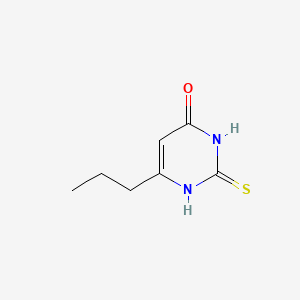
Propylthiouracil
Overview
Description
Propylthiouracil: is a thiourea antithyroid agent primarily used to manage hyperthyroidism, including conditions such as Graves’ disease and toxic multinodular goiter . It works by inhibiting the synthesis of thyroid hormones and blocking the conversion of thyroxine (T4) to triiodothyronine (T3) . This compound has been in medical use since the 1940s and is listed on the World Health Organization’s List of Essential Medicines .
Preparation Methods
Synthetic Routes and Reaction Conditions: Propylthiouracil can be synthesized through the reaction of thiourea with propyl iodide in the presence of a base . The reaction typically involves heating the reactants under reflux conditions to facilitate the formation of the desired product.
Industrial Production Methods: Industrial production of this compound involves similar synthetic routes but on a larger scale. The process includes the purification of the final product through recrystallization to ensure high purity and quality .
Chemical Reactions Analysis
Types of Reactions:
Oxidation: Propylthiouracil can undergo oxidation reactions, leading to the formation of disulfide bonds.
Reduction: It can be reduced to its corresponding thiol derivative.
Substitution: this compound can participate in substitution reactions, particularly nucleophilic substitution.
Common Reagents and Conditions:
Oxidation: Hydrogen peroxide or other oxidizing agents.
Reduction: Reducing agents such as sodium borohydride.
Substitution: Nucleophiles like amines or alcohols.
Major Products Formed:
Oxidation: Disulfide derivatives.
Reduction: Thiol derivatives.
Substitution: Various substituted thiouracil derivatives.
Scientific Research Applications
Clinical Applications
1. Management of Hyperthyroidism
- PTU is primarily indicated for patients with hyperthyroidism. It is especially useful for those who are intolerant to methimazole or require rapid control of thyroid hormone levels.
- Case Study : A study involving 19 hypothyroid patients showed that PTU effectively reduced serum T3 levels while enhancing reverse T3 concentrations .
2. Treatment of Thyroid Storm
- PTU is recommended for managing thyroid storm due to its rapid action in lowering thyroid hormone levels.
- Research Findings : A comparative study indicated that PTU may be more effective than methimazole in critically ill patients experiencing a thyroid storm, although this warrants further investigation .
3. Hepatotoxicity Concerns
- While effective, PTU has been associated with severe liver toxicity in rare cases. Reports indicate that agranulocytosis and fulminant hepatitis can occur, necessitating careful monitoring.
- Case Report : A 13-year-old girl developed severe liver dysfunction after PTU treatment for hyperthyroidism, highlighting the need for vigilance when prescribing this medication .
Research Applications
1. Endocrine Disruption Studies
- PTU has been used in various toxicological studies to evaluate its effects on endocrine function and development.
- Findings : Research has shown that low-level exposure to PTU can disrupt normal thyroid function and affect brain development in animal models .
2. Carcinogenicity Studies
- Animal studies have investigated the potential carcinogenic effects of PTU. Results indicated a significant increase in thyroid follicular-cell tumors among treated rodents.
- Data Summary : In a study with male Long Evans rats treated with PTU, 64 out of 65 rats developed thyroid tumors when combined with other agents .
Data Tables
| Study Type | Subjects | Duration | Findings |
|---|---|---|---|
| Hyperthyroidism Management | 19 hypothyroid patients | 5 days | Decreased T3 levels; increased rT3 levels |
| Thyroid Storm Comparison | Critically ill patients | N/A | Lower risk of death compared to methimazole |
| Carcinogenicity (Rats) | Long Evans rats | 1 year | High incidence of thyroid tumors (64/65) |
| Hepatotoxicity Case Report | 13-year-old girl | N/A | Severe liver dysfunction reported |
Mechanism of Action
Propylthiouracil exerts its effects by inhibiting the enzyme thyroid peroxidase, which is crucial for the iodination of tyrosine residues in thyroglobulin, a key step in the synthesis of thyroid hormones . Additionally, it inhibits the peripheral conversion of thyroxine (T4) to triiodothyronine (T3), thereby reducing the overall levels of active thyroid hormones in the body .
Comparison with Similar Compounds
Methimazole: Another antithyroid agent used to treat hyperthyroidism.
Carbimazole: A prodrug that is converted to methimazole in the body.
Comparison:
Methimazole: Unlike propylthiouracil, methimazole does not inhibit the peripheral conversion of T4 to T3.
Carbimazole: Similar to methimazole in its mechanism of action, carbimazole is converted to methimazole in the body and is used for similar indications.
This compound is unique in its dual action of inhibiting both thyroid hormone synthesis and peripheral conversion, making it particularly useful in acute thyrotoxic crises .
Biological Activity
Propylthiouracil (PTU) is a thiourea derivative primarily used as an antithyroid medication for managing hyperthyroidism, particularly in conditions like Graves' disease. This article delves into the biological activity of PTU, discussing its mechanisms of action, efficacy in various clinical settings, and potential side effects, supported by data tables and case studies.
PTU functions by inhibiting thyroid peroxidase, an enzyme crucial for the synthesis of thyroid hormones. It prevents the conversion of iodide to iodine and inhibits the incorporation of iodine into thyroglobulin, which is essential for the production of thyroxine (T4) and triiodothyronine (T3). Additionally, PTU interferes with the conversion of T4 to the more active T3, further reducing thyroid hormone levels in circulation .
Pharmacokinetics
- Absorption : PTU is well absorbed following oral administration.
- Protein Binding : Approximately 82% of PTU is bound to plasma proteins.
- Metabolism : The metabolic pathways for PTU are not fully characterized, but it is known to undergo hepatic metabolism.
- Elimination Route : The exact route of elimination remains unspecified; however, it is primarily excreted through urine .
1. Treatment during Pregnancy
A meta-analysis involving 18,948 infants assessed the safety and efficacy of PTU during pregnancy. The analysis indicated no significant increase in congenital anomalies associated with PTU use, with an odds ratio (OR) of 1.03 (95% CI: 0.84–1.25) confirming its safety profile during pregnancy .
| Outcome | Odds Ratio (OR) | 95% Confidence Interval (CI) | P-value |
|---|---|---|---|
| Congenital Anomalies | 1.03 | 0.84 – 1.25 | 0.80 |
| Neonatal Hypothyroidism | 0.55 | 0.06 – 4.92 | 0.593 |
| Hepatotoxicity | 0.34 | 0.08 – 1.48 | 0.151 |
2. Comparative Effectiveness for Thyroid Storm
A multicenter study compared PTU with methimazole in treating thyroid storm among critically ill patients. The results suggested that while both medications were effective, there may be specific scenarios where one could be preferred over the other based on patient response and side effects .
Hepatotoxicity Concerns
Despite its benefits, PTU has been associated with hepatotoxicity in some patients. A review highlighted that while most patients recover after discontinuation of PTU, there have been cases leading to severe liver damage and even fatalities .
Impact on Olfactory Function
Research has shown that PTU can impair olfactory function in both humans and animal models. In mice, PTU treatment disrupted the survival and differentiation of olfactory precursor neurons, suggesting a potential neurotoxic effect linked to its metabolism .
Q & A
Basic Research Questions
Q. What experimental design considerations are critical when evaluating the efficacy of propylthiouracil in hyperthyroidism models?
- Methodological Guidance :
- Use randomized controlled trials (RCTs) with clearly defined inclusion/exclusion criteria (e.g., thyroid hormone levels, patient demographics).
- Incorporate placebo or active comparator groups (e.g., methimazole) to isolate this compound-specific effects .
- Measure outcomes such as free thyroxine (FT4), triiodothyronine (FT3), and thyroid-stimulating hormone (TSH) at baseline and post-treatment intervals. Statistical heterogeneity in meta-analyses should be addressed using fixed- or random-effects models depending on values .
Q. How can researchers ensure reproducibility in this compound pharmacokinetic studies?
- Methodological Guidance :
- Follow USP Reference Standards for chemical identity and purity verification (e.g., USP this compound RS). Use validated analytical methods such as HPLC or LC-MS with deuterated internal standards (e.g., this compound-d5) to improve quantification accuracy .
- Report detailed protocols for sample preparation, storage conditions (e.g., protected from light at 20–25°C), and instrument calibration .
Q. What are the key steps to validate this compound’s mechanism of action in preclinical models?
- Methodological Guidance :
- Employ in vitro assays (e.g., thyroid peroxidase inhibition) and in vivo models (e.g., rat hyperthyroidism induction via thyroxine administration).
- Include dose-response studies to establish therapeutic windows and toxicity thresholds. Adverse effects like leukopenia or hepatotoxicity should be monitored via complete blood counts (CBC) and liver function tests .
Advanced Research Questions
Q. How can contradictory findings in this compound’s hematological toxicity profiles be resolved?
- Methodological Guidance :
- Conduct longitudinal studies with frequent monitoring of hematopoietic parameters (e.g., neutrophil counts, bone marrow biopsies).
- Use multivariate regression to adjust for confounding variables such as concurrent medications or autoimmune comorbidities .
- Compare results across species (e.g., rodents vs. primates) to assess translational relevance .
Q. What advanced analytical techniques are recommended for detecting this compound metabolites in biological samples?
- Methodological Guidance :
- Utilize high-resolution mass spectrometry (HR-MS) coupled with stable isotope tracing to identify metabolites like this compound sulfate.
- Apply metabolomics workflows (e.g., untargeted LC-MS/MS) to uncover novel metabolic pathways .
- Cross-validate findings with in silico models (e.g., pharmacokinetic simulations using PK-Sim) .
Q. How should researchers address ethical and methodological challenges in studying this compound’s teratogenicity?
- Methodological Guidance :
- Design animal studies adhering to IACUC guidelines, with strict controls for gestational timing and dose stratification.
- Use placental transfer assays and embryonic stem cell models to assess direct vs. indirect effects.
- For human studies, apply PICOT frameworks (Population: pregnant women; Intervention: this compound; Comparison: alternative therapies; Outcome: congenital anomalies; Timeframe: longitudinal follow-up) to structure ethical hypotheses .
Q. What strategies improve the statistical power of meta-analyses on this compound’s long-term outcomes?
- Methodological Guidance :
- Perform systematic literature reviews using PRISMA guidelines to minimize selection bias.
- Aggregate individual patient data (IPD) from RCTs to enable subgroup analyses (e.g., age, comorbidities).
- Address heterogeneity via sensitivity analyses and meta-regression .
Q. Methodological Frameworks and Tools
Q. Which frameworks are most effective for formulating hypothesis-driven research questions on this compound?
- Methodological Guidance :
- Apply FINER criteria (Feasible, Interesting, Novel, Ethical, Relevant) to prioritize questions. Example: "Is this compound’s anti-inflammatory effect in Graves’ disease mediated via NF-κB inhibition?" .
- Use PICO (Population/Problem, Intervention, Comparison, Outcome) to structure clinical queries .
Q. How can interdisciplinary approaches enhance this compound research?
- Methodological Guidance :
Properties
IUPAC Name |
6-propyl-2-sulfanylidene-1H-pyrimidin-4-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C7H10N2OS/c1-2-3-5-4-6(10)9-7(11)8-5/h4H,2-3H2,1H3,(H2,8,9,10,11) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
KNAHARQHSZJURB-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCCC1=CC(=O)NC(=S)N1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C7H10N2OS | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID5021209 | |
| Record name | 6-Propyl-2-thiouracil | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5021209 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
170.23 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Propylthiouracil | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014690 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
>25.5 [ug/mL] (The mean of the results at pH 7.4), 1 part dissolves in about 900 parts water at 20 °C, in 100 parts boiling water, in 60 parts ethyl alc, in 60 parts acetone; practically insol in ether, chloroform, benzene; freely sol in aq soln of ammonia and alkali hydroxides, In water, 1204 mg/L at 25 °C, 4.66e-01 g/L | |
| Record name | SID855783 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Propylthiouracil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00550 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Propylthiouracil | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014690 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Propylthiouracil binds to thyroid peroxidase and thereby inhibits the conversion of iodide to iodine. Thyroid peroxidase normally converts iodide to iodine (via hydrogen peroxide as a cofactor) and also catalyzes the incorporation of the resulting iodide molecule onto both the 3 and/or 5 positions of the phenol rings of tyrosines found in thyroglobulin. Thyroglobulin is degraded to produce thyroxine (T4) and tri-iodothyronine (T3), which are the main hormones produced by the thyroid gland. Therefore propylthiouracil effectively inhibits the production of new thyroid hormones., Propylthiouracil inhibits the synthesis of thyroid hormones by interfering with the incorporation of iodine into tyrosyl residues of thyroglobulin; the drug also inhibits the coupling of these iodotyrosyl residues to form iodothyronine. Although the exact mechanism(s) has not been fully elucidated, propylthiouracil may interfere with the oxidation of iodide ion and iodotyrosyl groups. Based on limited evidence it appears that the coupling reaction is more sensitive to antithyroid agents than the iodination reaction. Propylthiouracil does not inhibit the action of thyroid hormones already formed and present in the thyroid gland or circulation nor does the drug interfere with the effectiveness of exogenously administered thyroid hormones. Patients whose thyroid gland contains relatively high concentration of iodine (e.g., from prior ingestion or from administration during diagnostic radiologic procedures) may respond relatively slowly to antithyroid agents. Unlike methimazole, propylthiouracil inhibits the peripheral deiodination of thyroxine to triiodothyronine. Although the importance of this inhibition has not been established, propylthiouracil has a theoretical advantage compared with methimazole or carbimazole in patients with thyrotoxic crisis, since a decreased rate of conversion of circulating thyroxine to triiodothyronine may be clinically beneficial in these patients., The thionamide /propylthiouracil/ inhibit organification of iodide and the coupling of iodotyrosines to form hormonally active iodothyronines., Inhibit synthesis of thyroid hormone within the thyroid gland by serving as substrates for thyroid peroxidase, which catalyzes the incorporation of oxidized iodide into tyrosine residues in thyroglobulin molecules and couples iodotyrosines. This diverts iodine from the synthesis of thyroid hormones. Antithyroid agents do not interfere with the actions of exogenous thyroid hormone or inhibit the release of thyroid hormones. Therefore, stores of thyroid hormones must be depleted before clinical effects will be apparent. Antithyroid agents may also have moderating effects on the underlying immunologic abnormalities, in hyperthyroidism due to Graves' disease (toxic-diffuse goiter), but evidence on this point reported to date is inconclusive., Type I 5'-deiodinase (D1) is inhibited by ... the antithyroid drug propylthiouracil., For more Mechanism of Action (Complete) data for PROPYL THIOURACIL (9 total), please visit the HSDB record page. | |
| Record name | Propylthiouracil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00550 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Impurities |
... Small amounts of thiourea present as an impurity., Small amounts of thiourea may be present in propylthiouracil as an impurity. | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White crystalline powder of starch-like appearance to eye and to touch | |
CAS No. |
51-52-5 | |
| Record name | Propylthiouracil | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=51-52-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Propylthiouracil [USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000051525 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Propylthiouracil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00550 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | propylthiouracil | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757302 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | propylthiouracil | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=70461 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | propylthiouracil | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=6498 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 6-Propyl-2-thiouracil | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5021209 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Propylthiouracil | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.095 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PROPYLTHIOURACIL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/721M9407IY | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Propylthiouracil | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014690 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
219-221 °C, 219 °C | |
| Record name | Propylthiouracil | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00550 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | PROPYL THIOURACIL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3390 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Propylthiouracil | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014690 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















