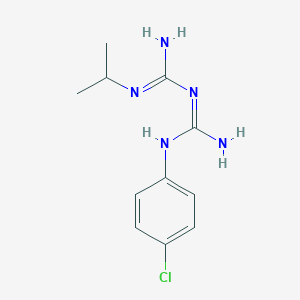
Proguanil
Overview
Description
Proguanil D6: is a deuterium-labeled derivative of this compound, an antimalarial agent. This compound is widely used for the prophylaxis and treatment of malaria caused by Plasmodium falciparum and Plasmodium vivax . The deuterium labeling in this compound D6 enhances its stability and allows for more precise pharmacokinetic studies .
Preparation Methods
Synthetic Routes and Reaction Conditions: : The synthesis of Proguanil D6 involves the incorporation of deuterium atoms into the this compound molecule. This can be achieved through various methods, including the use of deuterated reagents or solvents during the synthesis process . The reaction conditions typically involve the use of deuterated solvents such as deuterated dimethyl sulfoxide (DMSO) or deuterated ethanol, and the reactions are carried out under controlled temperatures and pressures to ensure the incorporation of deuterium atoms .
Industrial Production Methods: : Industrial production of this compound D6 follows similar synthetic routes but on a larger scale. The process involves the use of automated reactors and precise control of reaction conditions to ensure high yield and purity of the final product .
Chemical Reactions Analysis
Types of Reactions: : Proguanil D6 undergoes various chemical reactions, including:
Oxidation: This compound D6 can be oxidized to form its active metabolite, cycloguanil.
Reduction: Reduction reactions can convert this compound D6 back to its parent compound, this compound.
Substitution: Substitution reactions can occur at the chloro group, leading to the formation of different derivatives.
Common Reagents and Conditions
Reduction: Reducing agents such as sodium borohydride or lithium aluminum hydride are used under controlled conditions.
Substitution: Substitution reactions often involve nucleophiles such as amines or thiols under basic conditions.
Major Products
Cycloguanil: The major product formed from the oxidation of this compound D6.
Various Derivatives: Formed through substitution reactions.
Scientific Research Applications
Clinical Efficacy
Proguanil is most commonly administered in combination with atovaquone, marketed as Malarone. This combination has demonstrated high efficacy rates in preventing and treating malaria. For instance, a randomized placebo-controlled study showed that this combination was 100% effective in preventing malaria among children living in endemic areas . Another study highlighted that this compound effectively sensitizes malaria parasites to atovaquone, thereby enhancing treatment outcomes even in cases where resistance to other antimalarials is present .
Table 1: Efficacy Data from Clinical Studies
Case Studies
- Atovaquone/Proguanil-Induced Esophageal Ulcers : A case report documented a healthy medical student who developed esophageal ulcers after taking atovaquone/proguanil without water. This incident underscores the importance of proper administration methods for medications .
- Treatment of Imported Malaria : In a study involving travelers returning from endemic regions, atovaquone/proguanil was successfully used to treat multiple cases of P. falciparum and P. vivax malaria. The treatment was effective even in patients who had previously failed other treatments .
- Resistance Cases : A cluster of malaria cases treated with atovaquone/proguanil revealed resistance mutations in the P. falciparum genome. This highlights the ongoing challenge of drug resistance and the need for continuous monitoring and development of new treatment strategies .
Future Applications
Research is ongoing to explore additional applications of this compound beyond malaria treatment. Its potential as an antifungal agent is being investigated, particularly for use in immunocompromised patients at risk for fungal infections . Furthermore, this compound's role in combination therapies with other antimalarials continues to be a focus area due to its ability to enhance the efficacy of existing drugs.
Mechanism of Action
Proguanil D6 exerts its effects by inhibiting the enzyme dihydrofolate reductase, which is essential for the reproduction of the malaria parasite . This inhibition prevents the parasite from synthesizing DNA and replicating, thereby stopping the infection . The deuterium labeling in this compound D6 allows for more precise tracking of the drug’s distribution and metabolism in the body .
Comparison with Similar Compounds
Similar Compounds
Proguanil: The parent compound of this compound D6, used for malaria prophylaxis and treatment.
Cycloguanil: The active metabolite of this compound, formed through oxidation.
Chloroquine: Another antimalarial drug often used in combination with this compound.
Uniqueness: : this compound D6 is unique due to its deuterium labeling, which enhances its stability and allows for more precise pharmacokinetic studies compared to its non-deuterated counterparts . This makes it a valuable tool in scientific research and drug development.
Biological Activity
Proguanil is an antimalarial compound primarily used in the prevention and treatment of malaria, particularly against Plasmodium falciparum and Plasmodium vivax . Its biological activity is closely linked to its metabolism into the active metabolite cycloguanil, which exerts significant effects on the malaria parasites. This article explores the biological activity of this compound, including its mechanisms of action, pharmacokinetics, clinical efficacy, and safety profile.
This compound functions as a dihydrofolate reductase (DHFR) inhibitor , which is crucial for the biosynthesis of purines and pyrimidines necessary for DNA synthesis in malaria parasites. The inhibition of DHFR leads to a failure in nuclear division during the schizont formation phase within erythrocytes and liver cells .
Key Mechanisms:
- Inhibition of Dihydrofolate Reductase : this compound and its metabolite cycloguanil inhibit DHFR in malaria parasites, disrupting folate metabolism essential for DNA replication .
- Synergistic Action with Atovaquone : When combined with atovaquone (as in Malarone), this compound enhances the efficacy against resistant strains of malaria by targeting different pathways in the parasite's lifecycle .
Pharmacokinetics
This compound is rapidly absorbed following oral administration, with peak plasma concentrations occurring within 1-3 hours. It has a high bioavailability (approximately 75%) and is extensively metabolized in the liver to cycloguanil via cytochrome P450 enzymes (CYP2C19) .
Pharmacokinetic Parameters:
| Parameter | Value |
|---|---|
| Absorption | Rapid and well absorbed |
| Bioavailability | ~75% |
| Protein Binding | ~75% |
| Metabolism | Hepatic (CYP2C19) |
| Elimination Half-life | 8-10 hours |
Clinical Efficacy
Numerous studies have demonstrated the high efficacy of this compound, particularly when used in combination with atovaquone. A systematic review indicated that this combination therapy has a prophylactic efficacy of approximately 95.8% against malaria .
Case Studies:
- Study on Children : In a randomized controlled trial involving 320 children in Gabon, none of the children receiving atovaquone-proguanil developed positive blood smears during chemosuppression, compared to 25 cases in the placebo group (p<0.001)【6】【8】.
- Efficacy Against Resistant Strains : this compound has shown effectiveness even in regions where resistance to other antimalarial drugs is prevalent. For example, high antimalarial efficacy was observed in patients with poor metabolizer genotypes for CYP2C19【4】【5】.
Safety Profile
This compound is generally well-tolerated, with a lower incidence of adverse effects compared to alternative treatments. Common side effects include gastrointestinal disturbances such as nausea and vomiting【5】【6】. A meta-analysis reported fewer treatment-related adverse events leading to discontinuation among patients taking atovaquone-proguanil compared to those on other regimens【2】.
Adverse Effects Overview:
| Adverse Effect | Incidence (%) |
|---|---|
| Nausea | 33% |
| Vomiting | 29% |
| Abdominal Pain | Varies |
Q & A
Q. Basic: What experimental protocols are recommended for determining Proguanil hydrochloride’s solubility in Biopharmaceutics Classification System (BCS) studies?
This compound hydrochloride’s solubility should be assessed using standardized protocols aligned with BCS guidelines. Key steps include:
- pH conditions : Testing at critical pH values (1.2, 4.5, 6.8) to simulate gastrointestinal environments.
- Dose/Solubility (D/S) ratio : Ensuring the maximum D/S ratio remains <250 mL across all pH levels to confirm "high solubility" classification .
- Experimental design : Avoiding conditions that induce physicochemical incompatibilities (e.g., pH 3.3 instability) to prevent false-negative results .
- Validation : Reporting inconclusive results transparently to refine protocol accuracy .
Q. Advanced: How does CYP2C19 genetic polymorphism influence this compound’s pharmacokinetics and therapeutic efficacy?
This compound is a prodrug metabolized by CYP2C19 to its active metabolite, cycloguanil. Genetic variability impacts:
- Metabolic ratios : Poor metabolizers (PMs) with two non-functional CYP2C19 alleles exhibit urinary metabolic ratios >13, compared to <9 in heterozygotes .
- Pharmacokinetic disparities : PMs show 5–10× lower cycloguanil plasma concentrations, reducing antimalarial efficacy. Conversely, PMs have higher this compound and 4-chlorophenylbiguanide (CPB) levels, potentially increasing toxicity risks .
- Clinical implications : In populations with high CYP2C19*2/*3 allele frequencies (e.g., Vanuatu), genotyping is critical for dose optimization .
Q. Basic: What validated analytical methods are used for simultaneous quantification of this compound and its metabolites?
A robust RP-HPLC method employs:
- Column : Kromasil C18 (150 × 4.6 mm, 5 µm).
- Mobile phase : 0.1% OPA:ACN (50:50 v/v) at 1.0 mL/min flow rate.
- Detection : UV at 287 nm, yielding retention times of 2.15 min (this compound) and 2.48 min (Atovaquone).
- Validation : Linearity ranges of 25–150 µg/mL (this compound) and 62.5–375 µg/mL (Atovaquone), with recovery rates >98% . For plasma/urine, LC-MS/MS achieves LLOQs of 1 µg/L (this compound) and 0.5 µg/L (cycloguanil) .
Q. Advanced: How do contradictory efficacy outcomes arise in clinical trials of Atovaquone-Proguanil (AP) combinations?
AP’s efficacy against Plasmodium falciparum varies due to:
- PCR-adjusted endpoints : Trials differentiating recrudescence (treatment failure) from reinfection. For example, a 2014 Cameroon trial reported 9.4% PCR-adjusted failure rates for AP vs. 2.9% for artesunate-amodiaquine, though statistical power was limited .
- Regional resistance patterns : AP maintains >95% efficacy in non-African regions but shows reduced effectiveness in high-transmission areas due to prior exposure and parasite resistance .
- Study design : Small sample sizes (e.g., n=60 in Ethiopia) limit generalizability .
Q. Advanced: What methodological considerations address this compound’s folate antagonism in pregnancy-related research?
This compound inhibits dihydrofolate reductase, necessitating:
- Folic acid supplementation : Co-administration of 5 mg/day folic acid to mitigate teratogenicity risks, despite limited evidence of congenital malformations .
- Pharmacovigilance : Monitoring adverse outcomes (e.g., fetal loss, preterm birth) in trials, as seen in U.S. military data .
- Ethical constraints : Prioritizing alternative antimalarials in first-trimester studies due to residual uncertainties .
Q. Basic: What historical insights inform this compound’s mechanism of action as a causal prophylactic?
This compound’s unique prophylactic activity stems from:
- Prodrug activation : Hepatic conversion to cycloguanil, a dihydrofolate reductase inhibitor that blocks Plasmodium sporozoite development in hepatocytes .
- Mosquito-stage inhibition : Early studies demonstrated suppression of gametocyte maturation in Anopheles vectors, reducing transmission potential .
Q. Advanced: How do alternative metabolic pathways (e.g., CPB formation) impact this compound’s pharmacokinetic modeling?
CPB, a minor metabolite via CYP3A4-mediated N-dealkylation, complicates kinetic analyses by:
- Compartmental modeling : Requiring triexponential fitting to account for parallel elimination pathways (e.g., median AUCs: 3046 ng·h/mL for this compound vs. 257 ng·h/mL for CPB) .
- Tissue distribution : Higher whole-blood concentrations of this compound (5× plasma) and CPB (4× plasma) suggest erythrocyte binding, altering volume of distribution estimates .
Q. Advanced: What strategies optimize this compound dosing in CYP2C19-polymorphic populations?
Properties
Key on ui mechanism of action |
Proguanil inhibits the dihydrofolate reductase of plasmodia and thereby blocks the biosynthesis of purines and pyrimidines, which are essential for DNA synthesis and cell multiplication. This leads to failure of nuclear division at the time of schizont formation in erythrocytes and liver. |
|---|---|
CAS No. |
500-92-5 |
Molecular Formula |
C11H16ClN5 |
Molecular Weight |
259.77 g/mol |
IUPAC Name |
1-[amino-(4-chloroanilino)methylidene]-2-(1,1,1,3,3,3-hexadeuteriopropan-2-yl)guanidine |
InChI |
InChI=1S/C11H16ClN5/c1-7(2)15-10(13)17-11(14)16-9-5-3-8(12)4-6-9/h3-7H,1-2H3,(H5,13,14,15,16,17)/i1D3,2D3 |
InChI Key |
SSOLNOMRVKKSON-WFGJKAKNSA-N |
SMILES |
CC(C)N=C(N)N=C(N)NC1=CC=C(C=C1)Cl |
Isomeric SMILES |
[2H]C([2H])([2H])C(C([2H])([2H])[2H])N=C(N)N=C(N)NC1=CC=C(C=C1)Cl |
Canonical SMILES |
CC(C)N=C(N)N=C(N)NC1=CC=C(C=C1)Cl |
melting_point |
129 °C |
Key on ui other cas no. |
500-92-5 |
physical_description |
Solid |
Purity |
> 95% |
quantity |
Milligrams-Grams |
Related CAS |
637-32-1 (hydrochloride) |
solubility |
2.86e-01 g/L |
Synonyms |
Bigumal Chlorguanid Chloriguane Chloroguanide Chloroguanide Hydrochloride Hydrochloride, Chloroguanide Hydrochloride, Proguanil Paludrin Paludrine Proguanil Proguanil Hydrochloride |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















