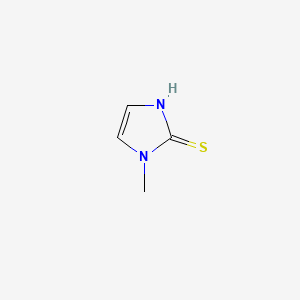
Methimazole
Overview
Description
Methimazole is a thionamide antithyroid agent used primarily to treat hyperthyroidism, a condition where the thyroid gland produces excessive thyroid hormones . It inhibits the synthesis of thyroid hormones by interfering with the enzyme thyroid peroxidase, which plays a crucial role in the production of these hormones .
Preparation Methods
Methimazole can be synthesized through several methods. One common method involves the reaction of N-methylimidazole with n-butyl lithium and sulfur powder in an organic solvent . The reaction is carried out under mild conditions, making it suitable for industrial production . Another method involves a diazotization reaction of 2-amino-1-methylimidazole followed by a Sandmeyer reaction to obtain an intermediate, which is then hydrolyzed to produce this compound .
Chemical Reactions Analysis
Methimazole undergoes various chemical reactions, including:
Scientific Research Applications
Methimazole has several scientific research applications:
Mechanism of Action
Methimazole exerts its effects by inhibiting the enzyme thyroid peroxidase, which is essential for the oxidation of iodide to iodine . This inhibition prevents the iodination of thyroglobulin, a key step in the synthesis of thyroid hormones . This compound’s sulfur moiety may also interact directly with the iron atom at the center of thyroid peroxidase’s heme molecule .
Comparison with Similar Compounds
Methimazole is often compared with other antithyroid agents such as propylthiouracil and carbimazole:
Propylthiouracil: Both this compound and propylthiouracil inhibit thyroid hormone synthesis, but this compound is generally preferred due to its lower risk of hepatotoxicity
Carbimazole: Carbimazole is a pro-drug that is converted to this compound in the body.
This compound’s unique properties, such as its potent inhibition of thyroid peroxidase and its suitability for various applications, make it a valuable compound in both medical and industrial fields.
Biological Activity
Methimazole is a thionamide antithyroid medication primarily used in the management of hyperthyroidism, particularly in conditions such as Graves' disease and toxic multinodular goiter. This article delves into the biological activity of this compound, its mechanism of action, pharmacokinetics, clinical efficacy, and safety profile based on diverse research findings.
This compound exerts its therapeutic effects by inhibiting thyroid peroxidase (TPO) , an enzyme responsible for catalyzing the iodination of tyrosine residues in thyroglobulin, which is essential for the synthesis of thyroid hormones T4 (thyroxine) and T3 (triiodothyronine). By binding to TPO, this compound disrupts the iodination process and subsequent hormone formation. The drug can act as a competitive substrate for TPO or interact directly with the enzyme's heme group, inhibiting its function .
Pharmacokinetics
- Absorption : this compound is rapidly absorbed after oral administration with an absolute bioavailability of approximately 93%. Peak plasma concentrations are typically reached within 0.25 to 4.0 hours post-dose.
- Metabolism : It undergoes hepatic metabolism primarily via cytochrome P450 enzymes (CYP1A2 and CYP2C9), resulting in the formation of 4-methyl-5-thiazolecarboxamide (MMI-4), which has weaker antithyroid activity .
- Half-life : The elimination half-life of this compound ranges from 4 to 6 hours, allowing for flexible dosing regimens.
Case Studies and Clinical Trials
-
Long-term Treatment in Graves' Disease :
- A randomized clinical trial demonstrated that continuous this compound therapy over five years resulted in an 84% remission rate , which persisted for up to four years after discontinuation .
- Another study involving 313 patients showed no significant difference in relapse rates between those treated with low (10 mg) versus high (40 mg) doses of this compound, indicating that lower doses can be equally effective .
- Comparison with Propylthiouracil :
- Juvenile Graves' Disease :
Safety Profile
This compound is generally well-tolerated, but it can lead to side effects such as rash, arthralgia, and agranulocytosis. The risk of serious adverse effects appears to be low when monitored appropriately. A case-control study found no increased risk of acute pancreatitis associated with cumulative this compound doses .
Summary Table of Key Findings
Q & A
Basic Research Questions
Q. What experimental models are standard for evaluating Methimazole’s efficacy in hyperthyroidism?
- Methodology : Use rodent models (e.g., Lgr5+ cell ablation in mice) to mimic thyroid dysfunction. Administer this compound intraperitoneally or orally, monitoring thyroid-stimulating hormone (TSH) and thyroxine (T4) levels via ELISA. Include dose-response studies (e.g., 10–30 mg/kg/day) to establish therapeutic windows. Validate results with histopathological analysis of thyroid tissue .
- Key Parameters : Serum T4/T3 reduction rate, TSH elevation, and goiter regression time.
Q. How can researchers measure this compound’s pharmacokinetics in preclinical studies?
- Methodology : Use high-performance liquid chromatography (HPLC) to quantify plasma concentrations post-administration. Calculate elimination half-life (e.g., ~5 hours in humans) and bioavailability. Incorporate radiolabeled this compound (e.g., ¹⁴C) for tissue distribution profiling. Adjust for species-specific metabolic differences (e.g., cytochrome P450 isoforms) .
Q. What synthesis routes are validated for this compound production in lab settings?
- Methodology : React aminoacetaldehyde diethyl acetal with methyl isothiocyanate under nitrogen atmosphere. Purify via recrystallization (ethanol/water). Confirm purity using melting point analysis (144–147°C), FTIR (thioamide νC=S at 680 cm⁻¹), and elemental analysis (C: 31.55%, H: 4.44%, N: 24.53%) .
Advanced Research Questions
Q. How can conflicting data on this compound’s carcinogenic potential be resolved?
- Methodology : Conduct systematic reviews (PRISMA guidelines) to assess bias in animal studies. Stratify by dose (e.g., >40 mg/kg/day in rats vs. therapeutic doses in humans) and exposure duration. Use meta-regression to identify confounders (e.g., concurrent thyroid hormone supplementation). Apply FINER criteria to evaluate study relevance and novelty .
Q. What mechanisms underlie this compound’s inhibition of flavin-containing monooxygenases (FMOs)?
- Methodology : Perform competitive inhibition assays with imipramine (FMO substrate) and varying this compound concentrations (0–500 μM). Analyze kinetics via nonlinear regression (Km, Vmax shifts). Validate with EPR spectroscopy to detect copper-thiolate complexes (g ~2.05 for rhombic symmetry) .
Q. How can epidemiological data on this compound prescriptions inform thyroid disorder trends?
- Methodology : Extract prescription data from national databases (e.g., Anatomical Therapeutic Chemical (ATC) codes H03BB02/H03BB52). Apply time-series analysis to correlate prescription rates with Graves’ disease incidence. Adjust for covariates (age, gender, geographic region) using multivariate regression .
Q. What strategies optimize this compound’s stability in copper complexation studies?
- Methodology : Synthesize copper(II)-Methimazole complexes under inert conditions. Monitor stability via thermogravimetric analysis (TGA) and UV-vis spectroscopy (λmax 640 nm for d-d transitions). Use EPR to confirm ligand coordination geometry (axial vs. rhombic symmetry) and solution-phase speciation .
Q. How do genetic ablation models clarify this compound’s role in thyroid regeneration?
- Methodology : Cross Lgr5-EGFP-IRES-CreERT2 mice with Rosa26-iDTR strains. Administer this compound post-diphtheria toxin-induced injury. Assess Lgr5+ cell proliferation via immunofluorescence (Ki67 staining) and RNA-seq for Wnt/β-catenin pathway markers. Compare recovery timelines (e.g., Days 3–31 post-injury) .
Q. Methodological Guidance for Data Contradictions
- Assessing Agranulocytosis Risk : Use case-control studies to compare this compound-treated cohorts (n > 10,000) with propylthiouracil (PTU) groups. Stratify by HLA-B*38:02 haplotype prevalence. Apply Naranjo criteria to classify adverse drug reactions .
- Resolving Enzyme Inhibition Discrepancies : Replicate experiments with controlled FMO/CYP isoform activity (e.g., anti-CYP2B1 antibodies). Use Michaelis-Menten plots to distinguish competitive vs. noncompetitive inhibition patterns .
Properties
IUPAC Name |
3-methyl-1H-imidazole-2-thione | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C4H6N2S/c1-6-3-2-5-4(6)7/h2-3H,1H3,(H,5,7) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
PMRYVIKBURPHAH-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CN1C=CNC1=S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C4H6N2S | |
| Record name | methimazole | |
| Source | Wikipedia | |
| URL | https://en.wikipedia.org/wiki/Methimazole | |
| Description | Chemical information link to Wikipedia. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID4020820 | |
| Record name | Methimazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4020820 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
114.17 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Methimazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014901 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Boiling Point |
280 °C WITH SOME DECOMP | |
| Record name | METHIMAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3361 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Solubility |
Freely soluble, Soluble in alcohol, chloroform. Sparingly soluble in ether, petroleum ether., 1 G SOL IN ABOUT 125 ML ETHER, ABOUT 4.5 ML CHLOROFORM, ABOUT 5 ML WATER, 5 ML ALCOHOL, SOL IN PYRIDINE, Slightly soluble in benzene., 1.13e+01 g/L | |
| Record name | Methimazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00763 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | METHIMAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3361 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Methimazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014901 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Methimazole's primary mechanism of action appears to be interference in an early step in thyroid hormone synthesis involving thyroid peroxidase (TPO), however the exact method through which methimazole inhibits this step is unclear. TPO, along with hydrogen peroxide, normally catalyzes the conversion of iodide to iodine and then further catalyzes the incorporation of this iodine onto the 3 and/or 5 positions of the phenol rings of tyrosine residues in thyroglobulin. These thyroglobulin molecules then degrade within thyroid follicular cells to form either thyroxine (T4) or tri-iodothyronine (T3), which are the main hormones produced by the thyroid gland. Methimazole may directly inhibit TPO, but has been shown in vivo to instead act as a competitive substrate for TPO, thus becoming iodinated itself and interfering with the iodination of thyroglobulin. Another proposed theory is that methimazole’s sulfur moiety may interact directly with the iron atom at the centre of TPO’s heme molecule, thus inhibiting its ability to iodinate tyrosine residues. Other proposed mechanisms with weaker evidence include methimazole binding directly to thyroglobulin or direct inhibition of thyroglobulin itself., ANTITHYROID DRUGS INHIBIT FORMATION OF THYROID HORMONE LARGELY BY INTERFERING WITH INCORPORATION OF IODINE INTO ORGANIC FORM. ...IMPLIES THAT THEY INTERFERE WITH OXIDATION OF IODIDE ION.../WHICH/ IS PROBABLY BROUGHT ABOUT BY PEROXIDASE. /ANTITHYROID DRUGS/, ANTITHYROID DRUGS INHIBIT THE FORMATION OF THYROID HORMONES BY ITERFERING WITH THE INCORPORATION OF IODINE INTO TYROSYL RESIDUES OF THYROGLOBULIN; THEY ALSO INHIBIT THE COUPLING OF THESE IODOTYROSYL RESIDUES TO FORM IODOTHYRONINES., Methimazole inhibits the synthesis of thyroid hormones by interfering with the incorporation of iodine into tyrosyl residues of thyroglobulin; the drug also inhibits the coupling of these iodotyrosyl residues to form iodothyronine. Although the exact mechanism(s) has not been fully elucidated, methimazole may interfere with the oxidation of iodide ion and iodotyrosyl groups. Based on limited evidence, it appears that the coupling reaction is more sensitive to antithyroid agents than the iodination reaction. Methimazole does not inhibit the action of thyroid hormones already formed and present in the thyroid gland or circulation nor does the drug interfere with the effectiveness of exogenously administered thyroid hormones. | |
| Record name | Methimazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00763 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | METHIMAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3361 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
LEAFLETS FROM ALCOHOL, WHITE TO PALE BUFF, CRYSTALLINE SUBSTANCE; STARCH-LIKE IN APPEARANCE & TO TOUCH | |
CAS No. |
60-56-0 | |
| Record name | Methimazole | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=60-56-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Methimazole [USP] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000060560 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Methimazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00763 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | methimazole | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757111 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | methimazole | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=38608 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 2H-Imidazole-2-thione, 1,3-dihydro-1-methyl- | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | Methimazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4020820 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Thiamazole | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.439 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | METHIMAZOLE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/554Z48XN5E | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | METHIMAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3361 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Methimazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014901 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
143-146 °C, 146-148 °C, 146 °C | |
| Record name | Methimazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00763 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | METHIMAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3361 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Methimazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014901 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















