, this compound showed non-inferior efficacy in maintaining viral suppression (70% vs. 69% at week 48) and a favorable CD4+ cell response (209 cells/mm³ vs. 155 cells/mm³) .
- A meta-analysis indicated that viral suppression rates ranged from 50% to 70% at six months and from 57% to 78% at twelve months in pediatric populations treated with this compound .
Safety Profile and Hypersensitivity Reactions
While this compound is generally well-tolerated, it is associated with hypersensitivity reactions (HSRs) in a subset of patients. The incidence of HSRs can be influenced by genetic factors such as HLA-B*5701 status:
- Case Study Example : A patient developed gastrointestinal symptoms and fever within days of initiating therapy with this compound. Upon discontinuation, symptoms resolved rapidly, indicating a probable hypersensitivity reaction .
Table 1: Summary of this compound Hypersensitivity Reactions
| Study/Case | Population | Symptoms | Outcomes |
|---|---|---|---|
| Case #3 | Adult male | Vomiting, diarrhea, fever | Symptoms resolved post-discontinuation |
| Patel et al. (2020) | Pediatric patients | Variable symptoms | Incidence rate varied from 0% to 5.49% |
Research Findings on Oncogenic Activity
Recent studies have also explored the potential oncogenic effects of this compound. Research indicates that this compound can activate oncogenic transcription factors in gastric cancer cells, suggesting a complex role beyond its antiviral activity:
Chemical Reactions Analysis
Metabolic Pathways and Enzymatic Reactions
Abacavir is metabolized via two primary enzymatic pathways: alcohol dehydrogenase (ADH) and UDP-glucuronosyltransferase (UGT) . These reactions produce inactive metabolites, ensuring systemic clearance while avoiding cytochrome P450 (CYP) interactions .
Table 1: Major Metabolites and Excretion Pathways
| Metabolite | Enzyme Responsible | % Urinary Excretion | Activity |
|---|---|---|---|
| Carbovir 5′-carboxylate | ADH | 30% | Inactive |
| This compound 5′-glucuronide | UGT | 36% | Inactive |
| Unchanged this compound | - | 1.2% | Active |
Activation to Carbovir Triphosphate (CBV-TP)
This compound’s antiviral activity depends on intracellular phosphorylation to carbovir triphosphate (CBV-TP) , a guanosine analogue. This occurs in a multi-step enzymatic process :
-
Phosphorylation : this compound → Carbovir monophosphate (via adenosine kinase).
-
Diphosphorylation : Carbovir monophosphate → Carbovir diphosphate (via guanylate kinase).
-
Triphosphorylation : Carbovir diphosphate → CBV-TP (via nucleoside diphosphate kinases).
CBV-TP competitively inhibits HIV reverse transcriptase and terminates viral DNA synthesis .
Drug-Drug Interactions Affecting Reactivity
This compound’s chemical stability and metabolism are influenced by co-administered drugs:
Ethanol Interaction
Ethanol inhibits ADH, increasing this compound plasma levels by reducing its oxidation to the carboxylate metabolite .
Protease Inhibitors (e.g., Tipranavir)
Tipranavir induces UGT activity, accelerating glucuronidation and reducing this compound bioavailability .
Table 2: Clinically Significant Interactions
| Interacting Drug | Effect on this compound | Mechanism | Outcome |
|---|---|---|---|
| Ethanol | ↑ Plasma levels | ADH inhibition | Risk of toxicity |
| Tipranavir/Ritonavir | ↓ Plasma levels | UGT induction | Reduced efficacy |
| Methadone | ↓ Methadone levels | Disputed mechanism | Potential opioid withdrawal |
Physicochemical Stability
This compound’s stability under varying conditions is critical for formulation and storage:
-
pH Sensitivity : Degrades rapidly in strongly acidic (pH < 3) or alkaline (pH > 9) environments.
-
Thermal Stability : Stable at room temperature but degrades above 40°C .
Hypersensitivity-Linked Conformational Dynamics
Though not a direct chemical reaction, this compound’s binding to HLA-B*57:01 alters peptide presentation in the antigen-binding cleft, triggering immune reactions. Molecular dynamics simulations show this compound adopts flexible conformations within HLA-B*57:01, enabling T-cell receptor activation .
Q & A
Basic Research Questions
Q. What methodologies are used to determine abacavir's resistance profile in HIV-1 reverse transcriptase?
Resistance profiling involves genotypic and phenotypic analyses. Key mutations (e.g., M184V, L74V) are identified via sequencing of the reverse transcriptase gene, followed by in vitro cross-resistance assays. Clinical validation includes correlating baseline mutations with virologic response using univariate/multivariate analyses and bootstrap resampling to ensure robustness . For example, mutations at codons 41, 67, 210, 215, 74, and 184 are linked to reduced this compound efficacy, with virologic response decreasing as mutation count increases .
Q. How is HLA-B*5701 screening validated for preventing this compound hypersensitivity?
Validation follows a stepwise approach: (1) Retrospective case-control studies identify HLA-B5701 as a risk allele (odds ratio >900); (2) Prospective randomized trials confirm screening efficacy, demonstrating elimination of immunologically confirmed hypersensitivity (0% in screened vs. 2.7% in controls); (3) Patch testing and CD8+ T-cell depletion validate immune mechanisms . Screening reduces hypersensitivity risk to <1% in HLA-B5701-negative populations .
Q. What experimental designs are used to assess this compound's impact on thrombin generation (TG)?
TG is quantified via calibrated automated thrombography, measuring parameters like endogenous thrombin potential (ETP) and peak thrombin. Studies compare this compound-treated vs. non-abacavir cohorts, adjusting for confounders (age, inflammation) using multivariate regression. For example, this compound increases ETP by 8% (p=0.006) and peak thrombin by 6% (p=0.083), indicating prothrombotic effects .
Advanced Research Questions
Q. How do conflicting data on this compound-associated cardiovascular risk inform study design?
Observational studies (e.g., D:A:D cohort) report increased myocardial infarction (MI) risk (RR 1.92), while randomized trials (ACTG A5001) show no association. Methodological reconciliation involves:
- Cohort stratification : Differentiating recent vs. cumulative this compound exposure .
- Confounder adjustment : Controlling for traditional cardiovascular risk factors (smoking, hypertension) and HIV-specific inflammation .
- Meta-analytic weighting : Pooling data from 27 studies, highlighting heterogeneity in study power and design .
Q. What advanced analytical techniques quantify this compound in antiretroviral formulations?
Molecular fluorescence spectroscopy coupled with UV-induced degradation generates second/third-order data. Chemometric tools like multivariate curve resolution-alternating least squares (MCR-ALS) and parallel factor analysis (PARAFAC) resolve spectral overlaps, achieving >95% accuracy in degradation profiling .
Q. How do pharmacokinetic (PK) models address variability in pediatric this compound dosing?
Population PK analyses using nonlinear mixed-effects modeling (e.g., NONMEM) identify covariates (age, weight) influencing drug clearance. For children, weight-normalized dosing (16 mg/kg/day) minimizes variability (CV <30%), with no formulation differences (tablet vs. solution) .
Q. What systems biology approaches elucidate this compound's metabolic interactions?
Pathway annotation integrates enzyme kinetics (e.g., UDP-glucuronosyltransferase activity) and transport data (e.g., ABCB1 efflux) into dynamic models. For example, this compound’s interaction with Hsp70-Hom (M493T polymorphism) is modeled to predict HLA-B*5701-restricted immune activation .
Q. Contradictory Data Analysis
Q. How to resolve discrepancies in this compound's association with thrombin generation vs. clinical thrombosis?
While ex vivo TG assays show increased ETP in this compound users , clinical thrombosis risk depends on:
- Protein C activity : No difference in normalized ETP after adjusting for protein C .
- Cohort selection : Studies prioritize PLHIV with low baseline CVD risk, diluting thrombotic endpoints .
- Temporal dynamics : Current vs. past this compound use shows divergent effects (current use: B=8.85, p=0.002) .
Q. Why does HLA-B*5701 not universally predict this compound hypersensitivity?
HLA-B*5701 sensitivity varies ethnically:
- Caucasians/Hispanics : Strong association (OR >100) .
- Black populations : No significant association due to haplotype diversity (Hsp70-Hom M493T not linked to HLA-B*5701) . Supplementary biomarkers (e.g., Hsp70-Hom genotyping) improve specificity to 98% in non-Black cohorts .
Q. Methodological Recommendations
Q. How to design cost-effectiveness studies for HLA-B*5701 screening?
Decision-analytic models compare testing costs (~5,000/episode). Monte Carlo simulations show screening is cost-effective (ICER: €22,811/hypersensitivity avoided) when alternative antiretrovirals (e.g., tenofovir) are equally efficacious .
Q. What statistical methods address time-varying confounders in this compound-CVD studies?
Marginal structural models with inverse probability weighting adjust for time-dependent variables (e.g., lipid changes, ART switches). Sensitivity analyses exclude patients with <6 months follow-up to mitigate immortal time bias .
Properties
IUPAC Name |
[4-[2-amino-6-(cyclopropylamino)purin-9-yl]cyclopent-2-en-1-yl]methanol | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C14H18N6O/c15-14-18-12(17-9-2-3-9)11-13(19-14)20(7-16-11)10-4-1-8(5-10)6-21/h1,4,7-10,21H,2-3,5-6H2,(H3,15,17,18,19) | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
MCGSCOLBFJQGHM-UHFFFAOYSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CC1NC2=C3C(=NC(=N2)N)N(C=N3)C4CC(C=C4)CO | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C14H18N6O | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID20861337 | |
| Record name | 4-[2-Amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID20861337 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
286.33 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
136470-78-5, 914348-29-1 | |
| Record name | NSC742406 | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=742406 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 4-[2-Amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID20861337 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















