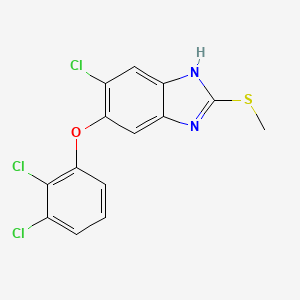
Triclabendazole
Overview
Description
Triclabendazole is a benzimidazole derivative used primarily as an anthelmintic agent to treat infections caused by liver flukes, specifically Fasciola hepatica and Fasciola gigantica . It is marketed under brand names such as Egaten and Fasinex . This compound is unique among benzimidazoles due to its efficacy against both immature and mature stages of liver flukes .
Preparation Methods
Synthetic Routes and Reaction Conditions
Triclabendazole can be synthesized using various methods. One common method involves starting with 1,2,3-trichlorobenzene, which undergoes hydrolysis in high-concentration alkali liquor to form 2,3-dichlorophenol sodium . This intermediate reacts with 4,5-dichloro-2-nitroaniline in a methylbenzene aqueous solution to form 4-chloro-5-(2,3-dichlorophenoxy)-2-nitroaniline . The nitro group is then reduced using a hydrogen catalytic transfer method, and the resulting compound undergoes methylation to yield this compound .
Another method involves using 3,4-dichloroaniline as the starting material, followed by acylation, nitration, hydrolysis, condensation, reduction with hydrazine hydrate, and ring-closure with S-methylisothiourea sulfate . This method avoids the use of hazardous reagents and high-pressure reactions, making it safer and more environmentally friendly .
Industrial Production Methods
Industrial production of this compound typically follows the synthetic routes mentioned above, with optimizations for large-scale manufacturing. The use of inexpensive and readily available starting materials, along with environmentally friendly reagents, makes the process cost-effective and suitable for large-scale production .
Chemical Reactions Analysis
Types of Reactions
Triclabendazole undergoes various chemical reactions, including:
Oxidation: This compound is metabolized in the liver to form sulfone and sulfoxide metabolites.
Reduction: The nitro group in the intermediate compound is reduced to an amine group during synthesis.
Substitution: The synthesis involves nucleophilic aromatic substitution reactions to introduce the dichlorophenoxy group.
Common Reagents and Conditions
Oxidation: Liver enzymes catalyze the oxidation of this compound to its metabolites.
Reduction: Hydrogen catalytic transfer or hydrazine hydrate is used for the reduction of the nitro group
Substitution: High-concentration alkali liquor and methylbenzene aqueous solution are used for nucleophilic aromatic substitution.
Major Products Formed
Sulfone and sulfoxide metabolites: Formed during the oxidation of this compound in the liver.
4-chloro-5-(2,3-dichlorophenoxy)-2-nitroaniline: An intermediate in the synthesis of this compound.
Scientific Research Applications
Treatment of Fascioliasis
Clinical Efficacy
Triclabendazole is the drug of choice for treating fascioliasis. Studies have demonstrated high cure rates following treatment:
- Cure Rates by Dosage :
In a retrospective cohort study involving children in Peru, the initial treatment with this compound resulted in a 55% cure rate , which decreased with subsequent treatments, indicating potential resistance or treatment failure in endemic areas .
Case Studies
- Case Study Example : In a report involving multiple cases of chronic fascioliasis, patients underwent several rounds of treatment with varying dosages of this compound. Despite initial improvements, many continued to shed Fasciola eggs post-treatment, highlighting challenges in achieving complete parasitological cure .
Potential Applications in Schistosomiasis
Recent studies have evaluated this compound's efficacy against Schistosoma mansoni , particularly in patients with co-infections of fascioliasis and schistosomiasis. A field survey showed that:
- Cure Rate for Schistosomiasis :
- In Vitro Studies : this compound demonstrated significant effects on adult schistosomes, leading to rapid destruction of their tegument within hours .
Cancer Research
Emerging research suggests that this compound may have applications in oncology:
- Mechanism of Action : It has been shown to induce pyroptosis—a form of programmed cell death—in breast cancer cells by activating caspase-3 pathways. This mechanism involves the elevation of reactive oxygen species and the regulation of apoptotic proteins .
- Tumor Volume Reduction : In xenograft models, this compound significantly reduced tumor volumes, indicating its potential as an anti-cancer agent .
Resistance and Treatment Challenges
Despite its efficacy, there are growing concerns regarding resistance to this compound:
Mechanism of Action
Triclabendazole and its metabolites are absorbed by the tegument of liver flukes, leading to a decrease in the resting membrane potential and inhibition of motility . This disruption affects the surface and ultrastructure of the flukes, ultimately leading to their death . This compound binds to beta-tubulin, preventing the polymerization of microtubules, which is essential for the survival of the flukes .
Comparison with Similar Compounds
Triclabendazole is unique among benzimidazoles due to its efficacy against both immature and mature liver flukes . Similar compounds include:
Albendazole: Used to treat a variety of parasitic infections but less effective against liver flukes.
Thiabendazole: Another benzimidazole derivative with a different mechanism of action, primarily used to treat strongyloidiasis.
Closantel: Effective against immature liver flukes but not as broad-spectrum as this compound.
This compound’s unique structure, including a chlorinated benzene ring and the absence of a carbamate group, contributes to its distinct mechanism of action and efficacy .
Biological Activity
Triclabendazole (TCBZ) is a benzimidazole derivative that is primarily used as an anthelmintic agent for the treatment of infections caused by Fasciola hepatica and Fasciola gigantica. This compound exhibits a range of biological activities, particularly against helminths, and has garnered attention for its potential applications beyond parasitic infections. This article delves into the biological activity of this compound, presenting research findings, case studies, and a comprehensive analysis of its mechanisms of action.
The precise mechanism of action of this compound remains partially understood; however, several key pathways have been identified:
- Microtubule Disruption : TCBZ disrupts microtubule formation in helminths, leading to tegumental damage and impaired motility. This effect has been demonstrated in studies involving F. hepatica, where TCBZ exposure resulted in autophagic changes and loss of tubulin immunoreactivity in the tegumental syncytium .
- Metabolite Activity : The sulphoxide metabolite of TCBZ is believed to play a significant role in its efficacy. This metabolite exhibits delayed but potent effects on parasite motility and may act through multiple targets, including inhibition of adenylate cyclase activity .
- Oxidative Phosphorylation : There is evidence suggesting that TCBZ may uncouple oxidative phosphorylation, which could contribute to its anthelmintic effects .
Efficacy in Treating Fascioliasis
This compound is notably effective against all stages of Fasciola infections. A review of clinical studies indicates high cure rates following treatment with TCBZ:
| Study | Dose Regimen | Cure Rate (%) |
|---|---|---|
| Talaie et al. (2019) | 10 mg/kg (1 dose) | 63.9% |
| Talaie et al. (2019) | 10 mg/kg (2 doses) | 68.6% |
| Talaie et al. (2019) | 10 mg/kg (3 doses) | 63.9% |
| Villegas et al. (2020) | 10 mg/kg (single dose) | 70% - 100% |
Despite these high efficacy rates, treatment failures have been documented, particularly in cases with high baseline egg counts or lower socioeconomic status . A retrospective cohort study in Peru found that only 55% of children achieved parasitologic cure after the first round of treatment, with cure rates declining significantly after multiple doses .
Case Studies
Several case studies highlight the variability in treatment outcomes with this compound:
- Case Study 1 : A 51-year-old female farmer experienced significant weight loss and abdominal pain due to Fasciola infection. After initial treatment with TCBZ (10 mg/kg), her symptoms improved; however, she relapsed after eight months and continued shedding Fasciola eggs despite subsequent treatments .
- Case Study 2 : In another case involving chronic fascioliasis, a patient received multiple courses of TCBZ but failed to achieve sustained parasitologic cure, emphasizing the need for ongoing research into resistance mechanisms and alternative treatments .
Broader Biological Activities
Recent studies have also explored the antibacterial properties of this compound. Research indicates that TCBZ exhibits activity against certain Gram-positive bacteria, including methicillin-resistant strains. In combination with other agents, it has demonstrated synergistic effects against Gram-negative pathogens like E. coli and Klebsiella pneumoniae .
Q & A
Basic Research Questions
Q. What is the primary mechanism of action of triclabendazole against Fasciola species, and how can researchers validate this experimentally?
this compound, a benzimidazole derivative, disrupts microtubule polymerization in parasitic tegumental cells, leading to impaired nutrient absorption and paralysis. However, its exact molecular targets remain debated. To validate this:
- Use in vitro assays with Fasciola hepatica larvae, measuring microtubule integrity via immunofluorescence microscopy .
- Compare dose-response curves of this compound with known microtubule-disrupting agents (e.g., albendazole) to identify overlapping pathways .
- Conduct RNA sequencing post-treatment to identify differentially expressed genes linked to cytoskeletal regulation .
Q. How can researchers design a robust in vitro model to assess this compound efficacy across Fasciola life stages?
- Experimental Design :
- Culture newly excysted juveniles (NEJs) and adult flukes in bile-containing media.
- Treat with this compound sulfoxide (active metabolite) at concentrations mimicking human pharmacokinetics (e.g., 0.1–10 µM) .
- Monitor motility inhibition (via video tracking) and tegument damage (scanning electron microscopy) over 72 hours .
Q. What methodologies are recommended for quantifying this compound and its metabolites in biological samples?
- Analytical Techniques :
- High-performance liquid chromatography (HPLC) with UV detection for plasma/serum samples, validated per ICH guidelines (linearity: 0.1–50 µg/mL; recovery >85%) .
- Liquid chromatography-mass spectrometry (LC-MS) for low-concentration metabolite detection (e.g., sulfoxide and sulfone derivatives) .
Advanced Research Questions
Q. How can conflicting data on this compound’s efficacy in resistant Fasciola strains be systematically analyzed?
- Contradiction Resolution :
- Perform comparative genomics on resistant vs. susceptible strains to identify mutations in β-tubulin or other putative targets .
- Use isothermal titration calorimetry (ITC) to measure this compound binding affinity to purified tubulin isoforms .
- Validate resistance markers via CRISPR-Cas9 editing in model organisms (e.g., Caenorhabditis elegans) .
Q. What experimental strategies can elucidate this compound’s role in modulating host-pathogen interactions beyond direct antiparasitic effects?
- Host-Centric Approaches :
- Transcriptomic profiling of human hepatocytes post-treatment to identify immunomodulatory pathways (e.g., cytokine signaling) .
- Co-culture Fasciola with host cells and measure this compound-induced changes in oxidative stress markers (e.g., glutathione levels) .
Q. How can researchers optimize this compound dosing regimens to mitigate resistance while maintaining efficacy in human trials?
- Pharmacokinetic/Pharmacodynamic (PK/PD) Modeling :
- Develop compartmental models integrating patient covariates (e.g., liver function, age) using NONMEM or Monolix .
- Simulate resistance emergence under different dosing strategies (e.g., pulsed vs. continuous) .
- Clinical Trial Design :
- Use adaptive trial designs with interim analyses to adjust dosing based on early efficacy/resistance data .
- Include coproantigen reduction as a primary endpoint to quantify parasitological response .
Q. What molecular pathways underlie this compound’s off-target effects, such as chronological lifespan extension in yeast?
- Mechanistic Studies :
- Deploy Saccharomyces cerevisiae knockout libraries to identify genes essential for this compound-induced lifespan extension (e.g., MSN2/4, RIM15) .
- Measure intracellular cAMP levels via ELISA after drug exposure to confirm adenylyl cyclase inhibition .
Q. Methodological Guidance
Q. How should researchers address ethical and logistical challenges in this compound clinical trials for fascioliasis in endemic regions?
- Participant Selection :
- Use stratified randomization based on infection severity (e.g., egg counts, imaging-confirmed liver damage) .
- Collaborate with local health authorities to ensure informed consent and post-trial access to treatment .
- Data Integrity : Implement double-blinding and third-party monitoring to reduce bias in resource-limited settings .
Q. What statistical approaches are most effective for analyzing this compound’s time-dependent efficacy in longitudinal studies?
- Mixed-Effects Models : Account for repeated measures and inter-patient variability using R’s lme4 or SAS PROC MIXED .
- Survival Analysis : Apply Cox proportional hazards models to evaluate time-to-parasitological cure .
Q. How can researchers ensure reproducibility when studying this compound’s metabolic fate in preclinical models?
Properties
IUPAC Name |
6-chloro-5-(2,3-dichlorophenoxy)-2-methylsulfanyl-1H-benzimidazole | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C14H9Cl3N2OS/c1-21-14-18-9-5-8(16)12(6-10(9)19-14)20-11-4-2-3-7(15)13(11)17/h2-6H,1H3,(H,18,19) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
NQPDXQQQCQDHHW-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CSC1=NC2=CC(=C(C=C2N1)Cl)OC3=C(C(=CC=C3)Cl)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C14H9Cl3N2OS | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID7043952 | |
| Record name | Triclabendazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7043952 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
359.7 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
0.5 [ug/mL] (The mean of the results at pH 7.4) | |
| Record name | SID50085431 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Triclabendazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB12245 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Triclabendazole is an anthelmintic agent against _Fasciola_ species. The mechanism of action against Fasciola species is not fully understood at this time. In vitro studies and animal studies suggest that triclabendazole and its active metabolites (_sulfoxide_ and _sulfone_) are absorbed by the outer body covering of the immature and mature worms, causing a reduction in the resting membrane potential, the inhibition of tubulin function as well as protein and enzyme synthesis necessary for survival. These metabolic disturbances lead to an inhibition of motility, disruption of the worm outer surface, in addition to the inhibition of spermatogenesis and egg/embryonic cells. **A note on resistance** In vitro studies, in vivo studies, as well as case reports suggest a possibility for the development of resistance to triclabendazole. The mechanism of resistance may be multifactorial and include changes in drug uptake/efflux mechanisms, target molecules, and changes in drug metabolism. The clinical significance of triclabendazole resistance in humans is not yet elucidated. | |
| Record name | Triclabendazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB12245 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
68786-66-3 | |
| Record name | Triclabendazole | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=68786-66-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Triclabendazole [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0068786663 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Triclabendazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB12245 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Triclabendazole | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759250 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Triclabendazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7043952 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 1H-Benzimidazole, 6-chloro-5-(2,3-dichlorophenoxy)-2-(methylthio) | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.127.414 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | TRICLABENDAZOLE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/4784C8E03O | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Melting Point |
189-191 | |
| Record name | Triclabendazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB12245 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















